Abstract
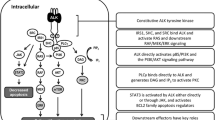
Anaplastic lymphoma kinase (ALK) is a tyrosine kinase receptor that affects a number of biological and biochemical functions through normal ligand-dependent signaling. It has oncogenic functions in a number of tumors including non-small cell lung cancer (NSCLC), anaplastic large cell lymphoma, and neuroblastoma when altered by translocation or amplification or mutation. On August 2011, a small molecule inhibitor against ALK, crizotinib, was approved for therapy against NSCLC with ALK translocations. As we determine the molecular heterogeneity of tumors, the potential of ALK as a relevant therapeutic target in a number of malignancies has become apparent. This review will discuss some of the tumor types with oncogenic ALK alterations. The activity and unique toxicities of crizotinib are described, along with potential mechanisms of resistance and new therapies beyond crizotinib.


Similar content being viewed by others
References
Morris SW, Kirstein MN, Valentine MB, Dittmer KG, Shapiro DN, Saltman DL et al (1994) Fusion of a kinase gene, ALK, to a nucleolar protein gene, NPM, in non-Hodgkin's lymphoma. Science 263(5151):1281–1284
Azarova AM, Gautam G, George RE (2011) Emerging importance of ALK in neuroblastoma. Semin Cancer Biol 21(4):267–275
Webb TR, Slavish J, George RE, Look AT, Xue L, Jiang Q et al (2009) Anaplastic lymphoma kinase: role in cancer pathogenesis and small-molecule inhibitor development for therapy. Expert Rev Anticancer Ther 9(3):331–356
Lee CC, Jia Y, Li N, Sun X, Ng K, Ambing E et al (2010) Crystal structure of the ALK (anaplastic lymphoma kinase) catalytic domain. Biochem J 430(3):425–437
Tartari CJ, Gunby RH, Coluccia AM, Sottocornola R, Cimbro B, Scapozza L et al (2008) Characterization of some molecular mechanisms governing autoactivation of the catalytic domain of the anaplastic lymphoma kinase. J Biol Chem 283(7):3743–3750
Iwahara T, Fujimoto J, Wen D, Cupples R, Bucay N, Arakawa T et al (1997) Molecular characterization of ALK, a receptor tyrosine kinase expressed specifically in the nervous system. Oncogene 14(4):439–449
The human protein atlas. Electronic citation 2012 Available from: www.proteinatlas.org
Stoica GE, Kuo A, Aigner A, Sunitha I, Souttou B, Malerczyk C et al (2001) Identification of anaplastic lymphoma kinase as a receptor for the growth factor pleiotrophin. J Biol Chem 276(20):16772–16779
Powers C, Aigner A, Stoica GE, McDonnell K, Wellstein A (2002) Pleiotrophin signaling through anaplastic lymphoma kinase is rate-limiting for glioblastoma growth. J Biol Chem 277(16):14153–14158
Miyake I, Hakomori Y, Shinohara A, Gamou T, Saito M, Iwamatsu A et al (2002) Activation of anaplastic lymphoma kinase is responsible for hyperphosphorylation of ShcC in neuroblastoma cell lines. Oncogene 21(38):5823–5834
Mourali J, Benard A, Lourenco FC, Monnet C, Greenland C, Moog-Lutz C et al (2006) Anaplastic lymphoma kinase is a dependence receptor whose proapoptotic functions are activated by caspase cleavage. Mol Cell Biol 26(16):6209–6222
Catalog of somatic mutations in cancer. Electronic citation 2012 Available from: http://www.sanger.ac.uk/perl/genetics/CGP/cosmic?action=gene&ln=ALK
Catlog of somatic mutations in cancer. Electronic citation 2012 Available from: http://www.sanger.ac.uk/perl/genetics/CGP/cosmic?action=bygene&ln=ALK&start=1&end=1621&coords=AA:AA
