Abstract
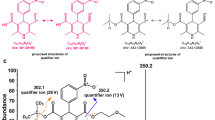
AB-FUBINACA, N-(1-amino-3-methyl-1-oxobutan-2-yl)-1-(4-fluorobenzyl)-1H-indazole-3-carboxamide, is an indazole synthetic cannabinoid identified in drug seizures around the world. Few metabolism data are available, despite the need for human urinary markers to detect AB-FUBINACA intake. Our main objective was to identify suitable analytical targets by analyzing human hepatocyte incubation samples with high-resolution mass spectrometry (HRMS) and to confirm the results in authentic urine specimens. We also determined AB-FUBINACA’s metabolic stability in human liver microsomes (HLMs) and compared hepatocyte and urine results with in silico predictions. The metabolic stability of AB-FUBINACA was determined in pooled HLMs (1 µmol/l, up to 1 h). The metabolite profile of human hepatocytes (10 µmol/l, 1 and 3 h) and urine samples from two subjects were determined by HRMS using information-dependent tandem-mass spectrometry (MS-MS) acquisition. Data were analyzed with MetabolitePilot™ software utilizing different processing algorithms, including generic peak finding, mass defect filtering, neutral loss, and product ion filtering. In silico metabolite prediction was performed with MetaSite™ software. AB-FUBINACA’s half-life in HLMs was 62.6 ± 4.0 min. AB-FUBINACA produced 11 metabolites (2 glucuronides) in human hepatocytes and 10 were identified in authentic human urine. Major metabolic pathways were terminal amide hydrolysis, acyl glucuronidation and hydroxylation at the aminooxobutane moiety. Epoxidation followed by hydrolysis, hydroxylation at the indazole moiety and dehydrogenation were minor pathways. Defluorination did not occur. Seventeen first-generation metabolites were predicted in silico, of which seven were observed in vitro and eight in vivo. We recommend AB-FUBINACA carboxylic acid, hydroxy AB-FUBINACA carboxylic acid, dihydrodiol AB-FUBINACA and dihydrodiol AB-FUBINACA carboxylic acid as suitable urinary markers.





Similar content being viewed by others
References
Castaneto MS, Gorelick DA, Desrosiers NA, Hartman RL, Pirard S, Huestis MA (2014) Synthetic cannabinoids: epidemiology, pharmacodynamics, and clinical implications. Drug Alcohol Depend 144:12–41
United Nations on Drugs and Crime. World Drug Report 2014 (2014) United Nations: Vienna.http://www.unodc.org/documents/wdr2014/World_Drug_Report_2014_web.pdf. Accessed Dec 2014
US Drug Enforcement Administration (2013) Establishment of drug codes for 26 substances. Final rule. Fed Regist 78:664–666
Buchler JP, Hayes MJ, Hegde SG, Hockerman SL, Jones DE, Kortum SW, Rico JG, Tenbrink RE, Wu KK (2009) Indazole derivatives. US Patent CA 2714573 A1:1–283
Brents LK, Reichard EE, Zimmerman SM, Moran JH, Fantegrossi WE, Prather PL (2011) Phase I hydroxylated metabolites of the K2 synthetic cannabinoid JWH -018 retain in vitro and in vivo cannabinoid 1 receptor affinity and activity. PLoS One 6:e21917
Uchiyama N, Matsuda S, Wakana D, Kikura-Hanajiri R, Goda Y (2013) New cannabimimetic indazole derivatives, N-(1-amino-3-methyl-1-oxobutan-2-yl)-1-pentyl-1H-indazole-3-carboxamide (AB-PINACA) and N-(1-amino-3-methyl-1-oxobutan-2-yl)-1-(4-fluorobenzyl)-1H-indazole-3-caboxamide (AB-FUBINACA) identified as designer drugs in illegal products. Forensic Toxicol 31:93–100
Drug Enforcement Administration (2014) 2013 Midyear Report. Department of Justice, Springfield, VA. http: http://www.deadiversion.usdoj.gov/nflis/2013midyear.pdf. Accessed Aug 2014
