Abstract
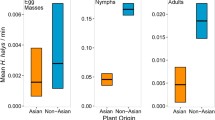
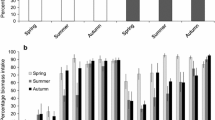
Thorns and hairs of plants can serve as defenses against herbivores, although they may not have evolved under selection by herbivory. Japanese nettles, Urtica thunbergiana, in Nara Park, Nara Prefecture, Japan, where sika deer have been protected for 1200 years, bear many more stinging hairs than those in areas with few or no deer. Previous studies suggested that such hairy nettles evolved under natural selection imposed by intense deer browsing, because stinging hairs deterred deer browsing and because among-population variation in hair density was associated with deer abundance. To confirm this hypothesis, we examined (1) whether stinging hairs affected oviposition and feeding preferences of herbivorous insects and (2) the degree to which they deterred deer via laboratory and field experiments with hairy nettles from Nara Park and with almost-hairless nettles from another area. A specialist butterfly, Indian red admiral, showed no oviposition or larval feeding preferences for either hairy or hairless nettles. Insect damage levels did not significantly differ between the two variants. In contrast, deer browsed hairless nettles more heavily than hairy ones. In hairy nettles, however, the level of deer browsing was not proportional to stinging-hair density, presumably because the hairy nettle population had reached a plateau for resistance as a result of long-term strong directional selection for stinging hairs. These results corroborate the hypothesis that hairy nettles in Nara Park evolved through natural selection under intense deer browsing.




Similar content being viewed by others
References
Barrett MA, Stiling P (2007) Relationships among Key deer, insect herbivores, and plant quality. Ecol Res 22:268–273
Cano-Santana Z, Oyama K (1992) Variation in leaf trichomes and nutrients of Wigandia urens (Hydrophyllaceae) and its implications for herbivory. Oecologia 92:405–409
Falconer DS, Mackay TFC (1996) An introduction to quantitative genetics, 4th edn. Longman, Essex
Grubb PJ (1992) A positive distrust in simplicity-lessons from plant defenses and from competition among plants and among animals. J Ecol 80:585–610
Hanley M, Lamont BB, Fairbanks MM, Rafferty CM (2007) Plant structural traits and their role in anti-herbivore defence. Perspect Plant Ecol Evol Syst 8:157–178
Kato T (2001) Nettles in Nara Park. Nara Bot 23:11–14 (in Japanese)
Kato T, Ishida K, Sato H (2007) The evolution of nettle resistance to heavy deer browsing. Ecol Res 23:339–345
Kitamura S, Murata G (1961) Colored illustrations of herbaceous plants of Japan, vol. II (Choripetalae) (in Japanese). Hoikusha, Osaka
Krebs C (1998) Ecological methodology, 2nd edn. Harper-Collins, New York
Milewski AV, Young TP, Madden D (1991) Thorns as induced defenses: experimental evidence. Oecologia 86:70–75
Molisch H, Dobat K (1979) Botanische Versuche und Beobachtungen mit einfachen Mitteln: ein Experimentierbuch für Schulen und Hochschulen, 5th edn. Gustav Fischer, Stuttgart
Mousseau TA, Roff DA (1987) Natural selection and the heritability of fitness components. Heredity 59:181–197
Ohgushi T, Schmitz O, Holt RD (eds) (2012) Trait-mediated indirect interactions: ecological and evolutionary perspectives. Cambridge University Press, Cambridge
Olsen C (1921) The ecology of Urtica dioica. J Ecol 9:1–8
Pollard AJ (1986) Variation in Cnidoscolus texanus in relation to herbivory. Oecologia 70:411–413
Pollard AJ, Briggs D (1982) Genecological studies of Urtica dioica L, I. The nature of intraspecific variation in U. dioica. New Phytol 92:453–470
Pollard AJ, Briggs D (1984a) Genecological studies of Urtica dioica L, II. Patterns of variation at Wicken Fen, Cambridgeshire, England. New Phytol 96:483–499
Pollard AJ, Briggs D (1984b) Genecological studies of Urtica dioica L, III. Stinging hairs and plant-herbivore interactions. New Phytol 97:505–522
Pullin AS, Gilbert JE (1989) The stinging nettle, Urtica dioica, increases trichome density after herbivore and mechanical damage. Oikos 54:275–280
Shikata M, Kato T, Shibata E, Sato H (2013) Among-population variation in resistance traits of a nettle and its relationship with deer habitat use frequency. Ecol Res 28:207–216
Shoonhoven LM, van Loon JJA, Dicke M (2005) Insect-plant biology, 2nd edn. Oxford University Press, Oxford
Sletvold N, Huttunen P, Handley R, Kärkkäinen K, Ågren J (2010) Cost of trichome production and resistance to a specialist insect herbivore in Arabidopsis lyrata. Evol Ecol 24:1307–1319
Sokal R, Rohlf JF (2012) Biometry, 4th edn. W H Freeman, New York
SPSS Inc (2006) SPSS base 15.0 syntax reference guide. SPSS Inc, Chicago
Teshirogi M (1990) An illustrated book of the Japanese Nymphalidae (in Japanese). Tokai University Press, Tokyo
Thurston EL (1974) Morphology, fine structure, and ontogeny of the stinging emergence of Urtica dioica. Am J Bot 61:809–817
Tuberville TD, Dudley PG, Pollard AJ (1996) Responses of invertebrate herbivores to stinging trichomes of Urtica dioica and Laportea canadensis. Oikos 75:83–88
Walters DR (2011) Plant defense: warding off attack by pathogens, herbivores, and parasitic plants. Wiley-Blackwell, Oxford
Yamamoto K (1997) LIA32 for Windows95 ver.0373. http://www.agr.nagoya-u.ac.jp/~shinkan/LIA32/index.html
Acknowledgments
We dedicate this paper to the memory of Emeritus Professor Ei’ichi Shibata, who always encouraged our studies. This study was supported financially in part by a Grant-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology of Japan ((C) No.22570019).
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Iwamoto, M., Horikawa, C., Shikata, M. et al. Stinging hairs on the Japanese nettle Urtica thunbergiana have a defensive function against mammalian but not insect herbivores. Ecol Res 29, 455–462 (2014). https://doi.org/10.1007/s11284-014-1137-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11284-014-1137-2