Siegel R, Ward E, Brawley O, Jemal A (2011) Cancer statistics, 2011: the impact of eliminating socioeconomic and racial disparities on premature cancer deaths. CA Cancer J Clin 61(4):212–236
Lung cancer—National Cancer Institute. Electronic citation 2012 Available from: http://www.cancer.gov/cancertopics/types/lung
Cancer of the lungs and bronchus—SEER Stat Fact Sheets. Electronic citation 2012 Available from: http://seer.cancer.gov/statfacts/html/lungb.html
Sandler A, Gray R, Perry MC, Brahmer J, Schiller JH, Dowlati A et al (2006) Paclitaxel–carboplatin alone or with bevacizumab for non-small-cell lung cancer. N Engl J Med 355(24):2542–2550
Scagliotti GV, Parikh P, von Pawel J, Biesma B, Vansteenkiste J, Manegold C et al. (2008) Phase III study comparing cisplatin plus gemcitabine with cisplatin plus pemetrexed in chemotherapy-naive patients with advanced-stage non-small-cell lung cancer. J Clin Oncol 26(21):3543–3551
Soda M, Choi YL, Enomoto M, Takada S, Yamashita Y, Ishikawa S et al (2007) Identification of the transforming EML4–ALK fusion gene in non-small-cell lung cancer. Nature 448(7153):561–566
Horn L, Pao W (2009) EML4–ALK: honing in on a new target in non-small-cell lung cancer. J Clin Oncol 27(26):4232–4235
Sasaki T, Rodig SJ, Chirieac LR, Janne PA (2010) The biology and treatment of EML4-ALK non-small cell lung cancer. Eur J Cancer 46(10):1773–1780
Soda M, Takada S, Takeuchi K, Choi YL, Enomoto M, Ueno T et al (2008) A mouse model for EML4–ALK-positive lung cancer. Proc Natl Acad Sci U S A 105(50):19893–19897
Shaw AT, Yeap BY, Mino-Kenudson M, Digumarthy SR, Costa DB, Heist RS et al (2009) Clinical features and outcome of patients with non-small-cell lung cancer who harbor EML4–ALK. J Clin Oncol 27(26):4247–4253
Shaw AT et al. Prognostic versus predictive value of EML4–ALK translocation in metastatic non-small cell lung cancer. J Clin Oncol 28. 4-3-2010. Ref Type: Abstract
Martinez P et al. ALK rearrangement in a selected population of advanced non-small cell lung cancer patients: FISH and inmunohistochemistry diagnostic methods, prevalence, and clinical outcomes. J Clin Oncol 29. 6-1-2011. Ref Type: Abstract
Rosell R, Moran T, Queralt C, Porta R, Cardenal F, Camps C et al (2009) Screening for epidermal growth factor receptor mutations in lung cancer. N Engl J Med 361(10):958–967
Kim ES, Salgia R (2009) MET pathway as a therapeutic target. J Thorac Oncol 4(4):444–447
Kwak EL, Bang YJ, Camidge DR, Shaw AT, Solomon B, Maki RG et al (2010) Anaplastic lymphoma kinase inhibition in non-small-cell lung cancer. N Engl J Med 363(18):1693–1703
Shaw AT, Yeap BY, Solomon BJ, Riely GJ, Gainor J, Engelman JA et al (2011) Effect of crizotinib on overall survival in patients with advanced non-small-cell lung cancer harbouring ALK gene rearrangement: a retrospective analysis. Lancet Oncol 12(11):1004–1012
Shaw AT et al. Impact of crizotinib on survival in patients with advanced, ALK-positive NSCLC compared with historical controls. J Clin Oncol 29. 3-12-2011. Ref Type: Abstract
Weickhardt AJ, Rothman MS, Salian-Mehta S, Kiseljak-Vassiliades K, Oton AB, Doebele RC et al. (2012) Rapid-onset hypogonadism secondary to crizotinib use in men with metastatic non-small cell lung cancer. Cancer. doi:10.1002/cncr.27450
Heuckmann JM, Holzel M, Sos ML, Heynck S, Balke-Want H, Koker M et al (2011) ALK mutations conferring differential resistance to structurally diverse ALK inhibitors. Clin Cancer Res 17(23):7394–7401
