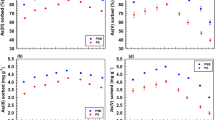
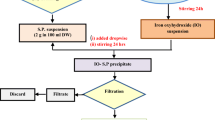
Abstract
The impact of arsenic (As) contamination of water is an ongoing concern worldwide with As released from anthropogenic activities including mining and agriculture. Biosorption is a promising As treatment methodology used currently for arsenate (As(V)) sorption from water. The biosorbent was developed by a simple and inexpensive treatment of coating of canola straw particles with iron hydroxides. The modification procedure was optimized with consideration of the concentration of iron solution, pH of modification process, and sonication time. A higher concentration of iron and lower pH led to an improved sorption capacity of the iron-loaded canola straw (ICS), while impacts of sonication time were not conclusive. Pareto analyses indicated that the magnitude of the effect of the pH was higher than that of the iron concentration. Overall, the maximum As(V) sorption capacity of the ICS was 5.5 mg/g for an 0.25 M FeCl3 solution concentration at pH 3. Analysis of kinetic data showed that the sorption processes of As(V) followed pseudo-second order and Elovich mechanisms, while sorption isotherm data were best represented by Freundlich and Temkin isotherm models. Studying the effect of ionic strength using NaCl suggested that the inner-sphere complex was the probable sorption mechanism. The thermodynamic parameters including ΔS°, ΔH°, and ΔG° showed that the As(V) sorption was thermodynamically favorable and spontaneous. Arsenic K-edge X-ray absorption near edge structure (XANES) spectroscopy indicated that no reaction to As(III) occurred during the sorption of As(V) using the optimum ICS biosorbent. The evolutionary polynomial regression (EPR) approach was able to closely match predicted vs. experimental sorption capacities (R2 = 0.95). Overall, the improved understanding of the biosorbent’s capability for removal of As(V) will be beneficial for assessment of its use for treatment of various water and wastewater matrices. In addition, knowledge gained from this research can assist in the understanding of sorption capacities of a variety of other biosorbents.








Similar content being viewed by others
Data availability
Not applicable.
References
Ahmadi J, Kahforoushan D, Fatehifar E et al (2016) Drying of Urmia Lake: modeling of level fluctuations. Environ Health Eng Manag 3:23–28
Ahmadi M, Zoroufchi Benis K, Faraji M, Shakerkhatibi M, Aliashrafi A (2019) Process performance and multi-kinetic modeling of a membrane bioreactor treating actual oil refinery wastewater. J Water Process Eng 28:115–122. https://doi.org/10.1016/J.JWPE.2019.01.010
Alam R, McPhedran K (2019) Applications of biological sulfate reduction for remediation of arsenic – A review. Chemosphere 222:932–944. https://doi.org/10.1016/J.CHEMOSPHERE.2019.01.194
Alchouron J, Navarathna C, Chludil HD, Dewage NB, Perez F, Hassan EB, Pittman Jr CU, Vega AS, Mlsna TE (2020) Assessing South American Guadua chacoensis bamboo biochar and Fe3O4 nanoparticle dispersed analogues for aqueous arsenic(V) remediation. Sci Total Environ 706:135943. https://doi.org/10.1016/j.scitotenv.2019.135943
Alkurdi SSA, Herath I, Bundschuh J, al-Juboori RA, Vithanage M, Mohan D (2019) Biochar versus bone char for a sustainable inorganic arsenic mitigation in water: what needs to be done in future research? Environ Int 127:52–69. https://doi.org/10.1016/J.ENVINT.2019.03.012
