Abstract
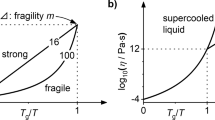
Differences in the thermodynamic functions between the liquid and crystalline states of La-based bulk metallic glasses alloys were calculated with the specific heat capacity C p and the fusion heat ΔH f, which we measured. Fragility indexes having different thermodynamic definitions were calculated from the temperature dependence of excess entropy ΔS liq-cry. It is ambiguous for La-based glass-forming liquid to evaluate fragility from the intercepts of ΔSliq-cry-temperature curves. We found that the thermodynamic fragility M ΔS , denoted ΔS, decreases at the onset melting temperatures T m rather than at glass transition temperatures T g, and relates linearly with the kinetic fragility m. The correlations between thermodynamic fragility and kinetic fragility are discussed within the frameworks of the Adam-Gibbs relationship and potential energy landscape theory.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Angell C A. Spectroscopy simulation and scattering, and the medium range order problem in glass. J Non-Cryst Solids, 1985, 73: 1–17
Bian X F, Sun B A, Hu L N, et al. Fragility of superheated melts and glass-forming ability in Al-based alloys. Phys Lett A, 2005, 335: 61–67
Song K K, Bian X F, Lv X Q, et al. The correlations among the fragility of supercooled liquids, the fragility of superheated melts, and the glass-forming ability for marginal metallic glasses. J Appl Phys, 2009, 105: 024304
Jia R, Bian X F, Lv X Q, et al. The relationship between viscosity and glass forming ability of Al-(Ni)-Yb alloy systems. Sci China Phys Mech Astron, 2010, 53: 390–393
Meng Q G, Zhou J K, Zheng H X, et al. Fragility of superheated melts and glass-forming ability in Pr-based alloys. Scripta Mater, 2006, 54: 777–781
Guo J, Bian X F, Li X L, et al. Correlation between superheated liquid fragility and potential energy landscape in Gd- and Pr-based glass-forming alloys. Sci China Ser G-Phys Mech Astron, 2008, 51: 387–393
Guo J, Bian X F. A correlation between superheated liquid fragility and supercooled liquid fragility in La- and Sm-based glass-forming alloys. J Alloy Compd, 2010, 504: 205–207
Kauzmann W. The nature of the glassy state and the behavior of liquids at low temperatures. Chem Rev, 1948, 43: 219–256
Adam G, Gibbs J H. On the temperature dependence of cooperative relaxation properties in glass-forming liquids. J Chem Phys, 1965, 43: 139–146
Huang D H, McKenna G B. New insights into the fragility dilemma in liquids. J Chem Phys, 2001, 114: 5621–5630
Fan G J, Choo H, Liaw P K. Fragility of metallic glass-forming liquids: A simple thermodynamic connection. J Non-Cryst Solids, 2005, 351: 3879–3883
Ito K, Moynihan C T, Angell C A. Thermodynamic determination of fragility in liquids and a fragile-to-strong liquid transition in water. Nature, 1999, 398: 492–495
Ngai K L, Yamamuro O. Thermodynamic fragility and kinetic fragility in supercooling liquids: A missing link in molecular liquids. J Chem Phys, 1999, 111: 10403–10406
Martinez L M, Angell C A. A thermodynamic connection to the fragility of glass-forming liquids. Nature, 2001, 410: 663–667
Ma H, Fecht H J. Thermodynamic and kinetic fragilities of Mg-based bulk metallic glass-forming liquids. J Mater Res, 2008, 23: 2816–2820
Fan G J, Fecht H J. A cluster model for the viscous flow of glass-forming liquids. J Chem Phys, 2002, 116: 5002–5006
Tanaka H. Relation between thermodynamics and kinetics of glass-forming liquids. Phys Rev Lett, 2003, 90: 055701
Lu Z P, Goh T T, Li Y. Glass formation in La-based La-Al-Ni-Cu-(Co) alloys by Bridgman solidification and their glass forming ability. Acta Mater, 1999, 47: 2215–2224
Lu Z P, Hu X, Li Y, et al. Glass forming ability of La-Al-Ni-Cu and Pd-Si-Cu bulk metallic glasses. Mater Sci Eng A, 2001, 304-306: 679–682
Lu Z P, Hu X, Li Y. Thermodynamics of La based La-Al-Cu-Ni-Co alloys studied by temperature modulated DSC. Intermetallics, 2000, 8: 477–480
Inoue A, Nakamure T, Sugita T, et al. Bulky La-Al-Tm (Tm=transition metal) amorphous alloys with high tensile strength produced by a high-pressure die casting method. Mater Trans JIM, 1993, 34: 351–358
Legg B A, Schroers J, Busch R. Thermodynamics, kinetics, and crystallization of Pt57.3Cu14.6Ni5.3P22.8 bulk metallic glass. Acta Mater, 2007, 55: 1109–1116
Kubaschewski O, Alcock C B, Spencer P J. Materials Thermochemistry. 6th ed. New York: Permagon, 1993
Glade S C, Busch R, Lee D S, et al. Thermodynamics of Cu47Ti34Zr11Ni8, Zr52.5Cu17.9Ni14.6Al10Ti5 and Zr57Cu15.4Ni12.6Al10Nb5 bulk metallic glass forming alloys. J Appl Phys, 2000, 87: 7242–7248
Busch R, Liu W, Johnson W L. Thermodynamics and kinetics of the Mg65Cu25Y10 bulk metallic glass forming liquid. J Appl Phys, 1998, 83: 4134–4141
Wilde G, Gorler G P, Willnecker R, et al. Thermodynamic properties of Pd40Ni40P20 in the glassy, liquid, and crystalline states. Appl Phys Lett, 1994, 65: 397–399
Busch R, Kim Y J, Johnson W L. Thermodynamics and kinetics of the undercooled liquid and the glass transition of the Zr41.2Ti13.8Cu2.5-Ni10.0Be22.5 alloy. J Appl Phys, 1995, 77: 4039–4043
Crowley K J, Zografi G. The use of thermal methods for predicting glass-former fragility. Thermochim Acta, 2001, 380: 79–93
Busch R, Bakke E, Johnson W L. Viscosity of the supercooled liquid and relaxation at the glass transition of the Zr46.75Ti8.25Cu7.5Ni10Be27.5 bulk metallic glass forming alloy. Acta Mater, 1998, 46: 4725–4732
Richert R, Angell C A. Dynamics of glassforming liquids. IV: On the link between molecular dynamics and configurational entropy. J Chem Phys, 1998, 108: 9016–9026
Angell C A. Thermodynamics: Liquid landscape. Nature, 1998, 393: 521–524
Sastry S. The relationship between fragility, configurational entropy and the potential energy landscape of glass-forming liquids. Nature, 2001, 409: 164–167
Hu L N, Bian X F, Wang W M, et al. Thermodynamics and dynamics of metallic glass formers: Their correlation for the investigation on potential energy landscape. J Phys Chem B, 2005, 109: 13737–13742
Takeuchi A, Inoue A. Calculations of mixing enthalpy and mismatch entropy for ternary amorphous alloys. Mater Trans JIM, 2005, 41: 1372–1378
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Jia, R., Bian, X. & Wang, Y. Thermodynamic determination of fragility in La-based glass-forming liquid. Chin. Sci. Bull. 56, 3912–3918 (2011). https://doi.org/10.1007/s11434-011-4844-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-011-4844-x