Abstract
Objectives
This study seeks to evaluate assessment of geriatric frailty and nutritional status in predicting postoperative mortality in gastric cancer surgery.
Methods
Preoperatively, patients operated for gastric adenocarcinoma underwent assessment of Groningen Frailty Indicator (GFI) and Short Nutritional Assessment Questionnaire (SNAQ). We studied retrospectively whether these scores were associated with in-hospital mortality.
Results
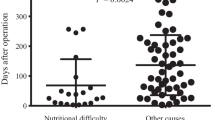
From 2005 to September 2012 180 patients underwent surgery with an overall mortality of 8.3 %. Patients with a GFI ≥ 3 (n = 30, 24 %) had a mortality rate of 23.3 % versus 5.2 % in the lower GFI group (OR 4.0, 95%CI 1.1–14.1, P = 0.03). For patients who underwent surgery with curative intent (n = 125), this was 27.3 % for patients with GFI ≥ 3 (n = 22, 18 %) versus 5.7 % with GFI < 3 (OR 4.6, 95 % CI 1.0–20.9, P = 0.05). SNAQ ≥ 1 (n = 98, 61 %) was associated with a mortality rate of 13.3 % versus 3.2 % in patients with SNAQ = 0 (OR 5.1, 95 % CI 1.1–23.8, P = 0.04). Given odds ratios are corrected in multivariate analyses for age, neoadjuvant chemotherapy, type of surgery, tumor stage and ASA classification.
Conclusions
This study shows a significant relationship between gastric cancer surgical mortality and geriatric frailty as well as nutritional status using a simple questionnaire. This may have implications in preoperative decision making in selecting patients who optimally benefit from surgery.

Similar content being viewed by others
References
Jemal, A., F. Bray, M.M. Center, J. Ferlay, E. Ward, and D. Forman, Global cancer statistics. CA Cancer J Clin, 2011. 61(2): p. 69-90.
Hartgrink, H.H., C.J. van de Velde, H. Putter, J.J. Bonenkamp, E. Klein Kranenbarg, I. Songun, K. Welvaart, J.H. van Krieken, S. Meijer, J.T. Plukker, P.J. van Elk, H. Obertop, D.J. Gouma, J.J. van Lanschot, C.W. Taat, P.W. de Graaf, M.F. von Meyenfeldt, H. Tilanus, and M. Sasako, Extended lymph node dissection for gastric cancer: who may benefit? Final results of the randomized Dutch gastric cancer group trial. J Clin Oncol, 2004. 22(11): p. 2069-77.
Saif, M.W., N. Makrilia, A. Zalonis, M. Merikas, and K. Syrigos, Gastric cancer in the elderly: an overview. Eur J Surg Oncol, 2010. 36(8): p. 709-17.
Hara, H., H. Isozaki, E. Nomura, K. Fujii, S. Sako, and N. Tanigawa, Evaluation of treatment strategies for gastric cancer in the elderly according to the number of abnormal parameters on preoperative examination. Surg Today, 1999. 29(9): p. 837-41.
Lepage, C., M. Sant, A. Verdecchia, D. Forman, J. Esteve, and J. Faivre, Operative mortality after gastric cancer resection and long-term survival differences across Europe. Br J Surg, 2010. 97(2): p. 235-9.
Nelson, H., Predictors of surgical outcomes. J Gastroenterol Hepatol, 2002. 17 Suppl: p. S167-71.
de Wilde, R.F., M.G. Besselink, I. van der Tweel, I.H. de Hingh, C.H. van Eijck, C.H. Dejong, R.J. Porte, D.J. Gouma, O.R. Busch, and I.Q. Molenaar, Impact of nationwide centralization of pancreaticoduodenectomy on hospital mortality. Br J Surg, 2012. 99(3): p. 404-10.
Birkmeyer, N.J., P.P. Goodney, T.A. Stukel, B.E. Hillner, and J.D. Birkmeyer, Do cancer centers designated by the National Cancer Institute have better surgical outcomes? Cancer, 2005. 103(3): p. 435-41.
van de Poll-Franse, L.V., V.E. Lemmens, J.A. Roukema, J.W. Coebergh, and G.A. Nieuwenhuijzen, Impact of concentration of oesophageal and gastric cardia cancer surgery on long-term population-based survival. Br J Surg, 2011. 98(7): p. 956-63.
Meyer, H.J., The influence of case load and the extent of resection on the quality of treatment outcome in gastric cancer. Eur J Surg Oncol, 2005. 31(6): p. 595-604.
