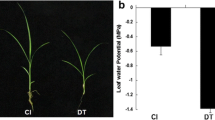
Abstract
Drought is one of the most devastating effects that severely reduce plant growth and development worldwide. In recent years, the availability of a reference Paulownia genome sequence has made it easier to explore gene expression, transcriptional regulation, and posttranscriptional regulation in Paulownia species. Here, we combined the analyses of the transcriptome, small RNAs, and degradome of Paulownia tomentosa seedlings to generate a comprehensive resource to describe the links between key regulatory miRNA-target gene pairs and drought stress. A total of 22,904 differentially expressed genes, 2073 differentially expressed miRNAs, and 198 target genes were identified by deep sequencing. Gene ontology function and KEGG pathway analyses of the differentially expressed genes and the target genes of the differentially expressed miRNAs revealed that momilactone A synthase, 14-3-3 protein, serine/threonine-protein kinase CTR1, and polyphenol oxidase, as well as alternative splicing, were associated directly or indirectly with drought stress in P. tomentosa. Our results will help to pave the way for further genomic studies, not only on P. tomentosa but also on other plants in family Paulowniaceae.





Similar content being viewed by others
References
Addoquaye C, Miller W, Axtell MJ (2009) CleaveLand: a pipeline for using degradome data to find cleaved small RNA targets. Bioinformatics 25(1):130–131. https://doi.org/10.1093/bioinformatics/btn604
Addo-Quaye C, Eshoo TW, Bartel DP, Axtell MJ (2008) Endogenous siRNA and miRNA targets identified by sequencing of the Arabidopsis degradome. Curr Biol 18(10):758–762. https://doi.org/10.1016/j.cub.2008.04.042
Ahmed CB, Rouina BB, Sensoy S, Boukhris M, Abdallah FB (2009) Changes in gas exchange, proline accumulation and antioxidative enzyme activities in three olive cultivars under contrasting water availability regimes. Environ Exp Bot 67(2):345–352. https://doi.org/10.1016/j.envexpbot.2009.07.006
Almssallem IS, Hu S, Zhang X, Lin Q, Liu W, Tan J, Yu X, Liu J, Pan L, Zhang T (2013) Genome sequence of the date palm Phoenix dactylifera L. Nat Commun 4:2274. https://doi.org/10.1038/ncomms3274
An FM, Chan MT (2012) Transcriptome-wide characterization of miRNA-directed and non-miRNA-directed endonucleolytic cleavage using Degradome analysis under low ambient temperature in Phalaenopsis aphrodite subsp. formosana. Plant Cell Physiol 53(10):1737–1750. https://doi.org/10.1093/pcp/pcs118
Anjum S, Wang L, Farooq M, Hussain M, Xue L, Zou C (2011) Brassinolide application improves the drought tolerance in maize through modulation of enzymatic antioxidants and leaf gas exchange. J Agron Crop Sci 197(3):177–185. https://doi.org/10.1111/j.1439-037X.2010.00459.x
Apel K, Hirt H (2004) Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol 55(1):373–399. https://doi.org/10.1146/annurev.arplant.55.031903.141701
Atawong A, Hasegawa M, Kodama O (2002) Biosynthesis of rice phytoalexin: enzymatic conversion of 3beta-hydroxy-9beta-pimara-7,15-dien-19,6beta-olide to momilactone A. Biosci Biotechnol Biochem 66(3):566–570. https://doi.org/10.1271/bbb.66.566
Audic S, Claverie JM (1997) The significance of digital gene expression profiles. Genome Res 7(10):986–995. https://doi.org/10.1101/gr.7.10.986
Aukerman MJ, Sakai H (2003) Regulation of flowering time and floral organ identity by a microRNA and its APETALA2-like target genes. Plant Cell 15(11):2730–2741. https://doi.org/10.1105/tpc.016238