A clinical trial testing the efficacy of crizotinib versus standard chemotherapy pemetrexed plus cisplatin or carboplatin in patients with ALK positive non-squamous cancer of the lung. Electronic citation 2012 Available from: http://clinicaltrials.gov/ct2/show/NCT01154140?term=alk+pemetrexed&rank=2
Swerdlow S, Campo E, Harris NL et al. (2008) WHO classification of tumours of haematopoietic and lymphoid tissues, 4th edn. World Health Organization, Lyon
Vose J, Armitage J, Weisenburger D (2008) International peripheral T-cell and natural killer/T-cell lymphoma study: pathology findings and clinical outcomes. J Clin Oncol 26(25):4124–4130
Savage KJ, Harris NL, Vose JM, Ullrich F, Jaffe ES, Connors JM et al (2008) ALK− anaplastic large-cell lymphoma is clinically and immunophenotypically different from both ALK+ ALCL and peripheral T-cell lymphoma, not otherwise specified: report from the International Peripheral T-Cell Lymphoma Project. Blood 111(12):5496–5504
Lamant L, de Reynies A, Duplantier MM, Rickman DS, Sabourdy F, Giuriato S et al (2007) Gene-expression profiling of systemic anaplastic large-cell lymphoma reveals differences based on ALK status and two distinct morphologic ALK+ subtypes. Blood 109(5):2156–2164
Salaverria I, Bea S, Lopez-Guillermo A, Lespinet V, Pinyol M, Burkhardt B et al (2008) Genomic profiling reveals different genetic aberrations in systemic ALK-positive and ALK-negative anaplastic large cell lymphomas. Br J Haematol 140(5):516–526
Thompson MA, Stumph J, Henrickson SE, Rosenwald A, Wang Q, Olson S et al (2005) Differential gene expression in anaplastic lymphoma kinase-positive and anaplastic lymphoma kinase-negative anaplastic large cell lymphomas. Hum Pathol 36(5):494–504
Shiota M, Nakamura S, Ichinohasama R, Abe M, Akagi T, Takeshita M et al (1995) Anaplastic large cell lymphomas expressing the novel chimeric protein p80NPM/ALK: a distinct clinicopathologic entity. Blood 86(5):1954–1960
Shiota M, Fujimoto J, Semba T, Satoh H, Yamamoto T, Mori S (1994) Hyperphosphorylation of a novel 80 kDa protein-tyrosine kinase similar to Ltk in a human Ki-1 lymphoma cell line, AMS3. Oncogene 9(6):1567–1574
Amin HM, Lai R (2007) Pathobiology of ALK+ anaplastic large-cell lymphoma. Blood 110(7):2259–2267
Chiarle R, Voena C, Ambrogio C, Piva R, Inghirami G (2008) The anaplastic lymphoma kinase in the pathogenesis of cancer. Nat Rev Cancer 8(1):11–23
Kuefer MU, Look AT, Pulford K, Behm FG, Pattengale PK, Mason DY et al (1997) Retrovirus-mediated gene transfer of NPM–ALK causes lymphoid malignancy in mice. Blood 90(8):2901–2910
Wasik MA, Zhang Q, Marzec M, Kasprzycka M, Wang HY, Liu X (2009) Anaplastic lymphoma kinase (ALK)-induced malignancies: novel mechanisms of cell transformation and potential therapeutic approaches. Semin Oncol 36(2 Suppl 1):S27–S35
Chiarle R, Simmons WJ, Cai H, Dhall G, Zamo A, Raz R et al (2005) Stat3 is required for ALK-mediated lymphomagenesis and provides a possible therapeutic target. Nat Med 11(6):623–629
Zamo A, Chiarle R, Piva R, Howes J, Fan Y, Chilosi M et al (2002) Anaplastic lymphoma kinase (ALK) activates Stat3 and protects hematopoietic cells from cell death. Oncogene 21(7):1038–1047
Marzec M, Kasprzycka M, Ptasznik A, Wlodarski P, Zhang Q, Odum N et al (2005) Inhibition of ALK enzymatic activity in T-cell lymphoma cells induces apoptosis and suppresses proliferation and STAT3 phosphorylation independently of Jak3. Lab Invest 85(12):1544–1554
