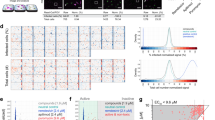
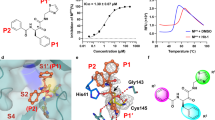
Abstract
The coronavirus disease 2019 (COVID-19) pandemic caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is a global crisis. Clinical candidates with high efficacy, ready availability, and that do not develop resistance are in urgent need. Despite that screening to repurpose clinically approved drugs has provided a variety of hits shown to be effective against SARS-CoV-2 infection in cell culture, there are few confirmed antiviral candidates in vivo. In this study, 94 compounds showing high antiviral activity against SARS-CoV-2 in Vero E6 cells were identified from 2,580 FDA-approved small-molecule drugs. Among them, 24 compounds with low cytotoxicity were selected, and of these, 17 compounds also effectively suppressed SARS-CoV-2 infection in HeLa cells transduced with human ACE2. Six compounds disturb multiple processes of the SARS-CoV-2 life cycle. Their prophylactic efficacies were determined in vivo using Syrian hamsters challenged with SARS-CoV-2 infection. Seven compounds reduced weight loss and promoted weight regain of hamsters infected not only with the original strain but also the D614G variant. Except for cisatracurium, six compounds reduced hamster pulmonary viral load, and IL-6 and TNF-α mRNA when assayed at 4 d postinfection. In particular, sertraline, salinomycin, and gilteritinib showed similar protective effects as remdesivir in vivo and did not induce antiviral drug resistance after 10 serial passages of SARS-CoV-2 in vitro, suggesting promising application for COVID-19 treatment.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Abu-Farha, M., Thanaraj, T.A., Qaddoumi, M.G., Hashem, A., Abubaker, J., and Al-Mulla, F. (2020). The role of lipid metabolism in COVID-19 virus infection and as a drug target. Int J Mol Sci 21, 3544.
Ahmad, J., Ikram, S., Ahmad, F., Rehman, I.U., and Mushtaq, M. (2020). SARS-CoV-2 RNA dependent RNA polymerase (RdRp)—a drug repurposing study. Heliyon 6, e04502.
Balasubramanian, A., Teramoto, T., Kulkarni, A.A., Bhattacharjee, A.K., and Padmanabhan, R. (2017). Antiviral activities of selected antimalarials against dengue virus type 2 and Zika virus. Antiviral Res 137, 141–150.
Berber, B., and Doluca, O. (2021). A comprehensive drug repurposing study for COVID19 treatment: novel putative dihydroorotate dehydrogenase inhibitors show association to serotonin-dopamine receptors. Brief Bioinform 22, 1023–1037.
Bouhaddou, M., Memon, D., Meyer, B., White, K.M., Rezelj, V.V., Correa Marrero, M., Polacco, B.J., Melnyk, J.E., Ulferts, S., Kaake, R.M., et al. (2020). The global phosphorylation landscape of SARS-CoV-2 infection. Cell 182, 685–712.e19.
Carpinteiro, A., Edwards, M.J., Hoffmann, M., Kochs, G., Gripp, B., Weigang, S., Adams, C., Carpinteiro, E., Gulbins, A., Keitsch, S., et al. (2020). Pharmacological inhibition of acid sphingomyelinase prevents uptake of SARS-CoV-2 by epithelial cells. Cell Rep Med 1, 100142.
Chen, Y., Lear, T.B., Evankovich, J.W., Larsen, M.B., Lin, B., Alfaras, I., Kennerdell, J.R., Salminen, L., Camarco, D.P., Lockwood, K.C., et al. (2021). A high-throughput screen for TMPRSS2 expression identifies FDA-approved compounds that can limit SARS-CoV-2 entry. Nat Commun 12, 3907.
Cheung, Y.Y., Chen, K.C., Chen, H., Seng, E.K., and Chu, J.J.H. (2014). Antiviral activity of lanatoside C against dengue virus infection. Antiviral Res 111, 93–99.
Dittmar, M., Lee, J.S., Whig, K., Segrist, E., Li, M., Kamalia, B., Castellana, L., Ayyanathan, K., Cardenas-Diaz, F.L., Morrisey, E.E., et al. (2021). Drug repurposing screens reveal cell-type-specific entry pathways and FDA-approved drugs active against SARS-CoV-2. Cell Rep 35, 108959.
Drayman, N., DeMarco, J.K., Jones, K.A., Azizi, S.A., Froggatt, H.M., Tan, K., Maltseva, N.I., Chen, S., Nicolaescu, V., Dvorkin, S., et al. (2021). Masitinib is a broad coronavirus 3CL inhibitor that blocks replication of SARS-CoV-2. Science 373, 931–936.
