Abstract
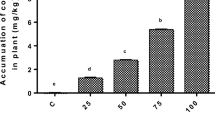
This study evaluated the toxic effects of arsenic (As) on the growth, total antioxidant activity, total content of phenolic compounds, and content of photosynthetic pigments of Azolla filiculoides. The aquatic fern was propagated and exposed to Yoshida nutrient solution contaminated with sodium arsenate (Na2HAsO4·7H2O) at six concentrations (5, 10, 20, 30, 60, and 120 μg As mL−1), including the control without As contamination. Azolla cultures were kept under environmental chamber conditions 26°C, 12 h photoperiod and 80% HR for 96 h. Increased As concentrations (>30 μg mL−1) significantly diminished growth of A. filiculoides and the total content of chlorophyll and total phenolic compounds, but significantly enhanced of total carotenoid + xanthophylls content. The concentrations of 5 and 10 μg As mL−1 significantly stimulated the growth of A. filiculoides. This aquatic fern tolerates As concentrations lower than 30 μg mL−1, and its maximum As accumulation (28 μg g−1 dry weight) was achieved when exposed to 60 μg As mL−1, but showing clear symptoms of As toxicity.




Similar content being viewed by others
References
Alarcón, A., Davies, F. T., Jr., Autenrieth, R. L., & Zuberer, D. A. (2008). Arbuscular mycorrhiza and petroleum-degrading microorganisms enhanced phytoremediaton of petroleum-contaminated soil. International Journal of Phytoremediation, 10, 251–263.
Bačkor, M., Kováčik, J., Piovár, J., Pisani, T., & Loppi, S. (2009). Physiological aspect of cadmium and nickel toxicity in the lichens Peltigera rufescens and Cladina arbuscula Subsp. Mitis. Water, Air, & Soil Pollution. doi:10.100/s11270-009-0133-6.
Bleeker, P. M., Schat, H., Vooijs, R., Verkleij, J. A. C., & Ernst, W. H. O. (2003). Mechanisms of arsenate tolerance in Cytisus striatus. New Phytologist, 157, 33–38.
Bruce, S. L., Noller, B. N., Grigg, A. H., Mullen, B. F., Mulligan, D. R., & Ritchie, P. J. (2003). A field study conducted at kidston gold mine to evaluate the impact of arsenic and zinc from mine tailing to grazing cattle. Toxicology Letters, 137, 23–34.
Caille, N., Zhao, F. J., & McGrath, S. P. (2005). Comparison of root absorption, translocation and tolerance of arsenic in the hyperaccumulator Pteris vittata and the nonhyperaccumulator Pteris tremula. New Phytologist, 165, 755–761.
Castro-Carrillo, L. A., Delgadillo-Martínez, J., Ferrera-Cerrato, R., & Alarcón, A. (2008). Phenanthrene dissipation by Azolla caroliniana utilizing bioaugmentation with hydrocarbonoclastic microorganisms. Interciencia, 33, 1–7 [In Spanish].
Chao-Yang, W., & Tong-Bin, C. (2006). Arsenic accumulation by two brake ferns growing on arsenic mine and their potential in phytoremediation. Chemosphere, 63, 1048–1053.
Dere, S., Günes, T., & Sivaci, R. (1998). Spectrophotometric determination of chlorophyll-A, B and total carotenoid contents of some algae species using different solvents. Turkish Journal of Botany, 22, 13–17.
Doh, S., Camusso, W., Mucciarelli, M., & Fusconi, A. (2010). Arsenate toxicity on the apices of Pisum sativum L. seedling roots: Effects on mitotic activity, chromatin integrity and microtubules. Environmental and Experimental Botany, 69, 17–23.
EPA (Environmental Protection Agency) (1994). Determination of trace elements in water and wastes by inductively coupled plasma-mass spectrometry. Method 200.8 Revision 5,4. In J. T. Creed, C. A. Brockhoff, T. D. Martin (eds.). US Environmental Protection Agency Cincinnati, OHIO 4568, 57pp.
Espinoza, Y., & Gutiérrez, R. (2003). Variabilidad intraespecifica de Azolla filiculoides colectadas en la zona centro-occidental de Venezuela. Revista de la Facultad de Agronomía (LUZ), 20, 156–167.
Francesconi, K., Visoottiviseth, P., Sridokchan, W., & Goessler, W. (2002). Arsenic species in an arsenic hyperaccumulating fern, Pityrogramma calomelanos: A potential phytoremediator of arsenic-contaminated soils. Science of the Total Environment, 284, 27–35.