Aranda-García E, Cristiani-Urbina E (2019) Effect of pH on hexavalent and total chromium removal from aqueous solutions by avocado shell using batch and continuous systems. Environ Sci Pollut Res 26:3157–3173. https://doi.org/10.1007/s11356-017-0248-z
Aryal M, Ziagova M, Liakopoulou-Kyriakides M (2010) Study on arsenic biosorption using Fe(III)-treated biomass of Staphylococcus xylosus. Chem Eng J 162:178–185. https://doi.org/10.1016/J.CEJ.2010.05.026
Baig JA, Kazi TG, Shah AQ, Kandhro GA, Afridi HI, Khan S, Kolachi NF (2010) Biosorption studies on powder of stem of Acacia nilotica: removal of arsenic from surface water. J Hazard Mater 178:941–948. https://doi.org/10.1016/J.JHAZMAT.2010.02.028
Baker MJ, Trevisan J, Bassan P, Bhargava R, Butler HJ, Dorling KM, Fielden PR, Fogarty SW, Fullwood NJ, Heys KA, Hughes C, Lasch P, Martin-Hirsch PL, Obinaju B, Sockalingum GD, Sulé-Suso J, Strong RJ, Walsh MJ, Wood BR, Gardner P, Martin FL (2014) Using Fourier transform IR spectroscopy to analyze biological materials. Nat Protoc 9:1771–1791. https://doi.org/10.1038/nprot.2014.110
Baltpurvins KA, Burns RC, Lawrance GA, Stuart AD (1996) Effect of pH and anion type on the aging of freshly precipitated iron(III) hydroxide sludges. Environ Sci Technol 30:939–944. https://doi.org/10.1021/es950401s
Benjamin MM, Sletten RS, Bailey RP, Bennett T (1996) Sorption and filtration of metals using iron-oxide-coated sand. Water Res 30:2609–2620. https://doi.org/10.1016/S0043-1354(96)00161-3
Bonakdari H, Gholami A, Sattar AMA, Gharabaghi B (2020) Development of robust evolutionary polynomial regression network in the estimation of stable alluvial channel dimensions. Geomorphology 350:106895. https://doi.org/10.1016/j.geomorph.2019.106895
Carijo PM, dos Reis GS, Lima ÉC, Oliveira MLS, Dotto GL (2019) Functionalization of corn stover with 3-aminopropyltrietoxysilane to uptake Reactive Red 141 from aqueous solutions. Environ Sci Pollut Res 26:32198–32208. https://doi.org/10.1007/s11356-019-06386-2
Cornell RM, Giovanoli R, Schneider W (1989) Review of the hydrolysis of iron(III) and the crystallization of amorphous iron(III) hydroxide hydrate. J Chem Technol Biotechnol 46:115–134. https://doi.org/10.1002/jctb.280460204
Corrales RCNR, Mendes FM, Perrone C, Sant’Anna C, de Souza W, Abud Y, Bon EPS, Ferreira-Leitão V (2012) Structural evaluation of sugar cane bagasse steam pretreated in the presence of CO2 and SO2. Biotechnol Biofuels 5:36. https://doi.org/10.1186/1754-6834-5-36
Davodia M, Alidadib H, Ramezanib A et al (2019) Study of the removal efficiency of arsenic from aqueous solutions using Melia azedarach sawdust modified with FeO: isotherm and kinetic studies. Desalin Water Treat 137:292–299
Dhanasekaran P, Satya Sai PM, Anand Babu C, Krishna Prabhu R, Rajan KK (2016) Arsenic removal from groundwater by Anjili tree sawdust impregnated with ferric hydroxide and activated alumina. Water Sci Technol Water Supply 16:115–127. https://doi.org/10.2166/ws.2015.119
Dos Reis GS, Wilhelm M, Silva TCDA et al (2016) The use of design of experiments for the evaluation of the production of surface rich activated carbon from sewage sludge via microwave and conventional pyrolysis. Appl Therm Eng 93:590–597. https://doi.org/10.1016/j.applthermaleng.2015.09.035