Eberle, R.J., Olivier, D.S., Amaral, M.S., Gering, I., Willbold, D., Arni, R. K., and Coronado, M.A. (2021). The repurposed drugs suramin and quinacrine cooperatively inhibit SARS-CoV-2 3CLproin vitro. Viruses 13, 873.
Farsalinos, K., Eliopoulos, E., Leonidas, D.D., Papadopoulos, G.E., Tzartos, S., and Poulas, K. (2020). Nicotinic cholinergic system and COVID-19: in silico identification of an interaction between SARS-CoV-2 and nicotinic receptors with potential therapeutic targeting implications. Int J Mol Sci 21, 5807.
Finch, C.L., Dyall, J., Xu, S., Nelson, E.A., Postnikova, E., Liang, J.Y., Zhou, H., DeWald, L.E., Thomas, C.J., Wang, A., et al. (2021). Formulation, stability, pharmacokinetic, and modeling studies for tests of synergistic combinations of orally available approved drugs against ebola virus in vivo. Microorganisms 9, 566.
Francis, M.E., Goncin, U., Kroeker, A., Swan, C., Ralph, R., Lu, Y., Etzioni, A.L., Falzarano, D., Gerdts, V., Machtaler, S., et al. (2021). SARS-CoV-2 infection in the syrian hamster model causes inflammation as well as type I interferon dysregulation in both respiratory and non-respiratory tissues including the heart and kidney. PLoS Pathog 17, e1009705.
Gao, K., Nguyen, D.D., Chen, J., Wang, R., and Wei, G.W. (2020a). Repositioning of 8565 existing drugs for COVID-19. J Phys Chem Lett 11, 5373–5382.
Gao, M., Yang, L., Chen, X., Deng, Y., Yang, S., Xu, H., Chen, Z., and Gao, X. (2020b). A study on infectivity of asymptomatic SARS-CoV-2 carriers. Respir Med 169, 106026.
Gehring, G., Rohrmann, K., Atenchong, N., Mittler, E., Becker, S., Dahlmann, F., PÖhlmann, S., Vondran, F.W.R., David, S., Manns, M.P., et al. (2014). The clinically approved drugs amiodarone, dronedarone and verapamil inhibit filovirus cell entry. J Antimicrob Chemother 69, 2123–2131.
Geleris, J., Sun, Y., Platt, J., Zucker, J., Baldwin, M., Hripcsak, G., Labella, A., Manson, D.K., Kubin, C., Barr, R.G., et al. (2020). Observational study of hydroxychloroquine in hospitalized patients with COVID-19. N Engl J Med 382, 2411–2418.
Ginex, T., Garaigorta, U., Ramirez, D., Castro, V., Nozal, V., Maestro, I., García-Cárceles, J., Campillo, N.E., Martinez, A., Gastaminza, P., et al. (2021). Host-directed FDA-approved drugs with antiviral activity against SARS-CoV-2 identified by hierarchical in silico/in vitro screening methods. Pharmaceuticals 14, 332.
Global report investigating novel coronavirus haplotypes. https://cov-lineages.org/global_report.html.
Guan, M., Wang, W., Liu, X., Tong, Y., Liu, Y., Ren, H., Zhu, S., Dubuisson, J., Baumert, T.F., Zhu, Y., et al. (2012). Three different functional microdomains in the hepatitis C virus hypervariable region 1 (HVR1) mediate entry and immune evasion. J Biol Chem 287, 35631–35645.
Han, Y., Duan, X., Yang, L., Nilsson-Payant, B.E., Wang, P., Duan, F., Tang, X., Yaron, T.M., Zhang, T., Uhl, S., et al. (2021). Identification of SARS-CoV-2 inhibitors using lung and colonic organoids. Nature 589, 270–275.
Herring, S., Oda, J.M., Wagoner, J., Kirchmeier, D., O’Connor, A., Nelson, E.A., Huang, Q., Liang, Y., DeWald, L.E., Johansen, L.M., et al. (2021). Inhibition of arenaviruses by combinations of orally available approved drugs. Antimicrob Agents Chemother 65.
Ianevski, A., Yao, R., Fenstad, M.H., Biza, S., Zusinaite, E., Reisberg, T., Lysvand, H., Løseth, K., Landsem, V.M., Malmring, J.F., et al. (2020). Potential antiviral options against SARS-CoV-2 infection. Viruses 12, 642.
Ju, X., Zhu, Y., Wang, Y., Li, J., Zhang, J., Gong, M., Ren, W., Li, S., Zhong, J., Zhang, L., et al. (2021). A novel cell culture system modeling the SARS-CoV-2 life cycle. PLoS Pathog 17, e1009439.
