Abstract
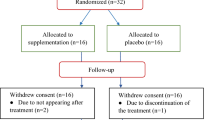
Previous studies have shown that n-3 polyunsaturated fatty acids n-3 (n-3 PUFA) have several anticancer effects, especially attributed to their ability to modulate a variety of genomic and immune responses. In this context, this randomized, prospective, controlled clinical trial was conducted in order to check whether supplementation of 2 g/day of fish oil for 9 weeks alters the production of inflammatory markers, the plasma fatty acid profile and the nutritional status in patients with colorectal cancer (CRC). Eleven adults with CRC in chemotherapy were randomized into two groups: (a) supplemented (SG) daily with 2 g/day of encapsulated fish oil [providing 600 mg/day of eicosapentaenoic acid (EPA) + docosahexaenoic acid (DHA)] for 9 weeks (n = 6), and (b) control (CG) (n = 5). All outcomes were evaluated on the day before the first chemotherapy session and 9 weeks later. Plasma TNF-α, IL-1β, IL-10 and IL-17A, the pro/anti-inflammatory balance (ratio TNF-α/IL-10 and IL-1β/IL10) and serum albumin, showed no significant changes between times and study groups (p > 0.05). C-reactive protein (CRP) and the CRP/albumin ratio showed opposite behavior in groups, significantly reducing their values in SG (p < 0.05). Plasma proportions of EPA and DHA increased 1.8 and 1.4 times, respectively, while the ARA reduced approximately 0.6 times with the supplementation (9 weeks vs baseline, p < 0.05). Patients from SG gained 1.2 kg (median) while the CG lost −0.5 kg (median) during the 9 weeks of chemotherapy (p = 0.72). These results demonstrate that 2 g/day of fish oil for 9 weeks of chemotherapy improves CRP values, CRP/albumin status, plasma fatty acid profile and potentially prevents weight loss during treatment.



Similar content being viewed by others
Abbreviations
- ARA:
-
Arachidonic acid
- BMI:
-
Body mass index
- CG:
-
Control group
- COX:
-
Cyclooxygenase
- CRC:
-
Colorectal cancer
- CRP:
-
C-Reactive protein
- DHA:
-
Docosahexaenoic acid
- ELISA:
-
Enzyme-linked immunosorbent assay
- EPA:
-
Eicosapentaenoic acid
- HPLC:
-
High pressure liquid chromatography
- IL:
-
Interleukin
- NF-κB:
-
Nuclear factor κB
- PG:
-
Prostaglandin
- PINI:
-
Prognostic inflammatory and nutritional index
- PPAR:
-
Peroxisome proliferator-activated receptors
- PUFA:
-
Polyunsaturated fatty acid
- SC:
-
Santa Catarina
- SG:
-
Supplemented group
- TNF:
-
Tumor necrosis factor
- TNM:
-
Tumor staging
- VB:
-
Visual basic
References
Connolly JM, Coleman M, Rose DP (1997) Effects of dietary fatty acids on DU145 human prostate cancer cell growth in athymic nude mice. Nutr Cancer 29:114–119
Connolly JM, Gilhooly EM, Rose DP (1999) Effects of reduced dietary linoleic acid intake, alone or combined with an algae source of docosahexaenoic acid on MDA-MB-231 breast cancer cell growth and apoptosis in nude mice. Nutr Cancer 35:44–49
Calder PC, Davis J, Yaqoob P et al (1998) Dietary fish oil suppresses human colon tumor growth in athymic mice. Clin Sci 94:303–311
Chen ZY, Istfan NW (2000) Docohexaenoic acid is a potent inducer of apoptosis in HT-29 colon cancer cells. Prostaglandins Leukot Essent Fatty Acids 63:301–308
Jordan A, Stein J (2003) Effect of an omega-3 fatty acid containing lipid emulsion alone and in combination with 5-fluorouracil (5-FU) on growth of the colon cancer cell line Caco-2. Eur J Nutr 42:324–331
DeGraffenried LA, Friedrichs WE, Fulcher L et al (2003) Eicosapentaenoic acid restores tamoxifen sensitivity in breast cancer cells with high Akt activity. Ann Oncol 14:969–970