Lemmens, V.E., K. Bosscha, G. van der Schelling, S. Brenninkmeijer, J.W. Coebergh, and I.H. de Hingh, Improving outcome for patients with pancreatic cancer through centralization. Br J Surg, 2011. 98(10): p. 1455-62.
Wouters, M.W., H.E. Karim-Kos, S. le Cessie, B.P. Wijnhoven, L.P. Stassen, W.H. Steup, H.W. Tilanus, and R.A. Tollenaar, Centralization of esophageal cancer surgery: does it improve clinical outcome? Ann Surg Oncol, 2009. 16(7): p. 1789-98.
Dasgupta, M., D.B. Rolfson, P. Stolee, M.J. Borrie, and M. Speechley, Frailty is associated with postoperative complications in older adults with medical problems. Arch Gerontol Geriatr, 2009. 48(1): p. 78-83.
Robinson, T.N., B. Eiseman, J.I. Wallace, S.D. Church, K.K. McFann, S.M. Pfister, T.J. Sharp, and M. Moss, Redefining geriatric preoperative assessment using frailty, disability and co-morbidity. Ann Surg, 2009. 250(3): p. 449-55.
Pol, R.A., B.L. van Leeuwen, L. Visser, G.J. Izaks, J.J. van den Dungen, I.F. Tielliu, and C.J. Zeebregts, Standardised frailty indicator as predictor for postoperative delirium after vascular surgery: a prospective cohort study. Eur J Vasc Endovasc Surg, 2011. 42(6): p. 824-30.
Aaldriks, A.A., E.J. Giltay, S. le Cessie, L.G. van der Geest, J.E. Portielje, B.C. Tanis, J.W. Nortier, and E. Maartense, Prognostic value of geriatric assessment in older patients with advanced breast cancer receiving chemotherapy. Breast, 2013.
Aaldriks, A.A., L.G.M. van der Geest, E.J. Giltay, S. le Cessie, J.E.A. Portielje, B.C. Tanis, J.W.R. Nortier, and E. Maartense, Frailty and malnutrition predictive of mortality risk in older patients with advanced colorectal cancer receiving chemotherapy. Journal of Geriatric Oncology, 2013. 4(3): p. 218-226.
Kruizenga, H.M., J.C. Seidell, H.C. de Vet, N.J. Wierdsma, and M.A. van Bokhorst-de van der Schueren, Development and validation of a hospital screening tool for malnutrition: the short nutritional assessment questionnaire (SNAQ). Clin Nutr, 2005. 24(1): p. 75-82.
Lomivorotov, V.V., S.M. Efremov, V.A. Boboshko, D.A. Nikolaev, P.E. Vedernikov, M.N. Deryagin, V.N. Lomivorotov, and A.M. Karaskov, Prognostic value of nutritional screening tools for patients scheduled for cardiac surgery. Interact Cardiovasc Thorac Surg, 2013. 16(5): p. 612-8.
Neelemaat, F., H.M. Kruizenga, H.C. de Vet, J.C. Seidell, M. Butterman, and M.A. van Bokhorst-de van der Schueren, Screening malnutrition in hospital outpatients. Can the SNAQ malnutrition screening tool also be applied to this population? Clin Nutr, 2008. 27(3): p. 439-46.
Schuurmans, H., N. Steverink, S. Lindenberg, N. Frieswijk, and J.P. Slaets, Old or frail: what tells us more? J Gerontol A Biol Sci Med Sci, 2004. 59(9): p. M962-5.
Heemskerk, V.H., F. Lentze, K.W. Hulsewe, and T.G. Hoofwijk, Gastric carcinoma: review of the results of treatment in a community teaching hospital. World J Surg Oncol, 2007. 5: p. 81.
Wolters, U., T. Wolf, H. Stutzer, and T. Schroder, ASA classification and perioperative variables as predictors of postoperative outcome. Br J Anaesth, 1996. 77(2): p. 217-22.
Clavien, P.A., J. Barkun, M.L. de Oliveira, J.N. Vauthey, D. Dindo, R.D. Schulick, E. de Santibanes, J. Pekolj, K. Slankamenac, C. Bassi, R. Graf, R. Vonlanthen, R. Padbury, J.L. Cameron, and M. Makuuchi, The Clavien-Dindo classification of surgical complications: five-year experience. Ann Surg, 2009. 250(2): p. 187-96.
Dindo, D., N. Demartines, and P.A. Clavien, Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg, 2004. 240(2): p. 205-13.