El-Moselhy MM, Ates A, Çelebi A (2017) Synthesis and characterization of hybrid iron oxide silicates for selective removal of arsenic oxyanions from contaminated water. J Colloid Interface Sci 488:335–347. https://doi.org/10.1016/J.JCIS.2016.11.003
Gautam SB, Alam MS, Kamsonlian S (2017) Adsorptive removal of As(III) from aqueous solution by raw coconut husk and iron impregnated coconut husk: kinetics and equilibrium analyses. Int J Chem React Eng 15. https://doi.org/10.1515/ijcre-2016-0097
Giustolisi O, Savic DA (2006) A symbolic data-driven technique based on evolutionary polynomial regression. J Hydroinf 8:207–222. https://doi.org/10.2166/hydro.2006.020b
Grossl PR, Eick M, Sparks DL et al (1997) Arsenate and chromate retention mechanisms on goethite. 2. Kinetic evaluation using a pressure-jump relaxation technique. Environ Sci Technol. https://doi.org/10.1021/es950654l
He R, Peng Z, Lyu H, Huang H, Nan Q, Tang J (2018) Synthesis and characterization of an iron-impregnated biochar for aqueous arsenic removal. Sci Total Environ 612:1177–1186. https://doi.org/10.1016/J.SCITOTENV.2017.09.016
Hokkanen S, Repo E, Lou S, Sillanpää M (2015) Removal of arsenic(V) by magnetic nanoparticle activated microfibrillated cellulose. Chem Eng J 260:886–894. https://doi.org/10.1016/J.CEJ.2014.08.093
Hu X, Ding Z, Zimmerman AR, Wang S, Gao B (2015) Batch and column sorption of arsenic onto iron-impregnated biochar synthesized through hydrolysis. Water Res 68:206–216. https://doi.org/10.1016/J.WATRES.2014.10.009
Huang Z, Liang X, Hu H, Gao L, Chen Y, Tong Z (2009) Influence of mechanical activation on the graft copolymerization of sugarcane bagasse and acrylic acid. Polym Degrad Stab 94:1737–1745. https://doi.org/10.1016/j.polymdegradstab.2009.06.023
Huertaa RR, Saldañaa MDA (2018) Biorefining of canola straw using pressurized aqueous ethanol: nanosized cellulose. In: Proceedings of the J. Supercrit. Fluids
Jang J, Lee DS (2018) Magnetite nanoparticles supported on organically modified montmorillonite for adsorptive removal of iodide from aqueous solution: optimization using response surface methodology. Sci Total Environ 615:549–557. https://doi.org/10.1016/j.scitotenv.2017.09.324
** H, Capareda S, Chang Z, Gao J, Xu Y, Zhang J (2014) Biochar pyrolytically produced from municipal solid wastes for aqueous As(V) removal: adsorption property and its improvement with KOH activation. Bioresour Technol 169:622–629. https://doi.org/10.1016/J.BIORTECH.2014.06.103
Kumar KV (2006) Linear and non-linear regression analysis for the sorption kinetics of methylene blue onto activated carbon. J Hazard Mater 137:1538–1544. https://doi.org/10.1016/j.jhazmat.2006.04.036
Kumar A, Gurian PL, Bucciarelli-Tieger RH, Mitchell-Blackwood J (2008) Iron oxide-coated fibrous sorbents for arsenic removal. J Am Water Works Assoc 100:151–1A4. https://doi.org/10.1002/j.1551-8833.2008.tb09611.x
Laucelli D, Giustolisi O (2011) Scour depth modelling by a multi-objective evolutionary paradigm. Environ Model Softw 26:498–509. https://doi.org/10.1016/j.envsoft.2010.10.013
Leite AB, Saucier C, Lima EC, dos Reis GS, Umpierres CS, Mello BL, Shirmardi M, Dias SLP, Sampaio CH (2018) Activated carbons from avocado seed: optimisation and application for removal of several emerging organic compounds. Environ Sci Pollut Res 25:7647–7661. https://doi.org/10.1007/s11356-017-1105-9