Li, Q., Cui, C., Xu, F., Zhao, J., Li, N., Li, H., Wang, T., Zhang, H., Liu, N., Wei, Y., et al. (2021). Evaluation of the efficacy and safety of hydroxychloroquine in comparison with chloroquine in moderate and severe patients with COVID-19. Sci China Life Sci 64, 660–663.
Liu, Z., Chu, R., Gong, L., Su, B., and Wu, J. (2020). The assessment of transmission efficiency and latent infection period in asymptomatic carriers of SARS-CoV-2 infection. Int J Infect Dis 99, 325–327.
Mahase, E. (2021). COVID-19: molnupiravir reduces risk of hospital admission or death by 50% in patients at risk, MSD reports. BMJ 375, n2422.
Mirabelli, C., Wotring, J.W., Zhang, C.J., McCarty, S.M., Fursmidt, R., Pretto, C.D., Qiao, Y., Zhang, Y., Frum, T., Kadambi, N.S., et al. (2021). Morphological cell profiling of SARS-CoV-2 infection identifies drug repurposing candidates for COVID-19. Proc Natl Acad Sci USA 118, e2105815118.
Mori, M., Kaneko, N., Ueno, Y., Yamada, M., Tanaka, R., Saito, R., Shimada, I., Mori, K., and Kuromitsu, S. (2017). Gilteritinib, a FLT3/AXL inhibitor, shows antileukemic activity in mouse models of FLT3 mutated acute myeloid leukemia. Invest New Drugs 35, 556–565.
Naujokat, C., and Steinhart, R. (2012). Salinomycin as a drug for targeting human cancer stem cells. J Biomed Biotechnol 2012, 1–17.
Nemerow, G.R., and Cooper, N.R. (1984). Infection of B lymphocytes by a human herpesvirus, Epstein-Barr virus, is blocked by calmodulin antagonists. Proc Natl Acad Sci U S A 81, 4955–4959.
Ochiai, H., Kurokawa, M., and Niwayama, S. (1991). Influence of trifluoperazine on the late stage of influenza virus infection in MDCK cells. Antiviral Res 15, 149–160.
Otręba, M., Kośmider, L., and Rzepecka-Stojko, A. (2020). Antiviral activity of chlorpromazine, fluphenazine, perphenazine, prochlorperazine, and thioridazine towards RNA-viruses. A review. Eur J Pharmacol 887, 173553.
Pandey, A., Nikam, A.N., Shreya, A.B., Mutalik, S.P., Gopalan, D., Kulkarni, S., Padya, B.S., Fernandes, G., Mutalik, S., and Prassl, R. (2020). Potential therapeutic targets for combating SARS-CoV-2: drug repurposing, clinical trials and recent advancements. Life Sci 256, 117883.
Patten, J.J., Keiser, P.T., Gysi, D., Menichetti, G., Mori, H., Donahue, C.J., Gan, X., Do Valle, I., Geoghegan-Barek, K., Anantpadma, M. et al. (2021). Multidose evaluation of 6,710 drug repurposing library identifies potent SARS-CoV-2 infection inhibitors in vitro and in vivo. bioRxiv doi: https://doi.org/10.1101/2021.04.20.440626.
Peng, H.R., Li, C.Z., Tang, H.L., Xue, J.Y., Chen, Z.H., Liang, X.S., Zhu, Y.M., **a, B.H., Zhu, Y.J., Wu, J.J. et al. (2020). Isolation and identification of two strains of severe acute respiratory syndrome coronavirus 2 from coronavirus disease 2019 patients in Shanghai (in Chinese). Acad J Sec Mil Med Univ 41, 365–370.
Plante, J.A., Liu, Y., Liu, J., **a, H., Johnson, B.A., Lokugamage, K.G., Zhang, X., Muruato, A.E., Zou, J., Fontes-Garfias, C.R., et al. (2021). Spike mutation d614g alters SARS-CoV-2 fitness. Nature 592, 116–121.
Puhl, A.C., Fritch, E.J., Lane, T.R., Tse, L.V., Yount, B.L., Sacramento, C. Q., Fintelman-Rodrigues, N., Tavella, T.A., Maranhão Costa, F.T., Weston, S., et al. (2021). Repurposing the Ebola and Marburg virus inhibitors tilorone, quinacrine, and pyronaridine: In vitro activity against SARS-CoV-2 and potential mechanisms. ACS Omega 6, 7454–7468.
Rakedzon, S., Neuberger, A., Domb, A.J., Petersiel, N., and Schwartz, E. (2021). From hydroxychloroquine to ivermectin: what are the anti-viral properties of anti-parasitic drugs to combat SARS-CoV-2? J Travel Med 28, taab005.
Riva, L., Yuan, S., Yin, X., Martin-Sancho, L., Matsunaga, N., Pache, L., Burgstaller-Muehlbacher, S., De Jesus, P.D., Teriete, P., Hull, M.V., et al. (2020). Discovery of SARS-CoV-2 antiviral drugs through large-scale compound repurposing. Nature 586, 113–119.
