Abstract
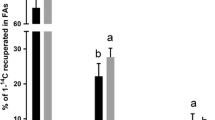
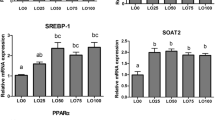
Atlantic salmon (Salmo salar) (90 g) were fed four different diets for 21 weeks (final weight 344 g). The levels of n-3 highly unsaturated fatty acids (HUFA) ranged from 11% of the total fatty acids (FA) in the low n-3 diet to 21% in the intermediate n-3 diet, to 55 and 58% in the high n-3 diets. The high n-3 diets were enriched with either docosahexaenoic acid (DHA) or eicosapentaenoic acid (EPA). Increasing dietary levels of n-3 HUFA led to increasing percentages (from 31 to 52%) of these FA in liver lipids. The group fed the highest level of DHA had higher expressions of peroxisome proliferator-activated receptor (PPAR) β and the FA β-oxidation genes acyl-CoA oxidase (ACO) and carnitine palmitoyltransferase (CPT)-II, compared to the low n-3 groups. The high n-3 groups had reduced activity of mitochondrial cytochrome c oxidase and β-oxidation capacity, together with increased activities of superoxide dismutase (SOD) and caspase-3 activities. In the group fed the highest level of n-3 HUFA, decreased percentages of major phospholipids (PL) in the mitochondrial and microsomal membranes of the liver were also apparent. The percentage of mitochondrial cardiolipin (Ptd2Gro) was 3.1 in the highest n-3 group compared to 6.6 in the intermediate group. These data clearly show an increased incidence of oxidative stress in the liver of fish fed the high n-3 diets.






Similar content being viewed by others
Abbreviations
- ACO:
-
Acyl-CoA oxidase
- BSA:
-
Bovine serum albumin
- CoA:
-
Coenzyme A
- CPT:
-
Carnitine palmitoyltransferase
- DHA:
-
Docosahexaenoic acid
- DTT:
-
Dithiothreitol
- EDTA:
-
Ethylene diamine tetra-acetic acid
- EF1-αβ :
-
Elongation factor 1-α beta isoform
- EGTA:
-
Ethylene glycol tetra-acetic acid
- EPA:
-
Eicosapentanoic acid
- FA:
-
Fatty acid
- FAD:
-
Flavin adenine dinucleotide
- FBS:
-
Fetal bovine serum
- FO:
-
Fish oil
- HPTLC:
-
High-performance thin-layer chromatography
- HUFA:
-
Highly unsaturated fatty acid
- L-15:
-
Leibowitz-15 medium
- NAD:
-
Nicotinamide adenine dinucleotide
- PBS:
-
Phosphate-buffered saline
- PL:
-
Phospholipid
- PPAR:
-
Peroxisome proliferator-activated receptor
- PtdCho:
-
Phosphatidyl-choline
- PtdEtn:
-
Phosphatidyl-ethanolamine
- Ptd2Gro:
-
Cardiolipin
- PtdIns:
-
Phosphatidyl-inositol
- RO:
-
Rapeseed oil
- ROS:
-
Reactive oxygen species
- SOD:
-
Superoxide dismutase
- TAG:
-
Triacylglycerol
References
Sargent JR, Tocher DR, Bell JG (2002) The lipids. In: Halver JE, Hardy RW (eds) Fish nutrition. Academic Press, New York, pp 181–257
Stubhaug I, Froyland L, Torstensen BE (2005) Beta-Oxidation capacity of red and white muscle and liver in Atlantic salmon (Salmo salar L.)—effects of increasing dietary rapeseed oil and olive oil to replace capelin oil. Lipids 40:39–47
Stubhaug I, Lie O, Torstensen BE (2006) Beta-oxidation capacity in liver increases during parr-smolt transformation of Atlantic salmon fed vegetable oil and fish oil. J Fish Biol 69:504–517
Stubhaug I, Lie O, Torstensen BE (2007) Fatty acid productive value and beta-oxidation capacity in Atlantic salmon (Salmo salar L.) fed on different lipid sources along the whole growth period. Aquacult Nutr 13:145–155
Vegusdal A, Gjoen T, Berge RK, Thomassen MS, Ruyter B (2005) Effect of 18:1n–9, 20:5n–3, and 22:6n–3 on lipid accumulation and secretion by Atlantic salmon hepatocytes. Lipids 40:477–486