León G, García F, Miguel B, Bayo J (2016) Equilibrium, kinetic and thermodynamic studies of methyl orange removal by adsorption onto granular activated carbon. Desalin Water Treat 57:17104–17117. https://doi.org/10.1080/19443994.2015.1072063
Lian F, Liu X, Gao M et al (2020) Effects of Fe-Mn-Ce oxide–modified biochar on As accumulation, morphology, and quality of rice (Oryza sativa L.). Environ Sci Pollut Res:1–12. https://doi.org/10.1007/s11356-020-08355-6
Lin L, Qiu W, Wang D, Huang Q, Song Z, Chau HW (2017) Arsenic removal in aqueous solution by a novel Fe-Mn modified biochar composite: Characterization and mechanism. Ecotoxicol Environ Saf 144:514–521. https://doi.org/10.1016/J.ECOENV.2017.06.063
Liu C-H, Chuang Y-H, Chen T-Y, Tian Y, Li H, Wang MK, Zhang W (2015) Mechanism of arsenic adsorption on magnetite nanoparticles from water: thermodynamic and spectroscopic studies. Environ Sci Technol 49:7726–7734. https://doi.org/10.1021/acs.est.5b00381
Lo S-L, Jeng H-T, Lai C-H (1997) Characteristics and adsorption properties of iron-coated sand. Water Sci Technol 35:63–70. https://doi.org/10.1016/S0273-1223(97)00115-7
Lunge S, Singh S, Sinha A (2014) Magnetic iron oxide (Fe3O4) nanoparticles from tea waste for arsenic removal. J Magn Magn Mater 356:21–31. https://doi.org/10.1016/J.JMMM.2013.12.008
Luo M, Lin H, He Y, Li B, Dong Y, Wang L (2019) Efficient simultaneous removal of cadmium and arsenic in aqueous solution by titanium-modified ultrasonic biochar. Bioresour Technol 284:333–339. https://doi.org/10.1016/J.BIORTECH.2019.03.108
Mahmood T, Aslam M, Naeem A, Siddique T, Din SU (2018) Adsorption of As (III) from aqueous solution onto iron impregnated used tea activated carbon: equilibrium, kinetic and thermodynamic study. J Chil Chem Soc 63:3855–3866
Marín-Rangel VM, Cortés-Martínez R, Cuevas Villanueva RA, Garnica-Romo MG, Martínez-Flores HE (2012) As (V) Biosorption in an aqueous solution using chemically treated lemon (Citrus aurantifolia Swingle) residues. J Food Sci 77:T10–T14. https://doi.org/10.1111/j.1750-3841.2011.02466.x
Martinez-Vargas S, Martínez AI, Hernández-Beteta EE et al (2018) As(III) and As(V) adsorption on manganese ferrite nanoparticles. J Mol Struct 1154:524–534. https://doi.org/10.1016/J.MOLSTRUC.2017.10.076
Murugesan GS, Sathishkumar M, Swaminathan K (2006) Arsenic removal from groundwater by pretreated waste tea fungal biomass. Bioresour Technol 97:483–487. https://doi.org/10.1016/J.BIORTECH.2005.03.008
Nadeem R, Manzoor Q, Iqbal M, Nisar J (2016) Biosorption of Pb(II) onto immobilized and native Mangifera indica waste biomass. J Ind Eng Chem 35:185–194. https://doi.org/10.1016/j.jiec.2015.12.030
Najafzadeh M, Shiri J, Rezaie-Balf M (2018) New expression-based models to estimate scour depth at clear water conditions in rectangular channels. Mar Georesour Geotechnol 36:227–235. https://doi.org/10.1080/1064119X.2017.1303009
Nashine A, Tembhurkar A (2016) Iron oxide impregnated sugarcane bagasse waste material as sorbent for As(III) removal from water: kinetic, equilibrium and thermodynamic studies. J Water Supply Res Technol 65:645–652. https://doi.org/10.2166/aqua.2016.054
