Abstract
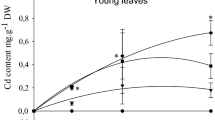
Halophytes have several advantages to be more effective in metal phytoextraction. Little is known about the Cd-phytoextraction potential of Atriplex lentiformis under different levels of Cd. Seven levels of Cd (0, 40, 80, 120, 160, 200, and 240 mg per kg of soil) were added to A. lentiformis plants grown on pots filled with 5 kg of sandy loam soil. A. lentiformis plants achieve different defense mechanisms to meet the high concentration of Cd in the soil and plant. These mechanisms include reducing the number and area of leaves, minimizing chlorophyll synthesis, and enhancing synthesizing of oxalic acid, phenols, and proline. The critical point of Cd was 9.35 and 183 mg kg−1 for available soil Cd and leaves concentrations, respectively. The maximum level of Cd displayed a 66% decrease in the chlorophyll content of the leaves. On the other hand, the oxalic acid, phenols, and proline in the leaves were increased significantly by 129, 100, and 200% when Cd increased from 0 to 240 mg. The tested plant removed 3.6% of the total soil Cd under the low Cd concentration (40 mg) but under the high level of Cd (240 mg), it only removed a negligible amount of soil Cd (0.74%). The current study confirmed that A. lentiformis plants lost the ability to cleanup Cd from contaminated soil under the high levels of contamination due to the high reduction in the production of dry matter.






Similar content being viewed by others
References
Ali B, Huang CR, Qi ZY, Ali S, Daud MK, Geng XX, Liu HB, Zhou WJ (2013a) 5-Aminolevulinic acid ameliorates cadmium-induced morphological, biochemical and ultra structural changes in seedlings of oilseed rape. Environ Sci Pollut Res 20:7256–7267. https://doi.org/10.1007/s11356-013-1735-5
Ali B, Qian P, ** R, Ali S, Khan M, Aziz R (2013b) Physiological and ultra-structural changes in Brassica napus seedlings induced by cadmium stress. Biol Plant 58:131–138. https://doi.org/10.1007/s10535-013-0358-5
Arnon DI (1949) Copper enzymes in isolated chloroplasts. Polyphenol oxidase in Beta vulgaris. Plant Physiol 24:1–15. https://doi.org/10.1104/pp.24.1
Bates LS, Walden RP, Teare ID (1973) Rapid determination of free proline for water stress studies. Plant Soil 39(1):205–207. https://doi.org/10.1007/BF00018060
Burt R (2004) Soil survey laboratory methods manual. Soil Survey Investigations Report No. 42, Version 4.0, Natural Resources Conservation Service, United States Department of Agriculture
Butcher DJ (2009) Phytoremediation of lead in soil: recent applications and future prospects. ApplSpectrosc Rev 44:123–139. https://doi.org/10.1080/05704920802352580
Chaney RL, Malik M, Li YM, Brown SL, Brewer EP, Angle JS, Baker AJ (1997) Phytoremediation of soil metals. CurrOpinBiotechnol 8:279–228. https://doi.org/10.1016/S0958-1669(97)80004-3
Chaturvedi I (2004) Phytotoxicity of cadmium and its effect on two genotypes of Brassica juncea L. Emir J Agric Sci 16(2):01–08
Daud MK, Variatha MT, Ali S, Najeeb U, Jamil M, Hayat Y, Dawood M, Khand MI, Zaffar M, Cheema SA, Tonga XH, Zhu SJ (2009) Cadmium-induced ultramorphological and physiological changes in leaves of two transgenic cotton cultivars and their wild relative. J Hazard Mater 168:614–625. https://doi.org/10.1016/j.jhazmat.2009.02.069
Diaz J, Bernal A, Pomar F, Merino F (2001) Induction of shikimate dehydrogenase and peroxidase in pepper (Capsicum annum L .) seedlings in response to copper stress and its relation to lignification. Plant Sci 161:179–188. https://doi.org/10.1016/S0168-9452(01)00410-1