Barbazuk WB, Fu Y, McGinnis KM (2008) Genome-wide analyses of alternative splicing in plants: opportunities and challenges. Genome Res 18(9):1381–1392. https://doi.org/10.1101/gr.053678.106
Biehler K, Fock H (1996) Evidence for the contribution of the Mehler-peroxidase reaction in dissipating excess electrons in drought-stressed wheat. Plant Physiol 112(1):265–272. https://doi.org/10.1104/pp.112.1.265
Boyer JS (1982) Plant productivity and environment. Science 218(4571):443–448. https://doi.org/10.1126/science.218.4571.443
Broberg P (2005) A comparative review of estimates of the proportion unchanged genes and the false discovery rate. BMC Bioinformatics 6(1):199. https://doi.org/10.1186/1471-2105-6-199
Brown JWS, Marshall DF, Echeverria M (2008) Intronic noncoding RNAs and splicing. Trends Plant Sci 13(7):335–342. https://doi.org/10.1016/j.tplants.2008.04.010
Cao X, Fan G, Cao L, Deng M, Zhao Z, Niu S, Wang Z, Wang Y (2017) Drought stress-induced changes of microRNAs in diploid and autotetraploid Paulownia tomentosa. Genes Genome 39(1):77–86. https://doi.org/10.1007/s13258-016-0473-8
Chao J, Zhang S, Chen Y, Tian WM (2016) Isolation and characterization of a plastidic glutamine synthetase (GS) gene in laticifers of Hevea brasiliensis Muell. Argric Acta Physiol Plant 38(4):1–11. https://doi.org/10.1007/s11738-016-2107-9
Chen C, Ridzon DA, Broomer AJ, Zhou Z, Lee DH, Nguyen JT, Barbisin M, NL X, Mahuvakar VR, Andersen MR (2005) Real-time quantification of microRNAs by stem-loop RT-PCR. Nucleic Acids Res 33(20):e179. https://doi.org/10.1093/nar/gni178
Clark KL, Larsen PB, Wang X, Chang C (1998) Association of the Arabidopsis CTR1 Raf-like kinase with the ETR1 and ERS ethylene receptors. P Natl Acad Sci USA 95(9):5401–5406. https://doi.org/10.1073/pnas.95.9.5401
Dong Y, Fan G, Deng M, Xu E, Zhao Z (2014) Genome-wide expression profiling of the transcriptomes of four Paulownia tomentosa accessions in response to drought. Genomics 104(4):295–305. https://doi.org/10.1016/j.ygeno.2014.08.008
Doumett S, Doumett S, Lamperi L, Checchini L, Azzarello E, Mugnai S, Mancuso S, Petruzzelli G, Del Bubba M (2008) Heavy metal distribution between contaminated soil and Paulownia tomentosa, in a pilot-scale assisted phytoremediation study: influence of different complexing agents. Chemosphere 72(10):1481–1490. https://doi.org/10.1016/j.chemosphere.2008.04.083
Duan B, Yang Y, Lu Y, Korpelainen H, Berninger F, Li C (2007) Interactions between water deficit, ABA, and provenances in Picea asperata. J Exp Bot 58(11):3025–3036. https://doi.org/10.1093/jxb/erm160
Eldem V, ÇA U, Ozhuner E, Bakır Y, Uranbey S, Unver T (2012) Genome-wide identification of miRNAs responsive to drought in peach (Prunus persica) by high-throughput deep sequencing. PLoS One 7(12):e50298. https://doi.org/10.1371/journal.pone.0050298
Fan G, Yang Z, Cao Y (2007) Induction of autotetraploid of Paulownia tomentosa (Thunb.) Steud. Plant Physiol Commun 43:109
Fan G, Wang L, Deng M, Niu S, Zhao Z, Xu E, Cao X, Zhang X (2015) Transcriptome analysis of the variations between autotetraploid Paulownia tomentosa and its diploid using high-throughput sequencing. Mol Genet Genomics 290(4):1627–1638. https://doi.org/10.1007/s00438-015-1023-9
Fan G, Niu S, Zhao Z, Deng M, Xu E, Wang Y, Yang L (2016a) Identification of microRNAs and their targets in Paulownia fortunei plants free from phytoplasma pathogen after methyl methane sulfonate treatment. Biochimie 127:271–280. https://doi.org/10.1016/j.biochi.2016.06.010
Fan G, Wang L, Deng M, Zhao Z, Dong Y, Zhang X, Li Y (2016b) Changes in transcript related to osmosis and intracellular ion homeostasis in Paulownia tomentosa under salt stress. Front Plant Sci 7. https://doi.org/10.3389/fpls.2016.00384