Dikken, J.L., J.W. van Sandick, H.A. Maurits Swellengrebel, P.A. Lind, H. Putter, E.P. Jansen, H. Boot, N.C. van Grieken, C.J. van de Velde, M. Verheij, and A. Cats, Neo-adjuvant chemotherapy followed by surgery and chemotherapy or by surgery and chemoradiotherapy for patients with resectable gastric cancer (CRITICS). BMC Cancer, 2011. 11: p. 329.
Makary, M.A., D.L. Segev, P.J. Pronovost, D. Syin, K. Bandeen-Roche, P. Patel, R. Takenaga, L. Devgan, C.G. Holzmueller, J. Tian, and L.P. Fried, Frailty as a predictor of surgical outcomes in older patients. J Am Coll Surg, 2010. 210(6): p. 901-8.
Saxton, A. and V. Velanovich, Preoperative frailty and quality of life as predictors of postoperative complications. Ann Surg, 2011. 253(6): p. 1223-9.
Robinson, T.N., D.S. Wu, G.V. Stiegmann, and M. Moss, Frailty predicts increased hospital and six-month healthcare cost following colorectal surgery in older adults. Am J Surg, 2011. 202(5): p. 511-4.
Hoogendijk, E.O., H.E. van der Horst, D.J. Deeg, D.H. Frijters, B.A. Prins, A.P. Jansen, G. Nijpels, and H.P. van Hout, The identification of frail older adults in primary care: comparing the accuracy of five simple instruments. Age Ageing, 2013. 42(2): p. 262-5.
Grant Support
None
Author information
Authors and Affiliations
Corresponding author
Additional information
Discussant
Dr. Michael Zenilman (Bethesda, Maryland): This is one of an impressive new series focusing on the importance of frailty indicators for general, and specifically, gastrointestinal surgery
We are on the midst of a paradigm shift in risk assessment. The old school focused on complicated mathematical models based on clinical factors such as comorbidities, a history of cardiac disease, while the new school is based on the simple presence of frailty indicators and geriatric syndromes. It is now established that the presence of frailty is a much more powerful indicator than the others.
The challenge is how to measure it. While this study, using preop questionairres, captured some of frailty markers, they did not measure any objective variables such as serum albumin, dementia (measured by the mini-mental score), timed up and go, gait speed, and simply obtaining a history of falls. All of these are powerful univariate variables for postop risk.
My first question is did the authors confirm the presumed nutritional status of the patients w a serum albumin?
Second, both GFI and SNAQ measure self reported nutritional assessment, so doesn’t including both in a mutivariate analysis bias your results towards statistical significance? I am concerned by using it twice in a MV analysis and then using a combine score again in the same analysis.
Lastly, I applaud the use of six month outcomes in assessing surgical risk. Why did they not use it as a primary outcome? Thirty-day rates are much less critical in centers such as yours where the surgeons and institutions almost guaranty success.
Closing Discussant
Dr. Juul Tegels: Thank you Dr. Zenilman for taking the time to discuss our study and the excellent questions.
Due to the retrospective design of the study we could not verify results of the nutritional status questionnaire with other tests like serum albumin. Also serum albumin testing is not part of routine lab testing of patients who are evaluated for gastric cancer surgery.
With regard to your second question, I can say that we did notice the fact that both questionnaires incorporate items regarding unintentional weight loss. So in the manuscript and multivariate analyses, we also performed analyses when we removed the data with regard to unintentional weight loss in one of the two questionnaires. In these analyses, predictive value of the questionnaires remained. The reason for analyzing combined scores was to evaluate whether these two questionnaires perhaps amplified each others’ predictive value for perioperative mortality.
Your last remark is a good one and last week at the Dutch national surgical conference where these data were also presented a similar question was raised by one of the attending surgeons. In the present study, we also looked at 6-month mortality, but as a secondary outcome parameter. We took 30-day mortality or in-hospital mortality as the primary outcome parameter. Most important reason for choosing 30-day or in-hospital mortality was the idea to research wheter these scores were predictive of outcomes in short postoperative period. But we were obviously interested in their predictive value of GFI and SNAQ for other outcomes; therefore, we took these (serious adverse events, length-of-stay, and 6-month mortality) as secondary outcome parameters.
Rights and permissions
About this article
Cite this article
Tegels, J.J.W., de Maat, M.F.G., Hulsewé, K.W.E. et al. Value of Geriatric Frailty and Nutritional Status Assessment in Predicting Postoperative Mortality in Gastric Cancer Surgery. J Gastrointest Surg 18, 439–446 (2014). https://doi.org/10.1007/s11605-013-2443-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-013-2443-7