Gravisu, C., Hernández-Allica, J., Barrutia, O., Alkorta, I., & Becerril, J. M. (2002). Phytoremediation: A technology using green plants to remove contaminants from polluted areas. Review of Environmental Health, 17, 75–90.
Hove, V. C., & Lejeune, A. (2002). The Azolla–Anabaena symbiosis. In. Biology and Environment. Proceeding of the Royal Irish Academy, 102B, 23–26.
Huang, J. W., Poynton, C. Y., Kochian, L. V., & Elless, M. P. (2004). Phytofiltration of arsenic from drinking water using arsenic-hyperaccumulating ferns. Environmental Science & Technology, 38, 3412–3417.
Kabata-Pendias, A., & Pendias, H. (1992). Trace elements in soils and plants. Boca Raton: CRC.
Kanoun-Boulé, M., & Vicente, J. A. F. (2009). Ecophysiological tolerance of duckweeds exposed to copper. Aquatic Toxicology, 91, 1–9.
Kováčik, J., & Backor, M. (2007). Phenylalanine ammonia-lyase and phenolic compounds in chamomile tolerance to cadmium and copper excess. Wate Air Soil Pollution, 185, 185–193.
Krosravi, M., Ganji, T. M., & Rakhshaee, R. (2005). Toxic effect of Pb, Cd, Ni and Zn on Azolla filiculoides in the international Anzali Wetland. International Journal of Environmental Science and Technology, 2, 35–40.
Lepp, N. W. (1981). Effect of heavy metals pollution on plants. Applied Science Publishers. London and New Jersey. 1, 111–141.
Lesikar, B. J., Melton, R. H., Hare, M. F., Hopkins, J., & Dozier, M. C. (2006). Drinking water problems—arsenic: Texas cooperative extension (pp. 1–8). Texas A & M. L-5467S.
Ma, L. Q., Komart, K. M., Tu, C., Zhang, W., Cai, Y., & Kennelly, E. D. (2001). A fern that hyperaccumulates arsenic. Nature, 409, 579–579.
Mascher, R., Lippmann, B., Holzinger, S., & Bergmann, H. (2002). Arsenate toxicity: Effects on oxidative stress reponse molecules and enzymes in red clover plants. Plant Science, 163, 961–969.
Masood, A., Zeeshan, M., & Abraham, G. (2008). Response of growth and antioxidant enzymes in Azolla plants (Azolla pinnata and Azolla filiculoides) exposed to UV-B. Acta Biologica Hungarica, 59, 247–258.
Matschullat, J. (2000). Arsenic in the geosphere—A review. Science of the Total Environment, 249, 297–312.
Matthäus, M. (2002). Antioxidant activity of extracts obtained from residues of different oilseeds. Journal of Agricultural and Food Chemistry, 50, 3444–3452.
Meharg, A. A. (1994). Relationship between plant phosphorus status and the kinetics of arsenate influx in clones of Deschampsia cespitosa (L.) Beauv that differ in their tolerance to arsenate. Plant and Soil, 162, 99–106.
Meharg, A. A. (2002). Arsenic and old plants. New Phytologist, 156, 1–8.
Meharg, A. A., & Hartley-Witaker, M. J. (2002). Arsenic uptake and metabolism in arsenic plant species. New Phytologist, 154, 29–43.
Meharg, A. A., & Macnair, M. R. (1991a). The mechanisms of arsenate tolerance in Deschampsia cespitosa (L.) Beauv and Agrostis capillaris L. New Phytologist, 119, 291–297.
Meharg, A. A., & Macnair, M. R. (1991b). Uptake, accumulation and translocation of arsenate in arsenate-tolerant and nontolerant Holcus lanatus L. New Phytologist, 117, 225–231.
Miller, D. S., Shaw, J. R., Staton, C. R., Barnaby, R., Karlson, K. H., Hamilton, J. W., et al. (2007). MRP2 and acquired tolerance to inorganic arsenic in the kidney of killifish (Fundulus heteroclitus). Toxicological Sciences, 27, 1–41.
Mittler, R. (2002). Oxidative stress, antioxidants, and stress tolerance. Trends in Plant Science, 7, 405–410.
Ortiz-Catón, A., Quintero, L. R., & Ferrera-Cerrato, R. (1992). Efecto de metales pesados en el simbiosistema Azolla Anabaena. Terra, 12, 317–322 [In Spanish].
Perales-Vela, H. V., González-Moreno, S., Montes-Horeasitas, C., & Cañizares-Villanueva, R. O. (2007). Growth, photosynthetic and respiratory responses to sub-lethal copper concentrations in Scenedesmus incrassatulus (Chlorophyceae). Chemosphere, 67, 2274–2281.