Nguyen TH, Pham TH, Nguyen Thi HT, Nguyen TN, Nguyen MV, Tran Dinh T, Nguyen MP, Do TQ, Phuong T, Hoang TT, Mai Hung TT, Thi VHT (2019) Synthesis of iron-modified biochar derived from rice straw and its application to arsenic removal. J Chem 2019. https://doi.org/10.1155/2019/5295610
Niazi NK, Bibi I, Shahid M, Ok YS, Shaheen SM, Rinklebe J, Wang H, Murtaza B, Islam E, Farrakh Nawaz M, Lüttge A (2018a) Arsenic removal by Japanese oak wood biochar in aqueous solutions and well water: investigating arsenic fate using integrated spectroscopic and microscopic techniques. Sci Total Environ 621:1642–1651. https://doi.org/10.1016/J.SCITOTENV.2017.10.063
Niazi NK, Bibi I, Shahid M, Ok YS, Burton ED, Wang H, Shaheen SM, Rinklebe J, Lüttge A (2018b) Arsenic removal by perilla leaf biochar in aqueous solutions and groundwater: an integrated spectroscopic and microscopic examination. Environ Pollut 232:31–41. https://doi.org/10.1016/J.ENVPOL.2017.09.051
Niu CH, Volesky B, Cleiman D (2007) Biosorption of arsenic (V) with acid-washed crab shells. Water Res 41:2473–2478. https://doi.org/10.1016/J.WATRES.2007.03.013
Ociński D, Mazur P (2020) Highly efficient arsenic sorbent based on residual from water deironing – sorption mechanisms and column studies. J Hazard Mater 382. https://doi.org/10.1016/j.jhazmat.2019.121062
Pahlavanzadeh S, Zoroufchi Benis K, Shakerkhatibi M, Karimi Jashni A, Taleb Beydokhti N, Alizadeh Kordkandi S (2018) Performance and kinetic modeling of an aerated submerged fixed-film bioreactor for BOD and nitrogen removal from municipal wastewater. J Environ Chem Eng 6:6154–6164. https://doi.org/10.1016/J.JECE.2018.09.045
Pehlivan E, Tran TH, Ouédraogo WKI, Schmidt C, Zachmann D, Bahadir M (2013) Removal of As(V) from aqueous solutions by iron coated rice husk. Fuel Process Technol 106:511–517. https://doi.org/10.1016/J.FUPROC.2012.09.021
Penke YK, Yadav AK, Sinha P, Malik I, Ramkumar J, Kar KK (2020) Arsenic remediation onto redox and photo-catalytic/electrocatalytic Mn-Al-Fe impregnated rGO: Sustainable aspects of sludge as supercapacitor. Chem Eng J 390:124000. https://doi.org/10.1016/j.cej.2019.124000
Phuengprasop T, Sittiwong J, Unob F (2011) Removal of heavy metal ions by iron oxide coated sewage sludge. J Hazard Mater 186:502–507. https://doi.org/10.1016/J.JHAZMAT.2010.11.065
Podder MS, Majumder CB (2016) Investigation on elimination of As(III) and As(V) from wastewater using bacterial biofilm supported on sawdust/MnFe2O4 composite. Water Conserv Sci Eng 1:21–48. https://doi.org/10.1007/s41101-016-0002-2
Premarathna KSD, Rajapaksha AU, Sarkar B, Kwon EE, Bhatnagar A, Ok YS, Vithanage M (2019) Biochar-based engineered composites for sorptive decontamination of water: a review. Chem Eng J 372:536–550. https://doi.org/10.1016/J.CEJ.2019.04.097
Pronyk C, Mazza G (2012) Fractionation of triticale, wheat, barley, oats, canola, and mustard straws for the production of carbohydrates and lignins. Bioresour Technol 106:117–124. https://doi.org/10.1016/j.biortech.2011.11.071
Qu G, Huang X, Yin Q, Ning P (2014) Dissolution of garlic stem in the 1-butylrpridinium bromide ionic liquid. J Chem Eng Jpn 47:435–441. https://doi.org/10.1252/jcej.13we163
Ramesh A, Hasegawa H, Maki T, Ueda K (2007) Adsorption of inorganic and organic arsenic from aqueous solutions by polymeric Al/Fe modified montmorillonite. Sep Purif Technol 56:90–100. https://doi.org/10.1016/J.SEPPUR.2007.01.025
