Abstract
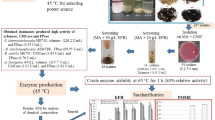
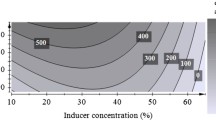
The aim of this work was to establish optimal conditions for the maximum production of endo-β-1,4 mannanases using cheaper sources. Eight thermotolerant fungal strains were isolated from garden soil and compost samples collected in and around the Gulbarga University campus, India. Two strains were selected based on their ability to produce considerable endo-β-1,4 mannanases activity while growing in liquid medium at 37 °C with locust bean gum (LBG) as the only carbon source. They were identified as Aspergillus niger gr and Aspergillus flavus gr. The experiment to evaluate the effect of different carbon sources, nitrogen sources, temperatures and initial pH of the medium on maximal enzyme production was studied. Enzyme productivity was influenced by the type of polysaccharide used as the carbon source. Copra meal defatted with n-hexane showed to be a better substrate than LBG and guar gum for endo-β-1,4 mannanases production by A. niger gr (40.011 U/ml), but for A. flavus gr (33.532 U/ml), the difference was not significant. Endo-β-1,4 mannanases produced from A. niger gr and A. flavus gr have high optimum temperature (65 and 60 °C) and good thermostability in the absence of any stabilizers (maintaining 50% of residual activity for 8 and 6 h, respectively, at 60 °C) and are stable over in a wide pH range. These new strains offer an attractive alternative source of enzymes for the food and feed processing industries.








Similar content being viewed by others
References
Harris, P. J. (2005). In K. M. Entwistle, & J. C. F. Walker (Eds.), Non-cellulosic polysaccharides in plant cell walls (pp.13–35). Christchurch, New Zealand: University of Canterbury.
Burke, R. M., & Cairney, J. W. G. (1997). Mycological Research, 101(9), 1135–1139.
Reese, T., & Shibata, Y. (1965). Canadian Journal of Microbiology, 11, 167–183.
Lin, T. C., & Chen, C. (2004). Process Biochemistry, 39, 1103–1109.
Ademark, P., Varga, A., Medve, J., Harjunpaa, V., Drakenberg, T., Tjerneld, F., et al. (1998). Journal of Biotechnology, 63, 199–210.
Regalado, C., Garcia-Almendarez, B. E., Venegas-Barrera, L. M., Tellez-Jurado, A., Rodriguez-Serrano, G., Huerta-Qchoa, S., et al. (2000). Journal of the Science of Food and Agriculture, 80, 1343–1350.
Arisan, A. I., Hodits, R., Kristufek, D., & Kubicek, C. P. (1993). Applied Microbiology and Biotechnology, 39, 58–62.
Gubitz, G. M., Hayn, M., Urbanz, G., & Steiner, W. (1996). Journal of Biotechnology, 45, 165–172.
Tomotari, M. (1990). Journal of Industrial Microbiology, 6, 263–268.
Kobayashi,Y., Echizen, R., Mada, M., & Mutai, M. (1984). In T. Mitsuoka (Ed.), Proceedings of the 4th Kiken symposium on intestinal flora (pp. 69-90). Tokyo: Japan Scientific Societies Press.
Christgan, S., Andersen, L. N., Kauppinen, S., Heldt-Hansen, H. P., & Dalboege, H. (1994). Patent Novo-Nordisk, 9425576, 10 November, 1994.
Gubitz, G. M., Lischnig, T., Stebbing, D., & Saddler, J. N. (1997). Biotechnology Letters, 19(5), 491–495.
Chandrakant, P., & Bisaria, V. S. (1998). Critical Reviews in Biotechnology, 18(4), 295–331.
Ray, S., Pubols, M. H., & Mgginnis, J. (1982). Poultry Science, 61, 488–494.
Hashimoto, Y., & Fukumoto, J. (1969). Nippon Nogei Kagakukaishi, 43(5), 317–322.
McCutchen, C. M., Duffaud, G. D., Leduc, P., Petersen, A. R. H., Tayal, A., Khan, S. A., et al. (1996). Biotechnology and Bioengineering, 52, 332–339.
Oda, Y., Komaki, T., & Tonomura, K. (1993). Food Microbiology, 10, 353–358.
Hossain, M. Z., Abe, J., & Hizukuri, S. (1996). Enzyme and Microbial Technology, 18, 95–108.
Guarte, R. C., Muhlbauer, W., & Kellert, M. (1996). Postharvest Biology and Technology, 9, 361–372.
Creswell, D. C., & Brooks, C. C. (1971). Journal of Animal Science, 33, 366–369.
NRC (1994). Washington, DC: National Academy.
Lin, T. C., & Chen, C. J. (2001). Journal of the Biomass Energy Society of China, 20(3–4), 57–68.
Nelson, N. (1944). Journal of Biological Chemistry, 153, 375–380.
Somogyi, M. (1952). Journal of Biological Chemistry, 195, 19–23.
Gibbs, P. A., Seviour, R. J., & Schmid, F. (2002). Critical Reviews in Biotechnology, 20(1), 17–48.
De Vries, R. P., & Visser, J. (2001). Microbiology and Molecular Biology Reviews, 65, 497–522.
de Souza, D. F., de Souza, C. G. M., & Peralta, R. M. (2001). Process Biochemistry, 36, 835–838.
Haltrich, D., Nidetzky, B., Kulbe, K. D., Steiner, W., & Zupancic, S. (1996). Bioresource Technology, 58, 137–161.
Lawrence, A. A. (1973). Park Ridge, NJ: Noyes Data.
Kato, K., & Matsuda, K. (1969). Agricultural and Biological Chemistry, 33(10), 1446–53.
Santoso, U., Kubok, K., Ota, T., Tadokoro, T., & Maekawa, A. (1996). Food Chemistry, 57(2), 299–304.
Rajoka, M. I., Akhtar, M. W., Hanif, A., & Khalid, A. M. (2006). World Journal of Microbiology & Biotechnology, 22, 991–998.
Christov, L. P., Szakacs, G., & Balakrishnan, H. (1999). Process Biochemistry, 34, 511–517.
Puchart, V., Vrsanska, M., Svoboda, P., Pohl, J., Ogel, Z. B., & Biely, P. (2004). Biochimica et Biophysica Acta, 1674, 239–250.
Barbasgaard, Hansen, H. P., & Diderichsen, B. (1992). Applied Microbiology and Biotechnology, 36, 569–572.
Acknowledgments
One of the authors, Mr. Naganagouda V Kote, wishes to thank the Indian Council of Medical Research (ICMR), New-Delhi, India for its financial support in the form of a Senior Research Fellowship (SRF) during this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kote, N.V., Patil, A.G.G. & Mulimani, V.H. Optimization of the Production of Thermostable endo-β-1,4 Mannanases from a Newly Isolated Aspergillus niger gr and Aspergillus flavus gr . Appl Biochem Biotechnol 152, 213–223 (2009). https://doi.org/10.1007/s12010-008-8250-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-008-8250-z