Pilon-Smits, E. (2005). Phytoremediation. Annual Review of Plant Biology, 56, 15–39.
Quintero-Lizaola, R., & Ferrera-Cerrato, R. (2000). Azolla helecho fijador de nitrógeno y su potencial en México. In J. J. Peña-Cabriales (Ed), La fijación biológica de nitrógeno en América Latina: El aporte de las técnicas isotópicas. Irapuato. México. [In Spanish].
Rahman, M. A., Hasegawa, H., Ueda, K., Maki, T., Okumura, C., & Rahman, M. M. (2007). Arsenic accumulation in duckweed (Spirodela polyrhiza L.): A good option for phytoremediation. Chemosphere, 69, 493–499.
Rudra, D. T., Sudhakar, S., Seema, M., Nandita, S., Rakesh, T., Dharmendra, K. G., et al. (2006). Arsenic hazards: Strategies for tolerance and remediation by plants. Trends in Biotechnology, 25, 159–165.
Sakihama, Y., Cohen, M. F., Grace, C. S., & Yamasaki, H. (2002). Plant phenolic antioxidant and prooxidant activities: Phenolics induces oxidative damage mediated by metals in plants. Toxicology, 177, 67–80.
Salt, D. E., Smith, R. D., & Raskin, I. (1998). Phytoremediation. Annual Review of Plant Physiology and Plant Molecular Biology, 49, 643–661.
Sánchez-Viveros, G., González-Mendoza, D., Alarcón, A., & Ferrera-Cerrato, R. (2010). Copper effects on photosynthetic activity and membrane leakage of Azolla filiculoides and A. caroliniana. International Journal of Agriculture & Biology, 12, 365–368.
SAS Institute Inc. (2002). The SAS system for windows, ver. 9.0. Cary, USA: SAS Institute Inc.
Sheng-Gen, H., Daryl, J., & Ming-Zu, W. (2005). Characterization of polyamine oxidase from the aquatic nitrogen-fixing fern Azolla imbricate. Plant Science, 169, 185–190.
Singh, N., Ma, L. Q., Srivastava, M., & Rathinasabapathi, B. (2006). Metabolic adaptations to arsenic-induced oxidative stress in Pteris vittata L and Pteris ensiformis L. Plan Science, 170, 274–282.
Singleton, V. L., & Rossi, J. A. (1965). Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. American Journal of Enology and Viticulture, 16, 144–147.
Stepniewska, Z., Bennicelli, R. P., Balakhnina, T. I., Szajnocha, K., Banach, A., & Wolinska, A. (2005). Potential of Azolla caroliniana for the removal of Pb and Cd from wastewaters. International Agrophysics, 19, 251–255.
Tu, C., & Ma, L. Q. (2002). Effects of arsenic concentrations and forms on arsenic uptake by the hyperaccumulator ladder brake. Journal of Environmental Quality, 31, 641–647.
Yoshida, S., Forno, D., Cock, J., & Gomez, K. (1971). Laboratory manual for physiological studies of rice (3rd ed.). Manila Philippines: International Rice Research Institute.
Zhang, X., Ai-jun, L., Fang-Jie, Z., Guo-Zhong, X., Gui-Lan, D., & Yong-Guan, Z. (2008). Arsenic accumulation by the aquatic fern Azolla: Comparison of arsenate uptake, speciation and efflux by A. caroliniana and A. filiculoides. Environmental Pollution, 156, 1156–1149.
Zhao, F. J., Dunham, S. J., & McGrath, S. P. (2002). Arsenic hyperaccumulation by different ferns species. New Phytologist, 156, 27–31.
Zimmerman, W. J., Quintero-Lizaola, R., & Ferrera-Cerrato, R. (1993). The genetic identification of species of agronomic Azolla Lam. indigenous to Mexico. American Fern Journal, 83, 97–104.
Acknowledgments
Authors thank Adan Castillo-Cervantes for technical support during both establishment and maintenance of the experiment. Authors gratefully thank anonymous reviewers for invaluable comments and thorough revisions.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sánchez-Viveros, G., Ferrera-Cerrato, R. & Alarcón, A. Short-Term Effects of Arsenate-Induced Toxicity on Growth, Chlorophyll and Carotenoid Contents, and Total Content of Phenolic Compounds of Azolla filiculoides . Water Air Soil Pollut 217, 455–462 (2011). https://doi.org/10.1007/s11270-010-0600-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11270-010-0600-0