Drug Enforcement Administration (2014) Schedules of controlled substances: temporary placement of four synthetic cannabinoids into schedule I: notice of intent. Fed Regist 79:7577–7582
Centers for Disease Control and Prevention (2013) Notes from the field: severe illness associated with synthetic cannabinoid use—Brunswick, Georgia, 2013. MMWR Morb Mortal Wkly Rep 62:939
Federal Drug Control Service of Russia (2014) Bыcтyплeниe пpeдceдaтeля ГAК, диpeктopa ФCКH Poccии B.П. Ивaнoвa нa зaceдaнии ГAК 6 oктябpя 2014 г. http://fskn.gov.ru/includes/periodics/speeches_fskn/2014/1006/124332682/detail.shtml. Accessed Dec 2014
Gurney SMR, Scott KS, Kacinko SL, Presley BC, Logan BK (2014) Pharmacology, toxicology, and adverse effects of synthetic cannabinoid drugs. Forensic Sci Rev 26:53–78
Research Chemical Review (2014) Trip report tme: 250 mg AB-FUBINACA and 500 mg AH-7921. http://researchchemicalreview.wordpress.com/2014/02/25/trip-report-tme-250mg-ab-fubinaca-and-500mg-ah-7921/ Accessed Aug 2014
De Brabanter N, Esposito S, Tudela E, Lootens L, Meuleman P, Leroux-Roels G, Deventer K, Van Eenoo P (2013) In vivo and in vitro metabolism of the synthetic cannabinoid JWH-200. Rapid Commun Mass Spectrom 27:2115–2126
Grigoryev A, Melnik A, Savchuk S, Simonov A, Rozhanets V (2011) Gas and liquid chromatography-mass spectrometry studies on the metabolism of the synthetic phenylacetylindole cannabimimetic JWH-250, the psychoactive component of smoking mixtures. J Chromatogr B 879:2519–2526
Wintermeyer A, Moller I, Thevis M, Jubner M, Beike J, Rothschild MA, Bender K (2010) In vitro phase I metabolism of the synthetic cannabimimetic JWH-018. Anal Bioanal Chem 398:2141–2153
Takayama T, Suzuki M, Todoroki K, Inoue K, Min JZ, Kikura-Hanajiri R, Goda Y, Toyo’oka T (2014) UPLC/ESI-MS/MS-based determination of metabolism of several new illicit drugs, ADB-FUBINACA, AB-FUBINACA, AB-PINACA, QUPIC, 5F-QUPIC and alpha-PVT, by human liver microsomes. Biomed Chromatogr 28:831–838
Jang M, Shin I, Yang W, Chang H, Yoo HH, Lee J, Kim E (2014) Determination of major metabolites of MAM-2201 and JWH-122 in in vitro and in vivo studies to distinguish their intake. Forensic Sci Int 244:85–91
Holm NB, Pedersen AJ, Dalsgaard PW, Linnet K (2015) Metabolites of 5F-AKB-48, a synthetic cannabinoid receptor agonist, identified in human urine and liver microsomal preparations using liquid chromatography high-resolution mass spectrometry. Drug Test Anal 7:199–206
** MJ, Lee J, In MK, Yoo HH (2013) Characterization of in vitro metabolites of CP 47,497, a synthetic cannabinoid, in human liver microsomes by LC–MS/MS. J Forensic Sci 58:195–199
Gandhi AS, Wohlfarth AW, Zhu M, Pang S, Castaneto M, Scheidweiler KB, Huestis MA (2015) High-resolution mass spectrometric metabolite profiling of a novel synthetic designer drug, N-(adamantan-1-yl)-1-(5-fluoropentyl)-1H-indole-3-carboxamide (STS-135), using cryopreserved human hepatocytes and assessment of metabolic stability with human liver microsomes. Drug Test Anal 7:187–198
Gandhi AS, Zhu M, Pang S, Wohlfarth A, Scheidweiler KB, Liu HF, Huestis MA (2013) First characterization of AKB-48 metabolism, a novel synthetic cannabinoid, using human hepatocytes and high-resolution mass spectrometry. AAPS J 15:1091–1098