Sharma, G., Champalal Sharma, D., Hwei Fen, L., Pathak, M., Bethur, N., Pendharkar, V., Peiris, M., and Altmeyer, R. (2013). Reduction of influenza virus-induced lung inflammation and mortality in animals treated with a phosophodisestrase-4 inhibitor and a selective serotonin reuptake inhibitor. Emerg Microbes Infect 2, 1–9.
Ueno, Y., Mori, M., Kamiyama, Y., Kaneko, N., Isshiki, E., and Takeuchi, M. (2016). Gilteritinib (ASP2215), a novel FLT3/AXL inhibitor: Preclinical evaluation in combination with azacitidine in acute myeloid leukemia. Blood 128, 2830.
Vagapova, E.R., Lebedev, T.D., and Prassolov, V.S. (2021). Viral fibrotic scoring and drug screen based on MAPK activity uncovers EGFR as a key regulator of COVID-19 fibrosis. Sci Rep 11, 11234.
Volz, E., Hill, V., McCrone, J.T., Price, A., Jorgensen, D., O’Toole, Á., Southgate, J., Johnson, R., Jackson, B., Nascimento, F.F., et al. (2021). Evaluating the effects of SARS-CoV-2 spike mutation D614G on transmissibility and pathogenicity. Cell 184, 64–75.e11.
Wang, M., Cao, R., Zhang, L., Yang, X., Liu, J., Xu, M., Shi, Z., Hu, Z., Zhong, W., and **ao, G. (2020). Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Res 30, 269–271.
Wang, S., Qiu, Z., Hou, Y., Deng, X., Xu, W., Zheng, T., Wu, P., **e, S., Bian, W., Zhang, C., et al. (2021). AXL is a candidate receptor for SARS-CoV-2 that promotes infection of pulmonary and bronchial epithelial cells. Cell Res 31, 126–140.
Wilson, A.J., Troy-Barnes, E., Subhan, M., Clark, F., Gupta, R., Fielding, A.K., Kottaridis, P., Mansour, M.R., O’Nions, J., Payne, E., et al. (2020). Successful remission induction therapy with gilteritinib in a patient with de novo FLT3-mutated acute myeloid leukaemia and severe COVID-19. Br J Haematol 190, e189–e190.
**ao, X., Wang, C., Chang, D., Wang, Y., Dong, X., Jiao, T., Zhao, Z., Ren, L., Dela Cruz, C.S., Sharma, L., et al. (2020). Identification of potent and safe antiviral therapeutic candidates against SARS-CoV-2. Front Immunol 11, 586572.
**ng, J., Paithankar, S., Liu, K., Uhl, K., Li, X., Ko, M., Kim, S., Haskins, J., and Chen, B. (2021). Published anti-SARS-CoV-2 in vitro hits share common mechanisms of action that synergize with antivirals. Brief Bioinform 22.
Yang, C.W., Peng, T.T., Hsu, H.Y., Lee, Y.Z., Wu, S.H., Lin, W.H., Ke, Y. Y., Hsu, T.A., Yeh, T.K., Huang, W.Z., et al. (2020). Repurposing old drugs as antiviral agents for coronaviruses. Biomed J 43, 368–374.
Yu, B., Li, C., Chen, P., Li, J., Jiang, H., and Wang, D.W. (2021). Beneficial effects exerted by hydroxychloroquine in treating COVID-19 patients via protecting multiple organs. Sci China Life Sci 64, 330–333.
Zhou, S., Hill, C.S., Sarkar, S., Tse, L.V., Woodburn, B.M.D., Schinazi, R. F., Sheahan, T.P., Baric, R.S., Heise, M.T., and Swanstrom, R. (2021). p-d-N4-hydroxycytidine inhibits SARS-CoV-2 through lethal mutagenesis but is also mutagenic to mammalian cells. J Infect Dis 224, 415–419.
Zhou, Y., Zhou, B., Pache, L., Chang, M., Khodabakhshi, A.H., Tanaseichuk, O., Benner, C., and Chanda, S.K. (2019). Metascape provides a biologist-oriented resource for the analysis of systems-level datasets. Nat Commun 10, 1523.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (31570170) and the National Key Research and Development Program of China (2016YFC1200401).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Compliance and ethics
The author(s) declare that they have no conflict of interest.
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
Peng, H., Ding, C., Jiang, L. et al. Discovery of potential anti-SARS-CoV-2 drugs based on large-scale screening in vitro and effect evaluation in vivo. Sci. China Life Sci. 65, 1181–1197 (2022). https://doi.org/10.1007/s11427-021-2031-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-021-2031-7