Zhang Q, Wang HY, Marzec M, Raghunath PN, Nagasawa T, Wasik MA (2005) STAT3- and DNA methyltransferase 1-mediated epigenetic silencing of SHP-1 tyrosine phosphatase tumor suppressor gene in malignant T lymphocytes. Proc Natl Acad Sci U S A 102(19):6948–6953
Zhang Q, Wang HY, Woetmann A, Raghunath PN, Odum N, Wasik MA (2006) STAT3 induces transcription of the DNA methyltransferase 1 gene (DNMT1) in malignant T lymphocytes. Blood 108(3):1058–1064
Slupianek A, Nieborowska-Skorska M, Hoser G, Morrione A, Majewski M, Xue L et al (2001) Role of phosphatidylinositol 3-kinase-Akt pathway in nucleophosmin/anaplastic lymphoma kinase-mediated lymphomagenesis. Cancer Res 61(5):2194–2199
Vega F, Medeiros LJ, Leventaki V, Atwell C, Cho-Vega JH, Tian L et al (2006) Activation of mammalian target of rapamycin signaling pathway contributes to tumor cell survival in anaplastic lymphoma kinase-positive anaplastic large cell lymphoma. Cancer Res 66(13):6589–6597
Marzec M, Kasprzycka M, Liu X, El-Salem M, Halasa K, Raghunath PN et al (2007) Oncogenic tyrosine kinase NPM/ALK induces activation of the rapamycin-sensitive mTOR signaling pathway. Oncogene 26(38):5606–5614
McDonnell SR, Hwang SR, Basrur V, Conlon KP, Fermin D, Wey E et al. (2011) NPM–ALK signals through glycogen synthase kinase 3beta to promote oncogenesis. Oncogene. doi:10.1038/onc.2011.542
Schmitz N, Trumper L, Ziepert M, Nickelsen M, Ho AD, Metzner B et al (2010) Treatment and prognosis of mature T-cell and NK-cell lymphoma: an analysis of patients with T-cell lymphoma treated in studies of the German High-Grade Non-Hodgkin Lymphoma Study Group. Blood 116(18):3418–3425
Skarbnik AP, Smith MR (2012) Brentuximab vedotin in anaplastic large cell lymphoma. Expert Opin Biol Ther 12(5):633–639
Younes A, Bartlett NL, Leonard JP, Kennedy DA, Lynch CM, Sievers EL et al (2010) Brentuximab vedotin (SGN-35) for relapsed CD30-positive lymphomas. N Engl J Med 363(19):1812–1821
Lowe EJ, Sposto R, Perkins SL, Gross TG, Finlay J, Zwick D et al (2009) Intensive chemotherapy for systemic anaplastic large cell lymphoma in children and adolescents: final results of Children's Cancer Group Study 5941. Pediatr Blood Cancer 52(3):335–339
Gambacorti-Passerini C, Messa C, Pogliani EM (2011) Crizotinib in anaplastic large-cell lymphoma. N Engl J Med 364(8):775–776
Delsol G, Lamant L, Mariame B, Pulford K, Dastugue N, Brousset P et al (1997) A new subtype of large B-cell lymphoma expressing the ALK kinase and lacking the 2; 5 translocation. Blood 89(5):1483–1490
Laurent C, Do C, Gascoyne RD, Lamant L, Ysebaert L, Laurent G et al (2009) Anaplastic lymphoma kinase-positive diffuse large B-cell lymphoma: a rare clinicopathologic entity with poor prognosis. J Clin Oncol 27(25):4211–4216
Gascoyne RD, Lamant L, Martin-Subero JI, Lestou VS, Harris NL, Muller-Hermelink HK et al (2003) ALK-positive diffuse large B-cell lymphoma is associated with Clathrin-ALK rearrangements: report of 6 cases. Blood 102(7):2568–2573
De Paepe P, Baens M, van Krieken H, Verhasselt B, Stul M, Simons A et al. (2003) ALK activation by the CLTC–ALK fusion is a recurrent event in large B-cell lymphoma. Blood 102(7):2638–2641
Cerchietti L, Damm-Welk C, Vater I, Klapper W, Harder L, Pott C et al. (2011) Inhibition of anaplastic lymphoma kinase (ALK) activity provides a therapeutic approach for CLTC–ALK-positive human diffuse large B cell lymphomas. PLoS One 6(4):e18436
Beltran B, Castillo J, Salas R, Quinones P, Morales D, Hurtado F et al (2009) ALK-positive diffuse large B-cell lymphoma: report of four cases and review of the literature. J Hematol Oncol 2:11