Raul PK, Devi RR, Umlong IM, Thakur AJ, Banerjee S, Veer V (2014) Iron oxide hydroxide nanoflower assisted removal of arsenic from water. Mater Res Bull 49:360–368. https://doi.org/10.1016/J.MATERRESBULL.2013.09.015
Ravel B, Newville M (2005) ATHENA, ARTEMIS, HEPHAESTUS: data analysis for X-ray absorption spectroscopy using IFEFFIT. In: Journal of Synchrotron Radiation. International Union of Crystallography, pp 537–541
Roy P, Mondal NKK, Das K (2014) Modeling of the adsorptive removal of arsenic: a statistical approach. J Environ Chem Eng 2:585–597. https://doi.org/10.1016/j.jece.2013.10.014
Sahu UK, Sahu S, Mahapatra SS, Patel RK (2019) Synthesis and characterization of magnetic bio-adsorbent developed from Aegle marmelos leaves for removal of As(V) from aqueous solutions. Environ Sci Pollut Res 26:946–958. https://doi.org/10.1007/s11356-018-3643-1
Schwertmann U, Murad E (1983) Effect of pH on the formation of Goethite and Hematite from Ferrihydrite. Clay Clay Miner 31:277–284. https://doi.org/10.1346/CCMN.1983.0310405
Shakerkhatibi M, Mohammadi N, Zoroufchi Benis K et al (2015) Using ANN and EPR models to predict carbon monoxide concentrations in urban area of Tabriz. Environ Heal Eng Manag J 2:117–122
Shakoor MB, Niazi NK, Bibi I, Shahid M, Sharif F, Bashir S, Shaheen SM, Wang H, Tsang DCW, Ok YS, Rinklebe J (2018) Arsenic removal by natural and chemically modified water melon rind in aqueous solutions and groundwater. Sci Total Environ 645:1444–1455. https://doi.org/10.1016/J.SCITOTENV.2018.07.218
Siddiqui SI, Chaudhry SA (2017) Iron oxide and its modified forms as an adsorbent for arsenic removal: a comprehensive recent advancement. Process Saf Environ Prot 111:592–626. https://doi.org/10.1016/j.psep.2017.08.009
StatCan (2017) Production of principal field crops. https://www150.statcan.gc.ca/n1/daily-quotidien/161206/dq161206b-eng.htm. Accessed 25 Dec 2019
Sumathi T, Alagumuthu G (2014) Adsorption studies for arsenic removal using activated Moringa oleifera. Int J Chem Eng 2014:1–6. https://doi.org/10.1155/2014/430417
Tran HN, You SJ, Hosseini-Bandegharaei A, Chao HP (2017) Mistakes and inconsistencies regarding adsorption of contaminants from aqueous solutions: a critical review. Water Res 120:88–116
Tsang S, Phu F, Baum MM, Poskrebyshev GA (2007) Determination of phosphate/arsenate by a modified molybdenum blue method and reduction of arsenate by S2O42−. Talanta 71:1560–1568. https://doi.org/10.1016/J.TALANTA.2006.07.043
Uddin MJ, Jeong Y-K (2020) Review: Efficiently performing periodic elements with modern adsorption technologies for arsenic removal. Environ Sci Pollut Res Int 27(32):39888–39912. https://doi.org/10.1007/s11356-020-10323-z
Ungureanu G, Santos S, Boaventura R, Botelho C (2015) Arsenic and antimony in water and wastewater: overview of removal techniques with special reference to latest advances in adsorption. J Environ Manag 151:326–342
Vieira BRC, Pintor AMA, Boaventura RAR, Botelho CMS, Santos SCR (2017) Arsenic removal from water using iron-coated seaweeds. J Environ Manag 192:224–233. https://doi.org/10.1016/J.JENVMAN.2017.01.054
Wang J, Guo X (2020) Adsorption kinetic models: physical meanings, applications, and solving methods. J Hazard Mater 390:122156. https://doi.org/10.1016/J.JHAZMAT.2020.122156