Fan G, Wang L, Dong Y, Zhao Z, Deng M, Niu S, Zhang X, Cao X (2017) Genome of Paulownia (Paulownia fortunei) illuminates the related transcripts, miRNA and proteins for salt resistance. Sci Rep 7(1):1285. https://doi.org/10.1038/s41598-017-01360-9
Farooq M, Wahid A, Kobayashi N, Fujita D, Basra SMA (2009) Plant drought stress: effects, mechanisms. Agron Sustain Dev 29(1):185–212. https://doi.org/10.1051/agro:2008021
Filichkin SA, Priest HD, Givan SA, Shen R, Bryant DW, Fox SE, Wong WK, Mockler TC (2010) Genome-wide map** of alternative splicing in Arabidopsis thaliana. Genome Res 20(1):45–58. https://doi.org/10.1101/gr.093302.109
Fridovich I (1986) Biological effects of the superoxide radical. Arch Biochem Biophys 247(1):1–11. https://doi.org/10.1016/0003-9861(86)90526-6
Fu J, Huang B (2001) Involvement of antioxidants and lipid peroxidation in the adaptation of two cool-season grasses to localized drought stress. Environ Exp Bot 45(2):105–114. https://doi.org/10.1016/S0098-8472(00)00084-8
German MA, German MA, Pillay M, Jeong DH, Hetawal A, Luo S, Janardhanan P, Kannan V, Rymarquis LA, Nobuta K, German R (2008) Global identification of microRNA-target RNA pairs by parallel analysis of RNA ends. Nat Biotechnol 26(8):941–946. https://doi.org/10.1038/nbt1417
Hall T (2008) Paulownia: an agroforestry gem. Trees Life J 3
Hasegawa M, Mitsuhara I, Seo S, Imai T, Koga J, Okada K, Yamane H, Ohashi Y (2010) Phytoalexin accumulation in the interaction between rice and the blast fungus. Mol Plant Microbe Interact 23(34):1000–1011. https://doi.org/10.1523/JNEUROSCI.1765-06.2006
Himmelbach A, Yang Y, Grill E (2003) Relay and control of abscisic acid signaling. Curr Opin Plant Biol 6(5):470–479. https://doi.org/10.1016/S1369-5266(03)00090-6
Hirsch J, Lefort V, Vankersschaver M, Boualem A, Lucas A, Thermes C, D'Aubentoncarafa Y, Crespi M (2006) Characterization of 43 non-protein-coding mRNA genes in Arabidopsis, including the MIR162a-derived transcripts. Plant Physiol 140(4):1192–1204. https://doi.org/10.1104/pp.105.073817
Ill Min Chung JTK, Kim S (2006) Evaluation of allelopathic potential and quantification of momilactone A,B from rice hull extracts and assessment of inhibitory bioactivity on paddy field weeds. J Agric Food Chem 54(7):2527–2536. https://doi.org/10.1021/jf052796x
Ipekci Z, Gozukirmizi N (2003) Direct somatic embryogenesis and synthetic seed production from Paulownia elongata. Plant Cell Rep 22(1):16–24. https://doi.org/10.1007/s00299-003-0650-5
Kato T, Kabuto C, Sasaki N, Tsunagawa M, Aizawa H, Fujita K, Kato Y, Kitahara Y, Takahashi N (1973) Momilactones, growth inhibitors from rice, Oryza sativa L. Tetrahedron Lett 14(39):3861–3864. https://doi.org/10.1016/S0040-4039(01)87058-1
Katonoguchi H, Ino T, Ota K (2008) Secretion of momilactone A from rice roots to the rhizosphere. J Plant Physiol 165(7):691–696. https://doi.org/10.1016/j.jplph.2007.07.018
Kurihara Y, Watanabe Y (2004) Arabidopsis micro-RNA biogenesis through Dicer-like 1 protein functions. Proc Natl Acad Sci USA 101(34):12753–12758. https://doi.org/10.1073/pnas.0403115101
Li R, Li Y, Kristiansen K, Wang J (2008) SOAP: short oligonucleotide alignment program. Bioinformatics 24(5):713–714. https://doi.org/10.1093/bioinformatics/btn025
Lu S, Sun YH, Shi R, Clark C, Li L, Chiang VL (2005) Novel and mechanical stress-responsive MicroRNAs in Populus trichocarpa that are absent from Arabidopsis. Plant Cell 17(8):2186–2203. https://doi.org/10.1105/tpc.105.033456
