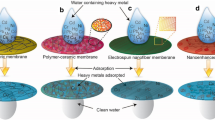
Abstract
Heavy metals like mercury, chromium, lead and copper present in groundwater at lower concentrations cause severe health issues and can even be fatal when consumed. The biopigment/biopolymer melanin can be reaped from different sources like bacterium, fungus, and human hair. It has excellent heavy metal ion scavenging property and can be exploited for non-biological applications, substantially including water purification. In this work, melanin nanoparticles were derived from the marine bacterium Pseudomonas stutzeri and were coated onto hydrophobic polyvinylidene fluoride (PVDF) membrane as a support, for batch and continuous removal of heavy metal studies. Batch studies on the effect of pH, temperature and adsorbate dose and continuous adsorption studies on the effect of flow rate, adsorbate and adsorbent mass loadings were carried out by using biosynthesised melanin-coated PVDF membranes for the removal of Hg(II), Cr(VI), Pb(II) and Cu(II). Scanning electron microscope (SEM) images revealed the surface morphology, Fourier-transform infrared spectroscopy (FTIR) and energy-dispersive X-ray spectroscopy (EDS) deciphered the chemical characteristics of melanin-coated PVDF membranes before and after adsorption. Contact angle measurement confirmed the improvement in hydrophilicity of PVDF membrane upon coating with melanin. The maximum removal percentages of heavy metals achieved by melanin-coated PVDF membranes under batch mode operation were 87.6%, 88.45%, 91.8% and 95.8% for mercury, chromium, lead and copper, respectively optimised at 318 K and pH of 3 for chromium and 5 for other metals. However, the continuous mode of operation with a flow rate of 0.5 mL/min having 1 mg/L of heavy metal solution concentration exposed to 50 mg of melanin loading with a working volume of 200 mL showed better removal efficiencies compared with batch mode. The dynamic studies using Thomas and Yoon–Nelson models described the transient stage of the breakthrough curve and the model constants were calculated for column design and scale-up.












Similar content being viewed by others
References
Acharya J, Sahu JN, Mohanty CR, Meikap BC (2009) Removal of lead(II) from wastewater by activated carbon developed from Tamarind wood by zinc chloride activation. Chem Eng J 149:249–262. https://doi.org/10.1016/j.cej.2008.10.029
Ahmad AA, Hameed BH (2010) Fixed-bed adsorption of reactive azo dye onto granular activated carbon prepared from waste. J Hazard Mater 175:298–303. https://doi.org/10.1016/j.jhazmat.2009.10.003
Anbia M, Amirmahmoodi S (2016) Removal of Hg (II) and Mn (II) from aqueous solution using nanoporous carbon impregnated with surfactants. Arab J Chem 9:S319–S325. https://doi.org/10.1016/j.arabjc.2011.04.004
Aydin YA, Aksoy ND (2009) Adsorption of chromium on chitosan: optimization, kinetics and thermodynamics. Chem Eng J 151:188–194. https://doi.org/10.1016/j.cej.2009.02.010
Banerjee S, Joshi SR, Mandal T, Halder G (2018) Application of zirconium caged activated biochar alginate beads towards deionization of Cr(VI) laden water in a fixed bed column reactor. J Environ Chem Eng 6:4018–4029. https://doi.org/10.1016/J.JECE.2018.06.011
Boributh S, Chanachai A, Jiraratananon R (2009) Modification of PVDF membrane by chitosan solution for reducing protein fouling. J Membr Sci 342:97–104. https://doi.org/10.1016/j.memsci.2009.06.022
Chen S, Xue C, Wang J et al (2009) Adsorption of Pb (II) and Cd (II) by squid Ommastrephes bartrami melanin. Bioinorg Chem Appl 2009