Wohlfarth A, Gandhi AS, Pang S, Zhu M, Scheidweiler KB, Huestis MA (2014) Metabolism of synthetic cannabinoids PB-22 and its 5-fluoro analog, 5F-PB-22, by human hepatocyte incubation and high-resolution mass spectrometry. Anal Bioanal Chem 406:1763–1780
Wohlfarth A, Pang S, Zhu M, Gandhi AS, Scheidweiler KB, Huestis MA (2014) Metabolism of RCS-8, a synthetic cannabinoid with cyclohexyl structure, in human hepatocytes by high-resolution MS. Bioanalysis 6:1187–2000
Wohlfarth A, Pang S, Zhu M, Gandhi AS, Scheidweiler KB, Liu HF, Huestis MA (2013) First metabolic profile of XLR-11, a novel synthetic cannabinoid, obtained by using human hepatocytes and high-resolution mass spectrometry. Clin Chem 59:1638–1648
Strano-Rossi S, Anzilotti L, Dragoni S, Pellegrino RM, Goracci L, Pascali VL, Cruciani G (2014) Metabolism of JWH-015, JWH-098, JWH-251, and JWH-307 in silico and in vitro: a pilot study for the detection of unknown synthetic cannabinoids metabolites. Anal Bioanal Chem 406:3621–3636
Thomsen R, Nielsen LM, Holm NB, Rasmussen HB, Linnet K (2014) Synthetic cannabimimetic agents metabolized by carboxylesterases. Drug Test Anal. doi:10.1002/dta.1731
Baranczewski P, Stanczak A, Sundberg K, Svensson R, Wallin A, Jansson J, Garberg P, Postlind H (2006) Introduction to in vitro estimation of metabolic stability and drug interactions of new chemical entities in drug discovery and development. Pharmacol Rep 58:453–472
McNaney CA, Drexler DM, Hnatyshyn SY, Zvyaga TA, Knipe JO, Belcastro JV, Sanders M (2008) An automated liquid chromatography-mass spectrometry process to determine metabolic stability half-life and intrinsic clearance of drug candidates by substrate depletion. ASSAY Drug Dev Technol 6:121–129
Lave T, Dupin S, Schmitt C, Valles B, Ubeaud G, Chou RC, Jaeck D, Coassolo P (1997) The use of human hepatocytes to select compounds based on their expected hepatic extraction ratios in humans. Pharm Res 14:152–155
Sobolevsky T, Prasolov I, Rodchenkov G (2012) Detection of urinary metabolites of AM-2201 and UR-144, two novel synthetic cannabinoids. Drug Test Anal 4:745–753
Regan SL, Maggs JL, Hammond TG, Lambert C, Williams DP, Park DK (2010) Acyl glucuronides: the good, the bad and the ugly. Biopharm Drug Dispos 31:367–395
Castaneto MS, Scheidweiler KB, Gandhi A, Wohlfarth A, Klette KL, Martin TM, Huestis MA (2014) Quantitative urine confirmatory testing for synthetic cannabinoids in randomly collected urine specimens. Drug Test Anal. doi:10.1002/dta.1709
Hutter M, Broecker S, Kneisel S, Auwarter V (2012) Identification of the major urinary metabolites in man of seven synthetic cannabinoids of the aminoalkylindole type present as adulterants in ‘herbal mixtures’ using LC-MS/MS techniques. J Mass Spectrom 47:54–65
Acknowledgments
This research was supported by the Intramural Research Program of the National Institute on Drug Abuse, National Institutes of Health. AB-FUBINACA was generously donated by the US Drug Enforcement Administration. Molecular Discovery Ltd kindly provided the MetaSite™ software.
Conflict of interest
There are no financial or other relations that could lead to a conflict of interest.
Ethical approval
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Castaneto, M.S., Wohlfarth, A., Pang, S. et al. Identification of AB-FUBINACA metabolites in human hepatocytes and urine using high-resolution mass spectrometry. Forensic Toxicol 33, 295–310 (2015). https://doi.org/10.1007/s11419-015-0275-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11419-015-0275-8