Eissa MA (2014) Performance of river saltbush (Atriplexamnicola) grown on contaminated soils as affected by organic fertilization. World App Sci J 30:1877–1881. https://doi.org/10.5829/idosi.wasj.2014.30.12.19
Eissa MA, Ghoneim MF, Elgharably GA, AbdElRazek M (2014) Phytoextraction of nickel, lead and cadmium from metals contaminated soils using different field crops and EDTA. World Appl Sci J 32:1045–1052. https://doi.org/10.5829/idosi.wasj.2014.32.06.912
Eissa MA (2016) Effect of sugarcane vinasse and EDTA on cadmium phytoextraction by two saltbush plants. Environ Sci Pollut Res 23(10):10247–10254. https://doi.org/10.1007/s11356-016-6261-9
Eissa MA, Ahmed EM, Reichman SM (2016) Production of the forage halophyte Atriplex amnicola in metal-contaminated soils. Soil Use Manage 32(3):350–356. https://doi.org/10.1111/sum.12275
Eissa MA (2017) Phytoextraction mechanism of Cd by Atriplexlentiformis using some mobilizing agents. Ecol Eng 108:220–226. https://doi.org/10.1016/j.ecoleng.2017.08.025
Eissa MA, Ahmed EM (2016) Nitrogen and phosphorus fertilization for some Atriplexplants grown on metal-contaminated soils. Soil Sediment Contam 25(4):431–442. https://doi.org/10.1080/15320383.2016.1158693
Emamverdian A, Ding Y, Mokhberdoran F, **e Y (2015) Heavy metal stress and some mechanisms of plant defense response. Sci World J 2:1–18. https://doi.org/10.1155/2015/756120
Folin O, Denis W (1915) A calorimetric method for the determination of phenols and phenol derivatives in urine. J Biol Chem 22:305–308
Guo B, Liu C, Ding N, Fu Q, Lin Y, Li h LN (2016) Silicon alleviates cadmium toxicity in two cypress varieties by strengthening the exodermis tissues and stimulating phenolic exudation of roots. J Plant Growth Regul 35:420–429
Hasanuzzaman M, Nahar K, Alam MM, Bhowmik PC, Hossain MA, Rahman MM, Prasad MNV, Ozturk M, Fujita M (2014) Potential use of halophytes to remediate saline soils. BioMed Res Int 1–12. https://doi.org/10.1155/2014/589341
Lindsay WL, Norvell WA (1969) Equilibrium relationship of Zn+2, Fe+3, Ca+2 and H+ with EDTA and DTPA in soils. Soil Sci Soc Am Proc 33:62–68
Lutts S, Lefe I, Delpe C, Kivits S (2004) Heavy metal accumulation by halophyte species. J Environ Qual33 33:1271–1279. https://doi.org/10.2134/jeq2004.1271
Manousaki E, Kalogerakis N (2009) Phytoextraction of Pb and Cd by the Mediterranean saltbush (Atriplexhalimus L.): metal uptake in relation to salinity. Environ Sci Pollut Res 16:844–854. https://doi.org/10.1007/s11356-009-0224-3
Naik VV, Patil NS, Aparadh VT, Karadge BA (2014) Methodology in determination of oxalic acid in plant tissue: a comparative approach. Journal of Global Trends in Pharmaceutical Sciences 5(2)-(2014) 1662–1672
Najeeb U, Jilani G, Ali S, Sarwar M, Xu L, Zhou WJ (2011) Insight into cadmium induced physiological and ultra-structural disorders in Juncuseffusus L. and its remediation through exogenous citric acid. J Hazard Mater 186:565–574. https://doi.org/10.1016/j.jhazmat.2010.11.037
Nedjimia B, Daoud Y (2009) Cadmium accumulation in Atriplexhalimus subsp. schweinfurthii and its influence on growth, proline, root hydraulic conductivity and nutrient uptake Flora, 204, 316–324.https://doi.org/10.1016/j.flora.2008.03.004
Pál M, Horváth E, Janda T, Páldi E, Szalai G (2006) Physiological changes and defense mechanisms induced by cadmium stress in maize. J Plant Nutr Soil Sci 169:239–246. https://doi.org/10.1002/jpln.200520573