Chen X, He Y, Fan Y et al (2019) Nature-inspired polyphenol chemistry to fabricate halloysite nanotubes decorated PVDF membrane for the removal of wastewater. Sep Purif Technol 212:326–336. https://doi.org/10.1016/j.seppur.2018.11.036
Darwish NB, Abdulgader HA, AlRomaih H, Alalawi A (2019) Effect of ultrafiltration membranes modifications by chitosan on humic acid fouling. J Water Process Eng 27:32–36. https://doi.org/10.1016/j.jwpe.2018.11.008
Demiral H, Güngör C (2016) Adsorption of copper(II) from aqueous solutions on activated carbon prepared from grape bagasse. J Clean Prod 124:103–113. https://doi.org/10.1016/j.jclepro.2016.02.084
Dotzauer DM, Dai J, Sun L, Bruening ML (2006) Catalytic membranes prepared using layer-by-layer adsorption of polyelectrolyte/metal nanoparticle films in porous supports. Nano Lett 6:2268–2272
Dwivedi CP, Sahu JN, Mohanty CR et al (2008) Column performance of granular activated carbon packed bed for Pb(II) removal. J Hazard Mater 156:596–603
Gao C, Zhang X, Yuan Y et al (2018) Removal of hexavalent chromium ions by core-shell sand/Mg-layer double hydroxides (LDHs) in constructed rapid infiltration system. Ecotoxicol Environ Saf 166:285–293. https://doi.org/10.1016/J.ECOENV.2018.09.083
Gupta VK, Rastogi A, Nayak A (2010) Adsorption studies on the removal of hexavalent chromium from aqueous solution using a low cost fertilizer industry waste material. J Colloid Interface Sci 342:135–141. https://doi.org/10.1016/j.jcis.2009.09.065
Han R, Zou L, Zhao X et al (2009) Characterization and properties of iron oxide-coated zeolite as adsorbent for removal of copper(II) from solution in fixed bed column. Chem Eng J 149:123–131. https://doi.org/10.1016/j.cej.2008.10.015
Kataria N, Garg VK (2018) Optimization of Pb (II) and Cd (II) adsorption onto ZnO nanoflowers using central composite design: isotherms and kinetics modelling. J Mol Liq 271:228–239. https://doi.org/10.1016/J.MOLLIQ.2018.08.135
Kundu S, Gupta AK (2006) Arsenic adsorption onto iron oxide-coated cement (IOCC): regression analysis of equilibrium data with several isotherm models and their optimization. Chem Eng J 122:93–106
Manirethan V, Raval K, Rajan R et al (2018) Kinetic and thermodynamic studies on the adsorption of heavy metals from aqueous solution by melanin nanopigment obtained from marine source: Pseudomonas stutzeri. J Environ Manag 214:315–324. https://doi.org/10.1016/j.jenvman.2018.02.084
Mikhaylov VI, Maslennikova TP, Krivoshapkina EF et al (2018) Express Al/Fe oxide–oxyhydroxide sorbent systems for Cr(VI) removal from aqueous solutions. Chem Eng J 350:344–355. https://doi.org/10.1016/J.CEJ.2018.05.023
Mohan D, Pittman CU (2006) Activated carbons and low cost adsorbents for remediation of tri- and hexavalent chromium from water. J Hazard Mater 137:762–811. https://doi.org/10.1016/j.jhazmat.2006.06.060
Mohankumar K, Hariharan V, Rao NP (2016) Heavy metal contamination in groundwater around industrial estate vs residential areas in Coimbatore, India. J Clin Diagn Res 10:BC05–BC07. https://doi.org/10.7860/JCDR/2016/15943.7527
Mondal S, Derebe AT, Wang K (2018) Surface functionalized carbon microspheres for the recovery of copper ion from refinery wastewater. Korean J Chem Eng 35:147–152. https://doi.org/10.1007/s11814-017-0245-4
Moyo M, Chikazaza L, Nyamunda BC, Guyo U (2013) Adsorption batch studies on the removal of Pb ( II ) using maize tassel based activated carbon. 17:3–17:8. doi: https://doi.org/10.1155/2013/508934
Núñez-Gómez D, Rodrigues C, Lapolli FR, Lobo-Recio MÁ (2019) Adsorption of heavy metals from coal acid mine drainage by shrimp shell waste: isotherm and continuous-flow studies. J Environ Chem Eng 7:102787. https://doi.org/10.1016/J.JECE.2018.11.032