Maris JM, Hogarty MD, Bagatell R, Cohn SL (2007) Neuroblastoma. Lancet 369(9579):2106–2120
Brodeur GM, Seeger RC, Schwab M, Varmus HE, Bishop JM (1984) Amplification of N-myc in untreated human neuroblastomas correlates with advanced disease stage. Science 224(4653):1121–1124
Seeger RC, Brodeur GM, Sather H, Dalton A, Siegel SE, Wong KY et al (1985) Association of multiple copies of the N-myc oncogene with rapid progression of neuroblastomas. N Engl J Med 313(18):1111–1116
Brodeur GM, Green AA, Hayes FA, Williams KJ, Williams DL, Tsiatis AA (1981) Cytogenetic features of human neuroblastomas and cell lines. Cancer Res 41(11 Pt 1):4678–4686
George RE, Attiyeh EF, Li S, Moreau LA, Neuberg D, Li C et al (2007) Genome-wide analysis of neuroblastomas using high-density single nucleotide polymorphism arrays. PLoS One 2(2):e255
Mosse YP, Laudenslager M, Longo L, Cole KA, Wood A, Attiyeh EF et al (2008) Identification of ALK as a major familial neuroblastoma predisposition gene. Nature 455(7215):930–935
Ogawa S, Takita J, Sanada M, Hayashi Y (2011) Oncogenic mutations of ALK in neuroblastoma. Cancer Sci 102(2):302–308
Janoueix-Lerosey I, Lequin D, Brugieres L, Ribeiro A, de Pontual L, Combaret V et al. (2008) Somatic and germline activating mutations of the ALK kinase receptor in neuroblastoma. Nature 455(7215):967–970
Chen Y, Takita J, Choi YL, Kato M, Ohira M, Sanada M et al (2008) Oncogenic mutations of ALK kinase in neuroblastoma. Nature 455(7215):971–974
George RE, Sanda T, Hanna M, Frohling S, Luther W, Zhang J et al (2008) Activating mutations in ALK provide a therapeutic target in neuroblastoma. Nature 455(7215):975–978
Bresler SC, Wood AC, Haglund EA, Courtright J, Belcastro LT, Plegaria JS et al. (2011) Differential inhibitor sensitivity of anaplastic lymphoma kinase variants found in neuroblastoma. Sci Transl Med 3(108):108ra114
De BS, De PK, Kumps C, Zabrocki P, Porcu M, Westerhout EM et al (2010) Meta-analysis of neuroblastomas reveals a skewed ALK mutation spectrum in tumors with MYCN amplification. Clin Cancer Res 16(17):4353–4362
Schulte JH, Bachmann HS, Brockmeyer B, Depreter K, Oberthur A, Ackermann S et al (2011) High ALK receptor tyrosine kinase expression supersedes ALK mutation as a determining factor of an unfavorable phenotype in primary neuroblastoma. Clin Cancer Res 17(15):5082–5092
Passoni L, Longo L, Collini P, Coluccia AM, Bozzi F, Podda M et al (2009) Mutation-independent anaplastic lymphoma kinase overexpression in poor prognosis neuroblastoma patients. Cancer Res 69(18):7338–7346
Weiser D, Laudenslager M, Rappaport E, Carpenter E, Attiyeh EF, Diskin S et al. Stratification of patients with neuroblastoma for targeted ALK inhibitor therapy. J Clin Oncol 29. 6-6-2011. Ref Type: Abstract
Crizotinib in treating young patients with relapsed or refractory solid tumors or anaplastic large cell lymphoma. Electronic citation 2012 Available from: http://clinicaltrials.gov/ct2/show/NCT00939770?term=NCT00939770&rank=1
Doebele RC, Pilling AB, Aisner DL, Kutateladze TG, Le AT, Weickhardt AJ et al (2012) Mechanisms of resistance to crizotinib in patients with ALK gene rearranged non-small cell lung cancer. Clin Cancer Res 18(5):1472–1482
Camidge DR, Doebele RC (2012) Treating ALK-positive lung cancer—early successes and future challenges. Nat Rev Clin Oncol 9:268–277
Lovly CM, Pao W (2012) Esca** ALK inhibition: mechanisms of and strategies to overcome resistance. Sci Transl Med 4(120):120ps2