Maggio A, Mccully MG, Kerdnaimongkol K, Bressan RA, Hasegawa PM, Joly RJ (2002) The ascorbic acid cycle mediates signal transduction leading to stress-induced stomatal closure. Funct Plant Biol 29(7):845–852. https://doi.org/10.1071/PP01259
Mahajan S, Tuteja N (2005) Cold, salinity and drought stresses: an overview. Arch Biochem Bioph 444(2):139–158. https://doi.org/10.1016/j.abb.2005.10.018
Marquez Y, Brown JW, Simpson C, Barta A, Kalyna M (2012) Transcriptome survey reveals increased complexity of the alternative splicing landscape in Arabidopsis. Genome Res 22(6):1184–1195. https://doi.org/10.1101/gr.134106.111
Marrs KA (1996) The function and regulation of glutathione-S-transferase in plants. Ann Rev Plant Physiol 47(1):127–158. https://doi.org/10.1146/annurev.arplant.47.1.127
Masterson J (1994) Stomatal size in fossil plants: evidence for polyploidy in majority of angiosperms. Science 264(5157):421–424. https://doi.org/10.1126/science.264.5157.421
Mayer AM (2006) Polyphenol oxidases in plants and fungi: going places? A review. Phytochemistry 67(21):2318–2331. https://doi.org/10.1016/j.phytochem.2006.08.006
Meyers BC, Meyers BC, Axtell MJ, Bartel B, Bartel DP, Baulcombe D, Bowman JL, Cao X, Carrington JC, Chen X, Green PJ (2008) Criteria for annotation of plant MicroRNAs. Plant Cell 20(12):3186–2331. https://doi.org/10.1105/tpc.108.064311
Mi S, Mi S, Cai T, Hu Y, Chen Y, Hodges E, Ni F, Wu L, Li S, Zhou H, Long C (2008) Sorting of small RNAs into Arabidopsis argonaute complexes is directed by the 5′ terminal nucleotide. Cell 133(1):116–127. https://doi.org/10.1016/j.cell.2008.02.034
Mohamadi N, Rajaei P (2013) Effect of triamidefon fungicide on some growth parameters and antioxidant enzymes activity in tomato (Lycopersicon esculentum Mill.) plant under drought stress. Int J Adv Biol Biomed Res 1:341–350
Mortazavi A, Williams BA, McCue K, Schaeffer L, Wold B (2008) Map** and quantifying mammalian transcriptomes by RNA-Seq. Nat Methods 5(7):621–628. https://doi.org/10.1038/nmeth.1226
Muthukumarasamy M, Gupta SD, Panneerselvam R (2000) Enhancement of peroxidase, polyphenol oxidase and superoxide dismutase activities by triadimefon in NaCl stressed Raphanus sativus L. Biol Plantarum 43(2):317–320. https://doi.org/10.1023/A:1002741302485
Mutz KO, Heilkenbrinker A, Lönne M, Walter JG, Stahl F (2013) Transcriptome analysis using next-generation sequencing. Curr Opin Biotechnol 24(1):22–30. https://doi.org/10.1016/j.copbio.2012.09.004
Niu S, Fan G, Xu E, Deng M, Zhao Z, Dong Y (2014) Transcriptome/degradome-wide discovery of MicroRNAs and transcript targets in two Paulownia australis genotypes. PLoS One 9(9):e106736. https://doi.org/10.1371/journal.pone.0106736
Prochazkova D, Sairam R, Srivastava G, Singh D (2001) Oxidative stress and antioxidant activity as the basis of senescence in maize leaves. Plant Sci 161(4):765–771. https://doi.org/10.1016/S0168-9452(01)00462-9
Reddy ASN (2007) Alternative splicing of pre-messenger RNAs in plants in the genomic era. Annu Rev Plant Biol 58(1):267–294. https://doi.org/10.1146/annurev.arplant.58.032806.103754
Reinhart BJ, Weinstein EG, Rhoades MW, Bartel B, Bartel DP (2002) MicroRNAs in plants. Genes Dev 16(13):1616–1626. https://doi.org/10.1101/gad.1004402
Ruby JG, Jan CH, Bartel DP (2007) Intronic microRNA precursors that bypass Drosha processing. Nature 448(1):83–86. https://doi.org/10.1146/annurev.arplant.58.032806.103754
Schultz TF, Medina J, Hill A, Quatrano RS (1998) 14-3-3 proteins are part of an abscisic acid-VIVIPAROUS1 (VP1) response complex in the Em promoter and interact with VP1 and EmBP1. Curr Opin Plant Biol 1(5):837–847. https://doi.org/10.1105/tpc.10.5.837
