Abstract
Purpose
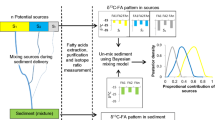
Compound-specific stable isotopes (CSSIs) of very long-chain fatty acids (VLCFAs) of plant origin were investigated in a soil and sediment tracing context in a watershed in Manitoba, Canada. Spatial and temporal variability in δ13CFA values and concentrations was examined at the point, transect, and field scales to determine the (1) ability to differentiate sediment sources in C3-cropped fields, (2) impact of subsampling on source tracer fingerprints, and (3) major sediment source for a downstream mixture using the Bayesian unmixing model MixSIAR.
Materials and methods
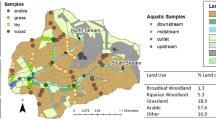
Analysis was performed for five agricultural fields over six sampling periods. Soil and sediment samples (320) were processed for VLCFA analyses (C20:0–C30:0, C32:0). Quantification was performed by gas chromatography–flame ionization detection (GC-FID) and 13C determination by GC combustion–isotope ratio mass spectrometry (GCC-IRMS). Data were analyzed using weighted t tests to differentiate fields by δ13CFA values. The major sediment source was determined using the following steps: (1) a point-in-polygon approach to identify VLCFA tracers; (2) unmixing using MixSIAR; (3) source apportioning using VLCFA concentrations and %C.
Results and discussion
VLCFA δ13CFA values vary spatially within a cropped field due to environmental factors. Sediment source fingerprints are dependent on the variability in δ13CFA values and the quantitative combining of subsamples. Cropped fields that appeared homogeneous exhibited a large range in δ13CFA values, with variability greatest for fall and spring samples; concentrations were lowest at these times. Historical field boundaries played a role. A downstream sediment mixture (June 2013) was analyzed and found to correspond with source data from August 2012. Sediment mixture data (δ13CFA) for several VLCFAs were found to fall within the source mixing polygons produced by using two cultivated fields and a riparian zone sample as sources.
Conclusions
Variability in δ13CFA values increased in fall and spring, which could affect the number of subsamples required per source. Most fields could be spatially differentiated using a weighted t test, but not necessarily using the same VLCFA chain lengths. Two spatially separated fields with similar crop** histories were difficult to differentiate, but one of the fields was more prone to VLCFA losses. Only one of several source sampling periods led to successful unmixing, suggesting multiple sampling periods for source and/or mixture are necessary. Understanding the spatial and temporal variability affecting δ13CFA values in source sediments is particularly important for tracing studies using biomarkers in producing a representative fingerprint.












Similar content being viewed by others
References
Alewell C, Birkholz A, Meusburger K, Schindler Wildhaber Y, Mabit L (2016) Quantitative sediment source attribution with compound-specific isotope analysis in a C3 plant-dominated catchment (Central Switzerland). Biogeosciences 13:1587–1596
Bilotta GS, Brazier RE (2008) Understanding the influence of suspended solids on water quality and aquatic biota. Water Res 42:2849–2861
Blake WH, Ficken KJ, Taylor P et al (2012) Tracing crop-specific sediment sources in agricultural catchments. Geomorphology 139–140:322–329
Brantley SL, Goldhaber MB, Ragnarsdottir KV (2007) Crossing disciplines and scales to understand the critical zone. Elements 3:307–314
Chandrasekaran M, Kumar AS, Kannathasan K, Venkatesalu V (2010) Fatty-acid composition of some mangroves. Chem Nat Compd 46:92–94
Cochran WG (1977) Sampling techniques, third edn. Wiley, New York
Cooper RJ, Pedentchouk N, Hiscock KM, Disdle P, Krueger T, Rawlins BG (2015) Apportioning sources of organic matter in streambed sediments: an integrated molecular and compound-specific stable isotope approach. Sci Total Environ 520:187–197
Core Team R (2017) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
del Río JC, Prinsen P, Gutiérrez A (2013) A comprehensive characterization of lipids in wheat straw. J Agric Food Chem 61:1904–1913
Ferreira LMM, Celaya R, Falco V, Oliván M, Santos AS, Guedes C, Rodrigues MAM, Osoro K (2010) Evaluation of very long-chain fatty acids and n-alkane epicuticular compounds as markers for estimating diet composition of sheep fed heathland vegetation species. Anim Feed Sci Technol 156:75–88
Gatz DF, Smith L (1995) The standard error of a weighted mean concentration—I. Bootstrap** vs other methods. Atmos Environ 29:1185–1193