Willumsen N, Vaagenes H, Lie O, Rustan AC, Berge RK (1996) Eicosapentaenoic acid, but not docosahexaenoic acid, increases mitochondrial fatty acid oxidation and upregulates 2,4-dienoyl-CoA reductase gene expression in rats. Lipids 31:579–592
Vamecq J, Vallee L, de la Porte PL, Fontaine M, de Craemer D, van den Branden C, Lafont H, Grataroli R, Nalbone G (1993) Effect of various n-3/n-6 fatty acid ratio contents of high fat diets on rat liver and heart peroxisomal and mitochondrial [beta]-oxidation. Biochim Biophys Acta 1170:151–156
Kjaer MA, Vegusdal A, Gjoen T, Rustan AC, Todorcevic M, Ruyter B (2008) Effect of rapeseed oil and dietary n-3 fatty acids on triacylglycerol synthesis and secretion in Atlantic salmon hepatocytes. Biochim Biophys Acta 1781:112–122
Jordal AE, Torstensen BE, Tsoi S, Tocher DR, Lall SP, Douglas SE (2005) Dietary rapeseed oil affects the expression of genes involved in hepatic lipid metabolism in Atlantic salmon (Salmo salar L.). J Nutr 135:2355–2361
Bell JG, McEvoy J, Tocher DR, McGhee F, Campbell PJ, Sargent JR (2001) Replacement of fish oil with rapeseed oil in diets of Atlantic salmon (Salmo salar) affects tissue lipid compositions and hepatocyte fatty acid metabolism. J Nutr 131:1535–1543
Moya-Falcon C, Thomassen MS, Jakobsen JV, Ruyter B (2005) Effects of dietary supplementation of rapeseed oil on metabolism of [1–14C]18:1n–9, [1–14C]20:3n-6, and [1–14C]20:4n-3 in Atlantic salmon hepatocytes. Lipids 40:709–717
Ruyter B, Thomassen MS (1999) Metabolism of n-3 and n-6 fatty acids in Atlantic salmon liver: stimulation by essential fatty acid deficiency. Lipids 34:1167–1176
Tocher DR, Bell JG, Dick JR, Crampton VO (2003) Effects of dietary vegetable oil on Atlantic salmon hepatocyte fatty acid desaturation and liver fatty acid compositions. Lipids 38:723–732
Zheng X, Tocher DR, Dickson CA, Bell JG, Teale AJ (2004) Effects of diets containing vegetable oil on expression of genes involved in highly unsaturated fatty acid biosynthesis in liver of Atlantic salmon (Salmo salar). Aquaculture 236:467–483
Gray J (1978) Measurement of lipid oxidation: a review. J Am Oil Chem Soc 55:539–546
Nilsson A, Hjelte L, Strandvik B (1992) Incorporation of dietary [14C]arachidonic acid and [3H]eicosapentaenoic acid into tissue lipids during absorption of a fish oil emulsion. J Lipid Res 33:1295–1305
Winston GW, Di Giulio RT (1991) Prooxidant and antioxidant mechanisms in aquatic organisms. Aquat Toxicol 19:137–161
Halliwell B, Gutteridge J (1999) Free radicals in biology and medicine, 3 edn. Oxford University Press, Oxford
Schlame M, Rua D, Greenberg ML (2000) The biosynthesis and functional role of cardiolipin. Prog Lipid Res 39:257–288
Paradies G, Petrosillo G, Pistolese M, Di Venosa N, Serena D, Ruggiero FM (1999) Lipid peroxidation and alterations to oxidative metabolism in mitochondria isolated from rat heart subjected to ischemia and reperfusion. Free Radical Bio Med 27:42–50
Ostrander DB, Sparagna GC, Amoscato AA, McMillin JB, Dowhan W (2001) Decreased cardiolipin synthesis corresponds with cytochrome c release in palmitate-induced cardiomyocyte apoptosis. J Biol Chem 276:38061–38067
Petrosillo G, Ruggiero FM, Pistolese M, Paradies G (2001) Reactive oxygen species generated from the mitochondrial electron transport chain induce cytochrome c dissociation from beef-heart submitochondrial particles via cardiolipin peroxidation. Possible role in the apoptosis. FEBS Lett 509:435–438
Seglen PO (1976) Preparation of isolated rat liver cells. Methods Cell Biol 13:29–83