Katayama R, Shaw AT, Khan TM, Mino-Kenudson M, Solomon BJ, Halmos B et al. (2012) Mechanisms of acquired crizotinib resistance in ALK-rearranged lung cancers. Sci Transl Med 4(120):120ra17
Deng X, Wang J, Zhang J, Sim T, Kim ND, Sasaki T et al (2011) Discovery of 3,5-diamino-1,2,4-triazole ureas as potent anaplastic lymphoma kinase inhibitors. ACS Med Chem Lett 2(5):379–384
A first in patient, study of investigational drug PF-03446962 in patients with advanced solid tumors. Electronic citation 2012 Available from: http://clinicaltrials.gov/ct2/show/NCT00557856?term=PF-03446962&rank=2
A dose finding study with oral LDK378 in patients with tumors characterized by genetic abnormalities in anaplastic lymphoma kinase (ALK). Electronic citation 2012 Available from: http://clinicaltrials.gov/ct2/show/NCT01283516?term=ldk+378&rank=1
Study of an investigational drug, ASP3026, in patients with solid tumors. Electronic citation 2012 Available from: http://clinicaltrials.gov/ct2/show/NCT01401504?term=asp3026&rank=1
EML4–ALK mutation: consider Xalkori. Electronic citation 2012 Available from: http://www.collabrx.com/lung/lookup?subtype_3.1*no_others_3.1
West L, Vidwans SJ, Campbell NP, Shrager J, Simon GR, Bueno R et al (2012) A novel classification of lung cancer into molecular subtypes. PLoS One 7(2):e31906
A phase 1/2 study of the oral ALK/EGFR inhibitor AP26113. Electronic citation 2012 Available from: http://clinicaltrials.gov/ct2/show/NCT01449461?term=ap26113&rank=1
Carpenter EHE, Chow A, Christensen J, Maris J, Mosse Y. Mechanisms of resistance to small molecule inhibition of anaplastic lymphoma kinase in human neuroblastoma. Proceedings of the 102nd Annual Meeting of the American Association for Cancer Research Abstract 742. 4-1-2011. Ref Type: Abstract
Paolo D, Brignole C, Pastorino F, Carosio R, Zorzoli A, Rossi M et al (2011) Neuroblastoma-targeted nanoparticles entrap** siRNA specifically knockdown ALK. Mol Ther 19(6):1131–1140
Bonvini P, Gastaldi T, Falini B, Rosolen A (2002) Nucleophosmin–anaplastic lymphoma kinase (NPM–ALK), a novel Hsp90-client tyrosine kinase: down-regulation of NPM–ALK expression and tyrosine phosphorylation in ALK(+) CD30(+) lymphoma cells by the Hsp90 antagonist 17-allylamino,17-demethoxygeldanamycin. Cancer Res 62(5):1559–1566
Crizotinib and STA-9090 in ALK positive lung cancers. Electronic citation 2012 Available from: http://clinicaltrials.gov/ct2/show/NCT01579994?term=STA9090&rank=9
Sequist LV, Gettinger S, Senzer NN, Martins RG, Janne PA, Lilenbaum R et al (2010) Activity of IPI-504, a novel heat-shock protein 90 inhibitor, in patients with molecularly defined non-small-cell lung cancer. J Clin Oncol 28(33):4953–4960
Chen Z, Sasaki T, Tan X, Carretero J, Shimamura T, Li D et al (2010) Inhibition of ALK, PI3K/MEK, and HSP90 in murine lung adenocarcinoma induced by EML4–ALK fusion oncogene. Cancer Res 70(23):9827–9836
Inamura K, Takeuchi K, Togashi Y, Nomura K, Ninomiya H, Okui M et al (2008) EML4–ALK fusion is linked to histological characteristics in a subset of lung cancers. J Thorac Oncol 3(1):13–17
Inamura K, Takeuchi K, Togashi Y, Hatano S, Ninomiya H, Motoi N et al (2009) EML4–ALK lung cancers are characterized by rare other mutations, a TTF-1 cell lineage, an acinar histology, and young onset. Mod Pathol 22(4):508–515
Koivunen JP, Mermel C, Zejnullahu K, Murphy C, Lifshits E, Holmes AJ et al (2008) EML4–ALK fusion gene and efficacy of an ALK kinase inhibitor in lung cancer. Clin Cancer Res 14(13):4275–4283