Palmer CE, Warwick S, Keller W (2001) Brassicaceae (Cruciferae) family, plant biotechnology, and phytoremediation. Int J Phytorem 3:245–287. https://doi.org/10.1080/15226510108500059
Poniedziałek M, Sekara A, Jedrszczyk E, Ciura J (2010) Phytoremediation efficiency of crop plants in removing cadmium, lead and zinc from soil. Folia Hortic Ann 22:25–31. https://doi.org/10.2478/fhort-2013-0155
Rizwan M, Ali S, Rizvi H, Rinklebe J, Tsang D, Meers E, Sik Y, Ishaque W (2016) Phytomanagement of heavy metals in contaminated soils using sunflower: a review. Crit Rev Environ Sci Technol 46(18):1498–1528. https://doi.org/10.1080/10643389.2016.1248199
Ruley AT, Sharma NC, Sahi SV, Singh SR, Sajwan KS (2006) Effects of lead and chelators on growth, photosynthetic activity and Pb uptake in Sesbaniadrummondii grown in soil. Environ Pollut 144:11–18. https://doi.org/10.1016/j.envpol.2006.01.016
Sai Kachout S, Leclerc JC, Ben Mansoura A, Rejeb MN, Ouerghi Z (2009) Effects of heavy metals on growth and bioaccumulation of the annual halophytes Atriplexhortensis and A. rosea. J. Appl Sci Res 5:746–756
Salt DE, Smith RD, Raskin I (1998) Phytoremediation. Annu Rev Plant Physiol Plant Mol Biol 49:643–668. https://doi.org/10.1146/annurev.arplant.49.1.643
Sharma S, Dietz KJ (2006) The significance of amino acids and amino acid-derived molecules in plant responses and adaptation to heavy metal stress. J Exp Bot 57:711–726
Soliz D, Glen EP, Seaman R, Nelson SM, Brown P (2011) Water consumption, irrigation efficiency and nutritional value of Atriplexlentiformis grown on reverse osmosis brine in a desert irrigation district. Agric Ecosyst Environ 140:473–483. https://doi.org/10.1016/j.agee.2011.01.012
Song X, Hu X, Ji P, Li Y, Chi G, Song Y (2012) Phytoremediation of cadmium contaminated farmland soil by the hyperaccumulator Beta vulgaris L. var. cicla. Bull Environ Contamin Toxicol 88:623–626. https://doi.org/10.1007/s00128-012-0524-z
Sytar O, Kumar A, Latowski D, Kuczynska P, Strzałka K, Prasad MNV (2013) Heavy metal-induced oxidative damage, defense reactions, and detoxification mechanisms in plants. Acta Physiol Plant 35:985–999
Vrettors JS, Stone DA, Brudvig GW (2001) Quantifying the ion selectivity of the Ca2? Site in photosystem II: evidence for direct involvement of Ca2? In O2 formation. Biochemistry 40:7937–7945. https://doi.org/10.1021/bi010679z
Wagner GJ (1993) Accumulation of cadmium in crop plants and its consequences to human health. Adv Agron 51:173–212. https://doi.org/10.1016/S0065-2113(08)60593-3
Wojcik M, Skorzynska-Polit E, Tukiendorf A (2006) Organic acids accumulation and antioxidant enzyme activities in Thlaspi caerulescens under Zn and Cd stress. Plant Growth Regul 48:145–155
Zouari M, Ben Ahmed C, Elloumi N, Bellassoued K, Delmail D, Labrousse P, Ben Abdallah F, Ben Rouina B (2016) Impact of proline application on cadmium accumulation, mineral nutrition and enzymatic antioxidant defense system of Oleaeuropaea L. cv Chemlali exposed to cadmium stress. Ecotoxicol Environ Saf 128:195–205. https://doi.org/10.1016/j.ecoenv.2016.02.024
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Elena Maestri
Rights and permissions
About this article
Cite this article
Eissa, M.A., Abeed, A.H. Growth and biochemical changes in quail bush (Atriplex lentiformis (Torr.) S.Wats) under Cd stress. Environ Sci Pollut Res 26, 628–635 (2019). https://doi.org/10.1007/s11356-018-3627-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-018-3627-1