Wang A, Zhou K, Zhang X, Zhou D, Peng C, Chen W (2020) Arsenic removal from highly-acidic wastewater with high arsenic content by copper-chloride synergistic reduction. Chemosphere 238:124675. https://doi.org/10.1016/j.chemosphere.2019.124675
Wongrod S, Simon S, van Hullebusch ED, Lens PNL, Guibaud G (2019) Assessing arsenic redox state evolution in solution and solid phase during As(III) sorption onto chemically-treated sewage sludge digestate biochars. Bioresour Technol 275:232–238. https://doi.org/10.1016/J.BIORTECH.2018.12.056
Wu Y, Li B, Feng S, Mi XM, Jiang JL (2009) Adsorption of Cr(VI) and As(III) on coaly activated carbon in single and binary systems. Desalination 249:1067–1073. https://doi.org/10.1016/J.DESAL.2009.06.049
Wu C, Huang L, Xue SG, Huang YY, Hartley W, Cui MQ, Wong MH (2017) Arsenic sorption by red mud-modified biochar produced from rice straw. Environ Sci Pollut Res 24:18168–18178. https://doi.org/10.1007/s11356-017-9466-7
Wu Z, Su X, Lin Z, Owens G, Chen Z (2019) Mechanism of As(V) removal by green synthesized iron nanoparticles. J Hazard Mater 379:120811. https://doi.org/10.1016/J.JHAZMAT.2019.120811
Yamil YL, Georgin J, dos Reis GS et al (2020) Utilization of Pacara Earpod tree (Enterolobium contortisilquum) and Ironwood (Caesalpinia leiostachya) seeds as low-cost biosorbents for removal of basic fuchsin. Environ Sci Pollut Res 27:33307–33320. https://doi.org/10.1007/s11356-020-09471-z
Yao Y, Bing H, Feifei X, **aofeng C (2011) Equilibrium and kinetic studies of methyl orange adsorption on multiwalled carbon nanotubes. Chem Eng J 170:82–89. https://doi.org/10.1016/j.cej.2011.03.031
Zama EF, Zhu Y-G, Reid BJ, Sun G-X (2017) The role of biochar properties in influencing the sorption and desorption of Pb(II), Cd(II) and As(III) in aqueous solution. J Clean Prod 148:127–136. https://doi.org/10.1016/j.jclepro.2017.01.125
Zeng L (2003) A method for preparing silica-containing iron(III) oxide adsorbents for arsenic removal. Water Res 37:4351–4358. https://doi.org/10.1016/S0043-1354(03)00402-0
Zeng L (2004) Arsenic adsorption from aqueous solutions on an Fe(III)-Si binary oxide adsorbent. Water Qual Res J Can 39:267–275. https://doi.org/10.2166/wqrj.2004.037
Zhang G, Liu H, Liu R, Qu J (2009) Adsorption behavior and mechanism of arsenate at Fe–Mn binary oxide/water interface. J Hazard Mater 168:820–825. https://doi.org/10.1016/J.JHAZMAT.2009.02.137
Zhang F, Wang X, **onghui J, Ma L (2016) Efficient arsenate removal by magnetite-modified water hyacinth biochar. Environ Pollut 216:575–583. https://doi.org/10.1016/J.ENVPOL.2016.06.013
Zhang H, Zhao Z, Kang P, Wang Y, Feng J, Jia J, Zhang Z (2018) Biological nitrogen removal and metabolic characteristics of a novel aerobic denitrifying fungus Hanseniaspora uvarum strain KPL108. Bioresour Technol 267:569–577. https://doi.org/10.1016/j.biortech.2018.07.073
Zhou Z, Liu Y, Liu S, Liu HY, Zeng GM, Tan XF, Yang CP, Ding Y, Yan ZL, Cai XX (2017) Sorption performance and mechanisms of arsenic(V) removal by magnetic gelatin-modified biochar. Chem Eng J 314:223–231. https://doi.org/10.1016/J.CEJ.2016.12.113