Gibbs M (2008) Identifying source soils in contemporary estuarine sediments: a new compound-specific isotope method. Estuar Coasts 31:344–359
Govers G, Van Oost K, Wang Z (2014) Scratching the critical zone: the global footprint of agricultural soil erosion. Procedia Earth Planet Sci 10:313–318
Greenland S, Senn SJ, Rothman KJ, Carlin JB, Poole C, Goodman SN, Altman DG (2016) Statistical tests, P values, confidence intervals, and power: a guide to misinterpretations. Eur J Epidemiol 31:337–350
Guzmán G, Quinton JN, Nearing MA, Mabit L, Gómez JA (2013) Sediment tracers in water erosion studies: current approaches and challenges. J Soils Sediments 13:816–833
Haddadchi A, Ryder DS, Evrard O, Olley J (2013) Sediment fingerprinting in fluvial systems: review of tracers, sediment sources and mixing models. Int J Sediment Res 28:560–578
Hancock GJ, Revill AT (2013) Erosion source discrimination in a rural Australian catchment using compound-specific isotope analysis (CSIA). Hydrol Process 27:923–932
Kanstrup M, Thomsen IK, Andersen AJ, Bogaard A, Christensen BT (2011) Abundance of 13C and 15N in emmer, spelt and naked barley grown on differently manured soils: towards a method for identifying past manuring practice. Rapid Commun Mass Spectrom 25:2879–2887
Kaspar TC, Bland WL (1992) Soil temperature and root growth. Soil Sci 154:290–299
Kleinman P, Sharpley A, Buda A, McDowell R, Allen A (2011) Soil controls of phosphorus in runoff: management barriers and opportunities. Can J Soil Sci 91:329–338
Koiter AJ, Owens PN, Petticrew EL, Lobb DA (2013) The behavioural characteristics of sediment properties and their implications for sediment fingerprinting as an approach for identifying sediment sources in river basins. Earth-Sci Rev 125:24–42
Lake Winnipeg Stewardship Board (2006) Reducing nutrient loading to Lake Winnipeg and its watershed: Our collective responsibility and commitment to action
Lappi H, Alén R (2011) Pyrolysis of vegetable oil soaps—palm, olive, rapeseed and castor oils. J Anal Appl Pyrolysis 91:154–158
Liu Y, Yang W, Yu Z, Lung I, Gharabaghi B (2015) Estimating sediment yield from upland and channel erosion at a watershed scale using SWAT. Water Resour Manag 29:1399–1412
Lockheart MJ, Van Bergen PF, Evershed RP (1997) Variations in the stable carbon isotope compositions of individual lipids from the leaves of modern angiosperms: implications for the study of higher land plant-derived sedimentary organic matter. Proc 17th Int Meet Org Geochem 26:137–153. https://doi.org/10.1016/S0146-6380(96)00135-0
Mabit L, Gibbs M, Mbaye M, Meusburger K, Toloza A, Resch C, Klik A, Swales A, Alewell C (2018) Novel application of compound specific stable isotope (CSSI) techniques to investigate on-site sediment origins across arable fields. Geoderma 316:19–26
Matisoff G, Watson SB, Guo J, Duewiger A, Steely R, (2017) Sediment and nutrient distribution and resuspension in Lake Winnipeg. Sci Total Environ 575:173–186
Owens PN, Batalla RJ, Collins AJ, Gomez B, Hicks DM, Horowitz AJ, Kondolf GM, Marden M, Page MJ, Peacock DH, Petticrew EL, Salomons W, Trustrum NA (2005) Fine-grained sediment in river systems: environmental significance and management issues. River Res Appl 21:693–717. https://doi.org/10.1002/rra.878
Owens PN, Blake WH, Gaspar L, Gateuille D, Koiter AJ, Lobb DA, Petticrew EL, Reiffarth DG, Smith HG, Woodward JC (2016) Fingerprinting and tracing the sources of soils and sediments: earth and ocean science, geoarchaeological, forensic, and human health applications. Earth-Sci Rev 162:1–23
Pasek J (2018) Weights: weighting and weighted statistics. R package version 0.90
Phillips DL, Inger R, Bearhop S, Jackson AL, Moore JW, Parnell AC, Semmens BX, Ward EJ (2014) Best practices for use of stable isotope mixing models in food-web studies. Can J Zool 92:823–835
Pimentel D, Berger B, Filiberto D et al (2004) Water resources: agricultural and environmental issues. BioScience 54:909
Reiffarth DG, Petticrew EL, Owens PN, Lobb DA (2016) Sources of variability in fatty acid (FA) biomarkers in the application of compound-specific stable isotopes (CSSIs) to soil and sediment fingerprinting and tracing: a review. Sci Total Environ 565:8–27
Richter D d B, Mobley ML (2009) Monitoring Earth’s critical zone. Science 326:1067–1068
RStudio Team (2016) RStudio: integrated development for R. RStudio, Inc., Boston
Ruess L, Chamberlain PM (2010) The fat that matters: soil food web analysis using fatty acids and their carbon stable isotope signature. Soil Biol Biochem 42:1898–1910