Feng Z, Hu W, Marnett LJ et al (2006) Malondialdehyde, a major endogenous lipid peroxidation product, sensitizes human cells to UV- and BPDE-induced killing and mutagenesis through inhibition of nucleotide excision repair. Mutat Res 601:125–136
Moses AW, Slater C, Preston T et al (2004) Reduced total energy expenditure and physical activity in cachexia patients with pancreatic cancer can be modulated by an energy and protein dense oral supplement enriched with n-3 fatty acids. Br J Cancer 90:996–1002
Colomer R, Moreno-Nogueira JM, García-Luna PP et al (2007) n-3 fatty acids, cancer and cachexia: a systematic review of the literature. Br J Nutr 97:823–831
Bonatto SR, Oliveira HP, Nunes EA et al (2012) Fish oil supplementation improves neutrophil function during cancer chemotherapy. Lipids 47:383–389
Cury-Boaventura MF, Torrinhas RS, Godoy AB et al (2012) Human leukocyte death after a preoperative infusion of medium/long-chain triglyceride and fish oil parenteral emulsions: a randomized study in gastrointestinal cancer patients. JPEN J Parenter Enteral Nutr 36(6):677–684
Cockbain AJ, Toogood GJ, Hull MA (2012) Omega-3 polyunsaturated fatty acids for the treatment and prevention of colorectal cancer. Gut 61:135–149
Janakiram NB, Mohammed A, Rao CV (2011) Role of lipoxins, resolvins, and other bioactive lipids in colon and pancreatic cancer. Cancer Metastasis Rev 30:507–523
Hull MA (2011) Omega-3 polyunsaturated fatty acids. Best Pract Res Clin Gastroenterol 25:547–554
Yang E, Chan D, Felix E et al (2004) Formation and antiproliferative effect of prostaglandin E3 from eicosapentaenoic acid in human lung cancer cells. J Lipid Res 45:1030–1039
Chapkin RS, Kim W, Lupton JR et al (2009) Dietary docosahexaenoic and eicosapentaenoic acid: emerging mediators of inflammation. Prostaglandins Leukot Essent Fatty Acids 81:187–191
Szkaradkiewicz A, Marciniak R, Chudzicka-Strugała I et al (2009) Proinflammatory cytokines and IL-10 in inflammatory bowel disease and colorectal cancer patients. Arch Immunol Ther Exp 57:291–294
Chung YC, Chang YF (2003) Serum C-reactive protein correlates with survival in colorectal cancer patients but is not an independent prognostic indicator. Eur J Gastroenterol Hepatol 15:369–373
Rickham PP (1964) Human experimentation. Code of ethics of the World Medical Association. Declaration of Helsinki. Br Med J 2:0–177
Peltola H, Valmari P (1985) Serum C-reactive protein as detector of pretreated childhood bacterial meningitis. Neurology 35:251–253
Doumas BT, Watson WA, Biggs HG (1971) Albumin standards and the measurement of serum albumin with bromocresol green. Clin Chim Acta 31:87–96
Corrêa CR, Angelili AO, Camargo NR et al (2002) Comparação entre a relação PCR/albumina e o índice prognóstico inflamatório nutricional (IPIN). Jornal Brasileiro de Patologia e Medicina Laboratorial 38:183–190
Lohman TG, Roche AF, Martorell R (1988) Anthropometric standardization reference manual. Human Kinetics, Champaign
World Health Organization (1998) Obesity: preventing and managing the global epidemic. Report of a WHO consultation on obesity. WHO, Geneva
Detsky AS, McLaughlin JR, Baker JP et al (1987) What is subjective global assessment of nutritional status? J Parenter Enter Nutr 11:8–13
Durnin JV, Womersley J (1974) Body fat assessed from total body density and its estimation from skinfold thickness: measurements on 481 men and women aged from 16 to 72 years. Br J Nutr 32:77–97
Braga M, Gianotti L, Vignali A et al (2002) Preoperative oral arginine and fatty acid supplementation improves the immunometabolic host response and outcome after colorectal resection for cancer. Surgery 132:805–814