Dannevig BH, Berg T (1985) Endocytosis of galactose-terminated glycoproteins by isolated liver cells of the rainbow trout (Salmo gairdneri). Comp Biochem Physiol B 82:683–688
Folch J, Lees M, Sloane Stanley GH (1957) A simple method for the isolation and purification of total lipids from animal tissues. J Biol Chem 226:497–509
Mason ME, Waller GR (1964) Dimethoxypropane induced transesterification of fats + oils in preparation of methyl esters for gas chromatographic analysis. Anal Chem 36:583–586
Hoshi M, Williams M, Kishimoto Y (1973) Esterification of fatty acids at room temperature by chloroform-methanolic HCl-cupric acetate. J Lipid Res 14:599–601
Vitiello F, Zanetta JP (1978) Thin-layer chromatography of phospholipids. J Chromatogr 166:637–640
De Duve C, Pressman BC, Gianetto R, Wattiaux R, Appelmans F (1955) Tissue fractionation studies. 6. Intracellular distribution patterns of enzymes in rat-liver tissue. Biochem J 60:604–617
Storrie B, Madden EA (1990) Isolation of subcellular organelles. Methods Enzymol 182:203–225
Baudhuin P, Beaufay H, Rahman-Li Y, Sellinger OZ, Wattiaux R, Jacques P, De DC (1964) Tissue fractionation studies. 17. Intracellular distribution of monoamine oxidase, aspartate aminotransferase, alanine aminotransferase, D-amino acid oxidase and catalase in rat-liver tissue. Biochem J 92:179–184
Bergmeyer HU, Gawehn K, Grassl M (1974) Methods of enzymatic analysis, 2nd edn. Academic Press, Inc., New York, pp 495–496
Beaufay H, Amar-Costesec A, Feytmans E, Thines-Sempoux D, Wibo M, Robbi M, Berthet J (1974) Analytical study of microsomes and isolated subcellular membranes from rat liver: I. Biochemical methods. J Cell Biol 61:188–200
Lazarow PB (1981) Assay of peroxisomal beta-oxidation of fatty acids. Methods Enzymol 72:315–319
Small GM, Burdett K, Connock MJ (1985) A sensitive spectrophotometric assay for peroxisomal acyl-CoA oxidase. Biochem J 227:205–210
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Peterson GL (1977) A simplification of the protein assay method of Lowry et al. which is more generally applicable. Anal Biochem 83:346–356
Simon P (2003) Q-Gene: processing quantitative real-time RT-PCR data. Bioinformatics 19:1439–1440
Olsvik P, Lie K, Jordal AE, Nilsen T, Hordvik I (2005) Evaluation of potential reference genes in real-time RT-PCR studies of Atlantic salmon. BMC Mol Biol 6:21–29
Ruyter B, Rosjo C, Einen O, Thomassen MS (2000) Essential fatty acids in Atlantic salmon: time course of changes in fatty acid composition of liver, blood and carcass induced by a diet deficient in n-3 and n-6 fatty acids. Aquacult Nutr 6:109–117
Mannaerts GP, Debeer LJ, Thomas J, De Schepper PJ (1979) Mitochondrial and peroxisomal fatty acid oxidation in liver homogenates and isolated hepatocytes from control and clofibrate-treated rats. J Biol Chem 254:4585–4595
Madsen L, Froyland L, Dyroy E, Helland K, Berge RK (1998) Docosahexaenoic and eicosapentaenoic acids are differently metabolized in rat liver during mitochondria and peroxisome proliferation. J Lipid Res 39:583–593
Vladimirov YA, Olenev VI, Suslova TB, Cheremisina ZP (1980) Lipid peroxidation in mitochondrial membrane. Adv Lipid Res 17:173–249
Nomura K, Imai H, Koumura T, Kobayashi T, Nakagawa Y (2000) Mitochondrial phospholipid hydroperoxide glutathione peroxidase inhibits the release of cytochrome c from mitochondria by suppressing the peroxidation of cardiolipin in hypoglycaemia-induced apoptosis. Biochem J 351:183–193