Shomron N, Levy C (2009) MicroRNA-biogenesis and pre-mRNA splicing crosstalk. J Biomed Biotechnol 2009:594678. https://doi.org/10.1155/2009/594678
Song QX, Liu YF, XY H, Zhang WK, Ma B, Chen SY, Zhang JS (2011) Identification of miRNAs and their target genes in develo** soybean seeds by deep sequencing. BMC Plant Biol 11(1):5–58. https://doi.org/10.1101/gr.093302.109
Szittya G, Moxon S, Santos DM, **g R, Fevereiro MP, Moulton V, Dalmay T (2008) High-throughput sequencing of Medicago truncatula short RNAs identifies eight new miRNA families. BMC Genomics 9(1):593. https://doi.org/10.1186/1471-2164-9-593
Toyomasu T, Kagahara T, Okada K, Koga J, Hasegawa M, Mitsuhashi W, Sassa T, Yamane H (2008) Diterpene phytoalexins are biosynthesized in and exuded from the roots of rice seedlings. Biosci Biotechnol Biochem 72(2):562–567. https://doi.org/10.1271/bbb.70677
Tuskan GA, Tuskan GA, Difazio S, Jansson S, Bohlmann J, Grigoriev I, Hellsten U, Putnam N, Ralph S, Rombauts S, Salamov A (2006) The genome of black cottonwood, Populus trichocarpa (Torr. & Gray). Science 313(5793):1596–1604. https://doi.org/10.1126/science.1128691
Vanetten HD, Mansfield JW, Bailey JA, Farmer EE (1994) Two classes of plant antibiotics: phytoalexins versus "phytoanticipins". Plant Cell 6(9):1191–1192. https://doi.org/10.1105/tpc.6.9.1191
Voinnet O (2009) Origin, biogenesis, and activity of plant MicroRNAs. Cell 136(4):669–687. https://doi.org/10.1016/j.cell.2009.01.046
Wasilewska A, Vlad F, Sirichandra C, Redko Y, Jammes F, Valon C, Frei dit Frey N, Leung J (2008) An update on abscisic acid signaling in plants and more. Mol Plant 1(2):198–217. https://doi.org/10.1093/mp/ssm022
Wei M, Wei H, Wu M, Song M, Zhang J, Yu J, Fan S, Yu S (2013) Comparative expression profiling of miRNA during anther development in genetic male sterile and wild type cotton. BMC Plant Biol 13(1):66. https://doi.org/10.1186/1471-2229-13-66
Wood TE, Takebayashi N, Barker MS, Mayrose I, Greenspoon PB, Rieseberg LH (2009) The frequency of polyploid speciation in vascular plants. Proc Natl Acad Sci U S A 106(33):13875–13879. https://doi.org/10.1073/pnas.0811575106
Xu X, Yin L, Ying Q, Song H, Xue D, Lai T, Xu M, Shen B, Wang H, Shi X (2013) High-throughput sequencing and degradome analysis identify miRNAs and their targets involved in fruit senescence of Fragaria ananassa. PLoS One 8(8):e70959. https://doi.org/10.1371/journal.pone.0070959
Xuan TD, Minh TN, Anh LH, Khanh TD (2016) Allelopathic momilactones A and B are implied in rice drought and salinity tolerance, not weed resistance. Agron Sustain Dev 36(3):52. https://doi.org/10.1007/s13593-016-0383-9
Yan J, He C, Wang J, Mao Z, Holaday SA, Allen RD, Zhang H (2004) Overexpression of the Arabidopsis 14-3-3 protein GF14 lambda in cotton leads to a "stay-green" phenotype and improves stress tolerance under moderate drought conditions. Plant Cell Physiol 45(8):1007–1014. https://doi.org/10.1093/pcp/pch115
Yan K, Liu P, CA W, Yang GD, Xu R, Guo QH, Huang JG, Zheng CC (2012) Stress-induced alternative splicing provides a mechanism for the regulation of microRNA processing in Arabidopsis thaliana. Mol Cell 48(4):521–531. https://doi.org/10.1016/j.molcel.2012.08.032
Yang ZM, Chen J (2013) A potential role of microRNAs in plant response to metal toxicity. Metallomics Integr Biomet Sci 5(9):1184–1190. https://doi.org/10.1039/c3mt00022b
