Abstract
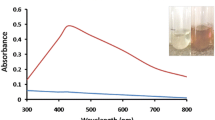
The biological synthesis methods have been emerging as a promising new approach for production of nanoparticles due to their simplicity and non-toxicity. In the present study, spores of Bacillus athrophaeus were used to achieve the objective of develo** a green synthesis method of silver nanoparticles. Enzyme assay revealed that the spores and their heat inactivated forms (microcapsules) were highly active and their enzymatic contents differed from the vegetative cells. Laccase, glucose oxidase, and alkaline phosphatase activities were detected in the dormant forms, but not in the vegetative cells. Although no nanoparticle was produced by active cells of B. athrophaeus, both spores and microcapsules were efficiently capable of reducing the silver ions (Ag+) to elemental silver (Ag0) leading to the formation of nanoparticles from silver nitrate (AgNO3). The presence of biologically synthesized silver nanoparticles was determined by obtaining broad spectra with maximum absorbance at 400 nm in UV–visible spectroscopy. The X-ray diffraction analysis pattern revealed that the nanoscale particles have crystalline nature with various topologies, as confirmed by transmission electron microscopy (TEM). The TEM micrograph showed the nanocrystal structures with dimensions ranging from 5 to 30 nm. Accordingly, the spore mixture could be employed as a factory for detoxification of heavy metals and subsequent production of nanoparticles. This research introduces an environmental friendly and cost effective biotechnological process for the extracellular synthesis of silver nanoparticles using the bacterial spores.





Similar content being viewed by others
References
Alani F, Moo-Young M, Anderson W (2012) Biosynthesis of silver nanoparticles by a new strain of Streptomyces sp. compared with Aspergillus fumigatus. World J Microbiol Biotechnol 28:1081–1086. doi:10.1007/s11274-011-0906-0
Anil Kumar S, Abyaneh MK, Gosavi Sulabha SW, Ahmad A, Khan MI (2007) Nitrate reductase-mediated synthesis of silver nanoparticles from AgNO3. Biotechnol Lett 29:439–445
Bai HJ, Yang BS, Chai CJ, Yang GE, Jia WL, Yi ZB (2011) Green synthesis of silver nanoparticles using Rhodobacter Sphaeroides. World J Microbiol Biotechnol 27:2723–2728. doi:10.1007/s11274-011-0747-x
Basnar B, Weizmann Y, Cheglakov Z, Willner I (2006) Synthesis of nanowires using dip-pen nanolithography and biocatalytic inks. Adv Mater 18:713–718
Breen A, Singleton FL (1999) Fungi in lignocellulose breakdown and biopul**. Curr Opin Biotechnol 10:252–258
Con TH, Loan DK (2011) Preparation of silver nano-particles and use as a material for water sterilization. EnvironmentAsia 4:62–66
De Oliveira DC, Pinto Tde J (2002) Study of sterilizing effectivity of different ethylene oxide gaseous mixtures using CFCs and HCFCs (Oxyfume 12R and 2002R). PDA J Pharm Sci Technol 56:242–247
Gleiter H (2000) Nanostructured materials: basic concepts and microstructure. Acta Mater 48:1–29
Greisen K, Loeffelholz M, Purohit A, Leong D (1994) PCR primers and probes for the 16S rRNA gene of most species of pathogenic bacteria, including bacteria found in cerebrospinal fluid. J Clin Microbiol 32:335–351
Harley S (1993) Use of a simple colorimetric assay to determine conditions for induction of nitrate reductase in plants. Am Biol Teach 55:161–164
Henriques AO, Beall BW, Roland K, Moran CP Jr (1995) Characterization of cotJ, a sigma E-controlled operon affecting the polypeptide composition of the coat of Bacillus subtilis spores. J Bacteriol 177:3394–3406