Shimizu S, Eguchi Y, Kamiike W, Waguri S, Uchiyama Y, Matsuda H, Tsujimoto Y (1996) Retardation of chemical hypoxia-induced necrotic cell death by Bcl-2 and ICE inhibitors: possible involvement of common mediators in apoptotic and necrotic signal transductions. Oncogene 12:2045–2050
Otsuki Y, Li Z, Shibata MA (2003) Apoptotic detection methods—from morphology to gene. Prog Histochem Cytochem 38:275–339
Du ZY, Clouet P, Zheng WH, Degrace P, Tian LX, Liu YJ (2006) Biochemical hepatic alterations and body lipid composition in the herbivorous grass carp (Ctenopharyngodon idella) fed high-fat diets. Br J Nutr 95:905–915
Du ZY, Clouet P, Huang LM, Degrace P, Zheng WH, He JG, Tian LX, Liu YJ (2008) Utilization of different dietary lipid sources at high level in herbivorous grass carp (Ctenopharyngodon idella): mechanism related to hepatic fatty acid oxidation. Aquacult Nutr 14:77–92
Stephan G, Guillaume J, Lamour F (1995) Lipid peroxidation in turbot (Scophthalmus maximus) tissue: effect of dietary vitamin E and dietary n-6 or n-3 polyunsaturated fatty acids. Aquaculture 130:251–268
Puangkaew J, Kiron V, Satoh S, Watanabe T (2005) Antioxidant defense of rainbow trout (Oncorhynchus mykiss) in relation to dietary n-3 highly unsaturated fatty acids and vitamin E contents. Comp Biochem Physiol C 140:187–196
Roem AJ, Kohler CC, Stickney RR (1990) Vitamin E requirements of the blue tilapia, Oreochromis aureus (Steindachner), in relation to dietary lipid level. Aquaculture 87:155–164
Watanabe T, Takeuchi T, Wada M, Uehara R (1981) The relationship between dietary-lipid levels and alpha-tocopherol requirement of rainbow-trout. Bull Jpn Soc Sci Fish 47:1463–1471
Waagboe R, Sandnes K, Sandvin A, Lie O (1991) Feeding three levels of N-3 polyunsaturated fatty acids at two levels of vitamin E to Atlantic salmon (Salmo salar). Growth and chemical composition, Fiskeridir Skr (Ernaering) vol 4, no 1, pp 51–63, 51–63
Foretz M, Foufelle F, Ferré P (1999) Polyunsaturated fatty acids inhibit fatty acid synthase and spot-14-protein gene expression in cultured rat hepatocytes by a peroxidative mechanism. Biochem J 341:371–376
Mikkelsen L, Hansen HS, Grunnet N, Dich J (1993) Inhibition of fatty acid synthesis in rat hepatocytes by exogenous polyunsaturated fatty acids is caused by lipid peroxidation. Biochim Biophys Acta 1166:99–104
Eder K, Kirchgessner M (1998) The effect of dietary vitamin E supply and a moderately oxidized oil on activities of hepatic lipogenic enzymes in rats. Lipids 33:277–283
Kim H, Choi S, Lee HJ, Lee JH, Choi H (2003) Suppression of fatty acid synthase by dietary polyunsaturated fatty acids is mediated by fat itself, not by peroxidative mechanism. J Biochem Mol Biol 36:258–264
Mundheim H, Aksnes A, Hope B (2004) Growth, feed efficiency and digestibility in salmon (Salmo salar L.) fed different dietary proportions of vegetable protein sources in combination with two fish meal qualities. Aquaculture 237:315–331
Acknowledgments
We wish to thank Inger Ø. Kristiansen and Målfrid T. Bjerke (Nofima) for skillful technical assistance. This work was carried out with support from the Norwegian Research Council.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Kjær, M.A., Todorčević, M., Torstensen, B.E. et al. Dietary n-3 HUFA Affects Mitochondrial Fatty Acid β-Oxidation Capacity and Susceptibility to Oxidative Stress in Atlantic Salmon. Lipids 43, 813–827 (2008). https://doi.org/10.1007/s11745-008-3208-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11745-008-3208-z