Yang X, Wang L, Yuan D, Lindsey K, Zhang X (2013) Small RNA and degradome sequencing reveal complex miRNA regulation during cotton somatic embryogenesis. J Exp Bot 64(6):1521–1536. https://doi.org/10.1093/jxb/ert013
Zeevaart JAD, Creelman RAD (2003) Metabolism and physiology of abscisic acid. Annu Rev Plant Biol 39:439–473
Zhang XS, Liu RN, Fan GQ, Zhao ZL, Deng MJ, University HA (2013a) Study on the physiological response of tetraploid Paulownia to drought. J Henan Agric Univ
Zhang H, Zhang J, Quan R, Pan X, Wan L, Huang R (2013b) EAR motif mutation of rice OsERF3 alters the regulation of ethylene biosynthesis and drought tolerance. Planta 237(6):1443–1451. https://doi.org/10.1007/s00425-013-1852-x
Zhang F, Zhu G, Du L, Shang X, Cheng C, Yang B, Hu Y, Cai C, Guo W (2016) Genetic regulation of salt stress tolerance revealed by RNA-Seq in cotton diploid wild species, Gossypium davidsonii. Sci Rep 6(1):20582. https://doi.org/10.1038/srep20582
Acknowledgements
This study was funded by the Natural Science Foundation of Henan Province of China (Grant No. 162300410158) and the Distinguished Talents Foundation of Henan Province of China (Grant No. 174200510001).
Data archiving statement
All the sequencing data generated in this study is available from the SRA-Archive (http://www.ncbi.nlm.nih.gov/sra) of NCBI. The transcriptome, small RNA, and degradome sequencing raw data from this article have been deposited under Accession Nos. SRP031515, SRP116066, and SRP116267.
Author information
Authors and Affiliations
Contributions
Guoqiang Fan conceived, designed, and carried out the study. Haifang Liu wrote the manuscript. Zhenli Zhao revised the manuscript. Limin Wang, Minjie Deng, **aoqiao Zhai, and Yanpeng Dong provided suggestions on the experimental design and analyses. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Open access
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Additional information
Communicated by A. Brunner
Haifang Liu and Zhenli Zhao are co-first authors.
Electronic supplementary material
Table S1
(XLSX 11 kb)
Table S2
(XLSX 3827 kb)
Table S3
(XLSX 1840 kb)
Table S4
(XLSX 969 kb)
Table S5
(XLSX 811 kb)
Table S6
(XLSX 27 kb)
Table S7
(XLSX 401 kb)
Table S8
(XLSX 387 kb)
Table S9
(XLSX 50 kb)
Fig. S1
Correlation coefficients of the expression of duplicate samples. X-axis represents the logarithmic value of diploid (A) or tetraploid (B) expression, while Y-axis represents the logarithmic value of the corresponding duplicate samples. (GIF 31 kb)
Fig. S2
Cluster analysis of the inter and union DEGs in the PT4 vs. PT2, PT4T vs. PT2T, PT4T vs. PT4, and PT2T vs. PT2 comparisons. (GIF 294 kb)
Fig. S3
Gene Ontology of the DEGs in Comparison A, B, C, and D. (GIF 67 kb)
Fig. S4
Gene Ontology of the DEGs in Comparison E, F, G, and H. (GIF 75 kb)
Fig. S5
KEGG annotation for the DEGs in Comparison A, B, C, and D. (GIF 142 kb)
Fig. S6
KEGG annotation for the DEGs in Comparison E, F, G, and H. (GIF 145 kb)
Fig. S7
Number of conserved miRNA families. (GIF 30 kb)
Fig. S8
Nucleotide bias at each position in miRNAs among the four sRNA libraries. A, B, C, and D represent the libraries made from the samples in PT2, PT2T, PT4, and PT4T, respectively. (GIF 140 kb)
Fig. S9
The cluster chart of miRNAs’ expression; (A): Conserved miRNAs; (B): Novel miRNAs. (GIF 219 kb)
Rights and permissions
About this article
Cite this article
Liu, H., Zhao, Z., Wang, L. et al. Genome-wide expression analysis of transcripts, microRNAs, and the degradome in Paulownia tomentosa under drought stress. Tree Genetics & Genomes 13, 128 (2017). https://doi.org/10.1007/s11295-017-1211-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11295-017-1211-3