Shinmura K, Kageyama S, Tao H, Bunai T, Suzuki M, Kamo T et al (2008) EML4–ALK fusion transcripts, but no NPM–, TPM3–, CLTC–, ATIC–, or TFG–ALK fusion transcripts, in non-small cell lung carcinomas. Lung Cancer 61(2):163–169
Martelli MP, Sozzi G, Hernandez L, Pettirossi V, Navarro A, Conte D et al (2009) EML4–ALK rearrangement in non-small cell lung cancer and non-tumor lung tissues. Am J Pathol 174(2):661–670
Wong DW, Leung EL, So KK, Tam IY, Sihoe AD, Cheng LC et al (2009) The EML4–ALK fusion gene is involved in various histologic types of lung cancers from nonsmokers with wild-type EGFR and KRAS. Cancer 115(8):1723–1733
Takeuchi K, Choi YL, Togashi Y, Soda M, Hatano S, Inamura K et al (2009) KIF5B–ALK, a novel fusion oncokinase identified by an immunohistochemistry-based diagnostic system for ALK-positive lung cancer. Clin Cancer Res 15(9):3143–3149
Choi YL, Takeuchi K, Soda M, Inamura K, Togashi Y, Hatano S et al (2008) Identification of novel isoforms of the EML4–ALK transforming gene in non-small cell lung cancer. Cancer Res 68(13):4971–4976
Takeuchi K, Choi YL, Soda M, Inamura K, Togashi Y, Hatano S et al (2008) Multiplex reverse transcription–PCR screening for EML4–ALK fusion transcripts. Clin Cancer Res 14(20):6618–6624
Fukuyoshi Y, Inoue H, Kita Y, Utsunomiya T, Ishida T, Mori M (2008) EML4–ALK fusion transcript is not found in gastrointestinal and breast cancers. Br J Cancer 98(9):1536–1539
Perner S, Wagner PL, Demichelis F, Mehra R, Lafargue CJ, Moss BJ et al (2008) EML4–ALK fusion lung cancer: a rare acquired event. Neoplasia 10(3):298–302
Takahashi T, Sonobe M, Kobayashi M, Yoshizawa A, Menju T, Nakayama E et al (2010) Clinicopathologic features of non-small-cell lung cancer with EML4–ALK fusion gene. Ann Surg Oncol 17(3):889–897
Wong DW, Leung EL, Wong SK, Tin VP, Sihoe AD, Cheng LC et al (2011) A novel KIF5B–ALK variant in nonsmall cell lung cancer. Cancer 117(12):2709–2718
Hernandez L, Bea S, Bellosillo B, Pinyol M, Falini B, Carbone A et al (2002) Diversity of genomic breakpoints in TFG–ALK translocations in anaplastic large cell lymphomas: identification of a new TFG–ALK(XL) chimeric gene with transforming activity. Am J Pathol 160(4):1487–1494
Drexler HG, Gignac SM, von Wasielewski R, Werner M, Dirks WG (2000) Pathobiology of NPM–ALK and variant fusion genes in anaplastic large cell lymphoma and other lymphomas. Leukemia 14(9):1533–1559
Caren H, Abel F, Kogner P, Martinsson T (2008) High incidence of DNA mutations and gene amplifications of the ALK gene in advanced sporadic neuroblastoma tumours. Biochem J 416(2):153–159
Murugan AK, **ng M (2011) Anaplastic thyroid cancers harbor novel oncogenic mutations of the ALK gene. Cancer Res 71(13):4403–4411
Acknowledgments
Supported in part by NIH/NCI, RO1 5R01CA100750-09, 5R01CA125541-05, Respiratory Health Association of Metropolitan Chicago, Geleerd Family Foundation (RS), the Elise Anderson Neuroblastoma Research Foundation (SLC), the Children’s Neuroblastoma Cancer Foundation (SLC), and Little Heroes Cancer Research Foundation (SLC).
Conflict of interest
None
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
La Madrid, A.M., Campbell, N., Smith, S. et al. Targeting ALK: a promising strategy for the treatment of non-small cell lung cancer, non-Hodgkin’s lymphoma, and neuroblastoma. Targ Oncol 7, 199–210 (2012). https://doi.org/10.1007/s11523-012-0227-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11523-012-0227-8