Ruess L, Häggblom MM, Garcı́a Zapata EJ, Dighton J (2002) Fatty acids of fungi and nematodes—possible biomarkers in the soil food chain? Soil Biol Biochem 34:745–756
Savchenko T, Walley JW, Chehab EW, **ao Y, Kaspi R, Pye MF, Mohamed ME, Lazarus CM, Bostock RM, Dehesh K (2010) Arachidonic acid: an evolutionarily conserved signaling molecule modulates plant stress signaling networks. Plant Cell 22:3193–3205
Schjoerring JK, Wang L (2012) Seasonal variation in nitrogen pools and 15N/13C natural abundances in different tissues of grassland plants. Biogeosciences 9:1583–1595
Smith JA, Mazumder D, Suthers IM, Taylor MD (2013) To fit or not to fit: evaluating stable isotope mixing models using simulated mixing polygons. Methods Ecol Evol 4:612–618
Stock BC, Semmens BX (2016) MixSIAR GUI user manual. Version 3.1
Tiessen KHD, Elliott JA, Yarotski J, Lobb DA, Flaten DN, Glozier NE (2010) Conventional and conservation tillage: influence on seasonal runoff, sediment, and nutrient losses in the Canadian prairies. J Environ Qual 39:964
Upadhayay HR, Bodé S, Griepentrog M, Huygens D, Bajracharya RM, Blake WH, Dercon G, Mabit L, Gibbs M, Semmens BX, Stock BC, Cornelis W, Boeckx P (2017) Methodological perspectives on the application of compound-specific stable isotope fingerprinting for sediment source apportionment. J Soils Sediments 17:1537–1553
Upadhayay HR, Bodé S, Griepentrog M, Bajracharya RM, Blake W, Cornelis W, Boeckx P (2018) Isotope mixing models require individual isotopic tracer content for correct quantification of sediment source contributions. Hydrol Process 32:981–989
Walling DE (2013) The evolution of sediment source fingerprinting investigations in fluvial systems. J Soils Sediments 13:1658–1675
Walling DE, Woodward JC (1992) Use of radiometric fingerprints to derive information on suspended sediment sources. In: Erosion and sediment transport monitoring programmes in river basins, International Association of Hydrological Sciences (IAHS) Publ. XXX. IAHS Press, Wallingford, UK, p 552
Wilkinson SN, Hancock GJ, Bartley R, Hawdon AA, Keen RJ (2013) Using sediment tracing to assess processes and spatial patterns of erosion in grazed rangelands, Burdekin River basin, Australia. Agric Ecosyst Environ 180:90–102
Wood PJ, Armitage PD (1997) Biological effects of fine sediment in the lotic environment. Environ Manag 21:203–217
Zelles L (1997) Phospholipid fatty acid profiles in selected members of soil microbial communities. Chemosphere 35:275–294
Zelles L (1999) Fatty acid patterns of phospholipids and lipopolysaccharides in the characterisation of microbial communities in soil: a review. Biol Fertil Soils 29:111–129
nZhang B, He H, Ding X, Zhang X, Zhang X, Yang X, Filley TR (2012) Soil microbial community dynamics over a maize (Zea mays L.) growing season under conventional- and no-tillage practices in a rainfed agroecosystem. Soil Tillage Res 124:153–160
Acknowledgments
We would like to thank the two anonymous reviewers for their time and effort in providing valuable feedback which we believe enhanced the manuscript. We would also like to thank Patrick Laceby for all his time, suggestions, and feedback, which further improved the quality of the manuscript.
This work is funded by a NSERC Strategic Grant STPGP 413426-2011—Development of environmental fingerprinting techniques for sources of sediment and associated phosphorus within agricultural watersheds of Canada—to D.A.L., P.N.O., and E.L.P. The purchasing of analytical equipment used in this project was made possible through the Canada Foundation for Innovation and the Western Economic Diversification Program. Analytical laboratory support was provided by Northern Analytical Laboratory Services (NALS) at UNBC. Additional thanks to the many undergraduate and graduate students at the University of Manitoba and UNBC who aided in the collection and processing of soil and sediment samples. This project would not have been possible without them.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: J. Patrick Laceby
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 6167 kb)
Rights and permissions
About this article
Cite this article
Reiffarth, D.G., Petticrew, E.L., Owens, P.N. et al. Spatial differentiation of cultivated soils using compound-specific stable isotopes (CSSIs) in a temperate agricultural watershed in Manitoba, Canada. J Soils Sediments 19, 3411–3426 (2019). https://doi.org/10.1007/s11368-019-02406-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-019-02406-3