Zhou Y, Cao S, ** C, Li X, Zhang L, Wang G, Chen Z (2019) A novel Fe3O4/graphene oxide/citrus peel-derived bio-char based nanocomposite with enhanced adsorption affinity and sensitivity of ciprofloxacin and sparfloxacin. Bioresour Technol 292:121951. https://doi.org/10.1016/J.BIORTECH.2019.121951
Zhu N, Yan T, Qiao J, Cao H (2016) Adsorption of arsenic, phosphorus and chromium by bismuth impregnated biochar: adsorption mechanism and depleted adsorbent utilization. Chemosphere 164:32–40. https://doi.org/10.1016/J.CHEMOSPHERE.2016.08.036
Zoroufchi Benis K, Damuchali AM, Soltan J, McPhedran K (2020a) Treatment of aqueous arsenic – a review of biochar modification methods. Sci Total Environ:139750. https://doi.org/10.1016/j.scitotenv.2020.139750
Zoroufchi Benis K, Motalebi Damuchali A, McPhedran KN, Soltan J (2020b) Treatment of aqueous arsenic – a review of biosorbent preparation methods. J Environ Manag 273:111126. https://doi.org/10.1016/j.jenvman.2020.111126
Acknowledgments
This research was financially supported by two NSERC Discovery Grants (K. McPhedran and J. Soltan). We would also like to extend our thanks to the College of Graduate and Postdoctoral Studies, the University of Saskatchewan, for financial support in the form of a Dean’s Scholarship (K. Benis).
The XANES and XRD analyses were performed using BioXAS-Spectroscopy and BXDS-WLE beamlines at the Canadian Light Source, which is supported by the Canada Foundation for Innovation (CFI), the Natural Sciences and Engineering Research Council (NSERC), the National Research Council (NRC), the Canadian Institutes of Health Research (CIHR), the Government of Saskatchewan, and the University of Saskatchewan. The authors are thankful to beamline scientists Roman Chernikov, Emilio Heredia, Beatriz Moreno, and Graham King for their contribution.
Funding
This research was financially supported by two NSERC Discovery Grants (K. McPhedran and J. Soltan). We would also like to extend our thanks to the College of Graduate and Postdoctoral Studies, the University of Saskatchewan, for financial support in the form of a Dean’s Scholarship (K. Benis).
The XANES and XRD analyses were performed using BioXAS-Spectroscopy and BXDS-WLE beamlines at the Canadian Light Source, which is supported by the Canada Foundation for Innovation (CFI), the Natural Sciences and Engineering Research Council (NSERC), the National Research Council (NRC), the Canadian Institutes of Health Research (CIHR), the Government of Saskatchewan, and the University of Saskatchewan.
Author information
Authors and Affiliations
Contributions
KZB performed all experiments with assistance from MS. KZB, JS, and KNM were major contributors to the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Ethical approval
Not applicable
Consent to participate
Not applicable
Consent to publish
Not applicable
Additional information
Responsible Editor: Tito Roberto Cadaval Jr
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 1157 kb).
Rights and permissions
About this article
Cite this article
Zoroufchi Benis, K., Shakouri, M., McPhedran, K. et al. Enhanced arsenate removal by Fe-impregnated canola straw: assessment of XANES solid-phase speciation, impacts of solution properties, sorption mechanisms, and evolutionary polynomial regression (EPR) models. Environ Sci Pollut Res 28, 12659–12676 (2021). https://doi.org/10.1007/s11356-020-11140-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-11140-0