Oehmen A, Vergel D, Fradinho J et al (2014) Mercury removal from water streams through the ion exchange membrane bioreactor concept. J Hazard Mater 264:65–70. https://doi.org/10.1016/j.jhazmat.2013.10.067
Pehlivan E, Cetin S (2009) Sorption of Cr (VI) ions on two Lewatit-anion exchange resins and their quantitative determination using UV–visible spectrophotometer. J Hazard Mater 163:448–453
Sabela MI, Kunene K, Kanchi S et al (2016) Removal of copper (II) from wastewater using green vegetable waste derived activated carbon: an approach to equilibrium and kinetic study. Arab J Chem. https://doi.org/10.1016/j.arabjc.2016.06.001
Sahinkaya E, Altun M, Bektas S, Komnitsas K (2012a) Bioreduction of Cr (VI) from acidic wastewaters in a sulfidogenic ABR. Miner Eng 32:38–44
Sahinkaya E, Kilic A, Altun M et al (2012b) Hexavalent chromium reduction in a sulfur reducing packed-bed bioreactor. J Hazard Mater 219:253–259
Saini AS, Melo JS (2013) Biosorption of uranium by melanin: kinetic, equilibrium and thermodynamic studies. Bioresour Technol 149:155–162
Sheng PX, Ting YP, Chen JP, Hong L (2004) Sorption of lead, copper, cadmium, zinc, and nickel by marine algal biomass: characterization of biosorptive capacity and investigation of mechanisms. J Colloid Interface Sci 275:131–141. https://doi.org/10.1016/j.jcis.2004.01.036
Sono K, Lye D, Moore CA et al (2012) Melanin-based coatings as lead-binding agents. Bioinorg Chem Appl 2012. https://doi.org/10.1155/2012/361803
Sun H, Wu T, Zhang Y et al (2018) Structure-enhanced removal of Cr (vi) in aqueous solutions using MoS 2 ultrathin nanosheets. New J Chem 42:9006–9015
Thaira H, Raval K, Manirethan V, Balakrishnan RM (2019) Melanin nano-pigments for heavy metal remediation from water. Sep Sci Technol 54:265–274. https://doi.org/10.1080/01496395.2018.1443132
Wilbur S, Abadin H, Fay M, et al (2012) Health effects
Yang J, Yu M, Chen W (2015) Adsorption of hexavalent chromium from aqueous solution by activated carbon prepared from longan seed: kinetics, equilibrium and thermodynamics. J Ind Eng Chem 21:414–422
Yang J, Yu M, Qiu T (2014) Adsorption thermodynamics and kinetics of Cr (VI) on KIP210 resin. J Ind Eng Chem 20:480–486
Yu J-G, Yue B-Y, Wu X-W et al (2016) Removal of mercury by adsorption: a review. Environ Sci Pollut Res 23:5056–5076. https://doi.org/10.1007/s11356-015-5880-x
Zhang G, Liu Y, Zheng S, Hashisho Z (2019) Adsorption of volatile organic compounds onto natural porous minerals. J Hazard Mater 364:317–324. https://doi.org/10.1016/j.jhazmat.2018.10.031
Zhu L-P, Yu J-Z, Xu Y-Y et al (2009) Surface modification of PVDF porous membranes via poly(DOPA) coating and heparin immobilization. Colloids Surf B: Biointerfaces 69:152–155. https://doi.org/10.1016/j.colsurfb.2008.11.011
Acknowledgements
The authors acknowledge the ICP-OES facility under FIST grant sanctioned by the Department of Science and Technology, Government of India. The authors also acknowledge the facility rendered by the Department of Metallurgical and Materials Engineering and Department of Chemistry, NITK Surathkal, for the scanning electron microscopic images and FTIR analysis.
Funding
This work was financially supported by the Department of Science and Technology, Government of India [DST/TSG/WP/2014/58]
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Angeles Blanco
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Manirethan, V., Gupta, N., Balakrishnan, R.M. et al. Batch and continuous studies on the removal of heavy metals from aqueous solution using biosynthesised melanin-coated PVDF membranes. Environ Sci Pollut Res 27, 24723–24737 (2020). https://doi.org/10.1007/s11356-019-06310-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-019-06310-8