Matsuda A, Furukawa K, Takasaki H et al (2006) Preoperative oral immune-enhancing nutritional supplementation corrects Th1/Th2 imbalance in patients undergoing elective surgery for colorectal cancer. Dis Colon Rectum 49:507–516
Read JA, Beale PJ, Volker DH et al (2007) Nutrition intervention using an eicosapenatenoic acid (EPA)-containing supplement in patients with advanced colorectal cancer. Effects on nutritional and inflammatory status: a phase II trial. Support Care Cancer 15:301–307
Liang B, Wang S, Ye YJ et al (2008) Impact of postoperative omega-3 fatty acid-supplemented parenteral nutrition on clinical outcomes and immunomodulations in colorectal cancer patients. World J Gastroenterol 21:2434–2439
Silva JA, Trindade EB, Fabre ME et al (2012) Fish oil supplement alters markers of inflammatory and nutritional status in colorectal cancer patients. Nutr Cancer 64:267–273
Zhu MW, Tang DN, Hou J et al (2012) Impact of fish oil enriched total parenteral nutrition on elderly patients after colorectal cancer surgery. Chin Med J 125:178–181
Purasiri P, Murray A, Richardson S et al (1994) Modulation of cytokine in vivo by dietary essential fatty acids in patients with colorectal cancer. Clin Sci 87:711–717
Erlinger TP, Platz EA, Rifai N et al (2004) C-reactive protein and the risk of incident colorectal cancer. JAMA 291:585–590
Gunter MJ, Stolzenberg-Solomon R, Cross AJ et al (2006) A prospective study of serum C-reactive protein and colorectal cancer risk in men. Cancer Res 66:2483–2487
Il’yasova D, Colbert LH, Harris TB et al (2005) Circulating levels of inflammatory markers and cancer risk in the health aging and body composition cohort. Cancer Epidemiol Biomarkers Prev 14:2413–2418
Shiu YC, Lin JK, Huang CJ et al (2008) Is C-reactive protein a prognostic factor of colorectal cancer? Dis Colon Rectum 51:443–449
Ingembleek Y, Carpentier Y (1984) A prognostic inflammatory and nutritional index scoring critically ill patients. Internat J Vit Nutr Res 55:91–101
Wu S, Rhee KJ, Albesiano E et al (2009) A human colonic commensal promotes colon tumorigenesis via activation of T helper type 17 T cell responses. Nat Med 15:1016–1022
Chae WJ, Gibson TF, Zelterman D et al (2010) Ablation of IL-17A abrogates progression of spontaneous intestinal tumorigenesis. PNAS 107:5540–5544
Chae WJ, Bothwell AL (2011) IL-17 and intestinal tumorigenesis. Drugs Discov Today: Dis Mech 8:e79–e83
Sánchez LK, Ugalde ME, Motola KD et al (2012) Gastrointestinal symptoms and weight loss in cancer patients receiving chemotherapy. Br J Nutr 12:1–4
Furukawa T, Tashiro H, Yamamori K et al (1999) Effects of soybean oil emulsion and eicosapentaenoic acid on stress response and immune function after a severely stressful operation. Ann Surg 229:255–261
Acknowledgments
We are grateful to the Post-Graduate Program in Nutrition—Federal University of Santa Catarina, Brazil; the Fellowship Program Social Demand/Coordination of Improvement of Higher Education Personnel (CAPES) with a scholarship grant to the senior author; and to the Foundation that Supports Research and Innovation in the State of Santa Catarina (FAPESC) by financing evaluations of inflammatory markers. Thanks also to Phytomare for the donation of the dietary supplements.
Conflict of interest
The authors state that there was no conflict of interests in any step of this study.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Mocellin, M.C., Pastore e Silva, J.A., Camargo, C.Q. et al. Fish Oil Decreases C-Reactive Protein/Albumin Ratio Improving Nutritional Prognosis and Plasma Fatty Acid Profile in Colorectal Cancer Patients. Lipids 48, 879–888 (2013). https://doi.org/10.1007/s11745-013-3816-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11745-013-3816-0