Hosseini Abari A, Emtiazi G, Roghanian R (2012) Production of none germinate spore ghost from a novel marine Bacillus with thermostable laccase activity. Afri J Microbiol Res 6:393–402
Hullo MF, Moszer I, Danchin A, Martin-Verstraete I (2001) CotA of Bacillus subtilis is a copper-dependent laccase. J Bacteriol 183:5426–5430
Jain D, Kachhaeaha S, Jain R, Sirvastava G, Kothari SL (2010) Novel microbial route to synthesize silver nanoparticles using spore crystal mixture of Bacillus thuringiensis. Indian J Exp Biol 48:1152–1156
Kalimuthu K, Babu RS, Venkataraman D, Mohd B, Gurunathan S (2008) Biosynthesis of silver nanocrystals by Bacillus licheniformis. Coll Surf B 65:150–153
Katz E, Willner I, Wang J (2004) Electroanalytical and bioelectroanalytical systems based on metal and semiconductor nanoparticles. Electroanalysis 16:19–44
Klaus T, Joerger R, Olsson E, Granqvist CG (1999) Silver-based crystalline nanoparticles, microbially fabricated. Proc Natl Acad Sci USA 96:13611–13614
Liu J, Qiao SZ, Hu QH, Lu GQ (2011) Magnetic nanocomposites with mesoporous structures: synthesis and applications. Small 7:425–443
Lu HW, Liu SH, Wang XF, Qian J, Yin J, Jhu JK (2003) Silver nanocrystals by hyperbranched polyurethane-assisted photochemical reduction of Ag+. Mater Chem Phys 81:104–107
Mandal D, Bolander ME, Mukhopadaya D, Sarkar G, Mukherjee P (2006) The use of microorganisms for the formation of metal nanoparticles and their application. Appl Microbiol Biotechnol 69:485–492
Miller GL (1959) Use of dinitrosaIicyIic acid reagent for determination of reducing sugar. Anal Chem 31:426–428
Mussing-Zufika M, Kornmuller A, Merkelbach B, Jekel M (1994) Isolation and analysis of intact polyphosphate chains from activated sludges associated with biological phosphate removal. Water Res 28:1725–1733
Nakamura LK (1989) Taxonomic relationship of black-pigmented Bacillus subtilis strains and a proposal for Bacillus atrophaeus sp. nov. Int J Syst Bacteriol 39:295–300
Nicholson WL, Setlow P (1990) Sporulation, germination, and out-growth. In: Harwood CR, Cutting SM (eds) Molecular biological methods for Bacillus. Wiley, Sussex, pp 391–450
Ruehle GLA (1923) The enzymatic content of bacterial spores. J Bacteriol 8:487–491
Ruttimann C, Schweber E, Salas L, Cullen D, Vicuna R (1992) Lygninolytic enzymes of white rot basidiomycetes Phlebia brevispora and Ceioporiopsis subvermispora. Biotechnol Appl Biochem 16:64–76
Silver S, Phung LT, Silver G (2006) Silver as biocides in burn and wound dressings and bacterial resistance to silver compounds. J Ind Microbiol Biotechnol 33:627–634
Suzuki S, Izawa Y, Kobayashi K, Eto Y, Yamanaka S, Kubota K, Yokozeki K (2000) Biosci Biotechnol Biochem 64:2344–2351
Whitesides GM (2003) The ‘right’ size in nanobiotechnology. Nat Biotechnol 21:1161–1165
Yu B, Xu P, Shi Q, Ma C (2006) Deep desulfurization of diesel oil and crude oils by a newly isolated Rhodococcus erythropolis strain. Appl Environ Microbiol 72:54–58
Zayats M, Baron R, Popov I, Willner I (2005) Biocatalytic growth of Au nanoparticles: from mechanistic aspects to biosensors design. Nano Lett 5:21–25
Acknowledgments
We thank the University of Isfahan for financial support given to PhD student for a training period in the Department of Biology and Microbiology.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Hosseini-Abari, A., Emtiazi, G. & Ghasemi, S.M. Development of an eco-friendly approach for biogenesis of silver nanoparticles using spores of Bacillus athrophaeus . World J Microbiol Biotechnol 29, 2359–2364 (2013). https://doi.org/10.1007/s11274-013-1403-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11274-013-1403-4