Abstract
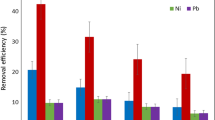
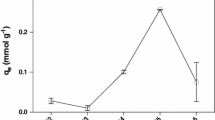
In this work, the potential removal of Cd, Cu, and Zn ions by non-living macrophytes Egeria densa has been studied. The adsorption kinetic and equilibrium experiments of these three metals on E. densa were performed in batch systems with controlled temperature and constant shaking. It was observed that all metal adsorption rates have increased when the pH was increasing. A pH threshold of 5 was established for use in adsorption experiments in order to avoid the metal precipitation. For adsorption kinetic tests, the equilibrium times for all metals were around 45 to 60 min. The equilibrium data at pH 5 were better described by the Langmuir isotherm than the Freundlich one, with the adsorption rate and maximum metal content values of 0.43 L g−1 and 1.25 mequiv g−1 for Cd, 4.11 L g-−1 and 1.43 mequiv g−1 for Cu, and 0.83 L g−1 and 0.93 mequiv g−1 for Zn. These adsorption parameters for E. densa resemble or are better than those for other biosorbents already studied, suggesting that the macrophytes E. densa as a biosorbent has a good metal removal potential for applications in effluent treatment systems.





Similar content being viewed by others
References
Ahluwalia, S. S., & Goyal, D. (2007). Microbial and plant derived biomass for removal of heavy metals from wastewater. Bioresource Technology, 98, 2243–2257. doi:10.1016/j.biortech.2005.12.006.
Al-Rub, F. A., El-Naas, M. H., Ashour, I., & Al-Marzoupi, M. (2006). Biosorption of koper on Chlorella vulgaris from single, binary and ternary metals aqueous solutions. Process Biochemistry, 41, 457–464. doi:10.1016/j.procbio.2005.07.018.
Bhattacharyya, K. G., & Sharma, A. (2005). Kinetics and thermodynamics of methylene blue adsorption on neem (Azadirachta indica) leaf powder. Dyes and Pigments, 65, 51–59. doi:10.1016/j.dyepig.2004.06.016.
Chojnacka, K., Chojnacki, A., & Gorékcka, H. (2005). Biosorption of Cr+3, Cd+2 and Cu+2 ions blue-green algae Spirulina sp.: Kinectics, equilibrium and the mechanism of the process. Chemosphere, 59, 75–84. doi:10.1016/j.chemosphere.2004.10.005.
Chubar, N., Behrends, T., & Cappellen, P. V. (2008). Biosorption of metals (Cu2+, Zn2+) and anions (F−, H2PO4 −) by viable and autoclaved cells of the Gram-negative bacterium Shewanella putrefaciens. Colloids and Surfaces. B, Biointerfaces, 65, 26–133. doi:10.1016/j.colsurfb.2008.03.006.
Cruz, C. C. V., Costa, A. C. A., Henriques, C. A., & Luna, A. S. (2004). Kinetic modeling and equilibrium studies during cadmium biosorption by dead Sargassum sp. Biomass. Bioresource Technology, 91, 249–257. doi:10.1016/S0960-8524(03)00194-9.
Dang, V. B. H., Doan, H. D., Dang-Vu, T., & Lohi, A. (2009). Equilibrium and kinetics of biosorption of cadmium(II) and copper(II) ions by wheat straw. Bioresource Technology, 100, 211–219. doi:10.1016/j.biortech.2008.05.031.
Davis, T. A., Volesky, B., & Mucci, A. (2003). A review of the biochemistry of heavy metal biosorption by brown algae. Water Research, 37, 4311–4330. doi:10.1016/S0043-1354(03)00293-8.
Fagundes-Klen, M. R., Ferri, P., Martins, T. D., Tavares, C. R. G., & Silva, E. A. (2007). Equilibrium study of the binary mixture of cadmium–zinc ions biosorption by the Sargassum filipendula species using adsorption isotherms models and neural network. Biochemical Engineering Journal, 34, 136–146.
Romera, E., González, F., Ballester, A., Blázquez, M. L., & Muñoz, J. A. (2008). Biosorption of heavy metals by Fucus spiralis. Bioresource Technology, 99, 4684–4693.
Ho, Y. S. (2005). Effect of pH on lead removal from water using tree fern as the sorbent. Bioresource Technology, 96, 1292–1296. doi:10.1016/j.biortech.2004.10.011.
Ho, Y. S., & Mckay, G. (1998). Pseudo-second order model for sorption processes. Process Biochemistry, 34, 451–465. doi:10.1016/S0032-9592(98)00112-5.
Ho, Y. S., & Mckay, G. (2000). The kinetics of sorption of divalent metal ions onto sphagnum moss peat. Water Research, 34, 735–742. doi:10.1016/S0043-1354(99)00232-8.
Liu, Y. G., Wang, X., Zeng, G. M., Qu, D., Gu, J. J., Zhou, M., & Chai, L. Y. (2007). Cadmium-induced oxidative stress and response of the ascorbate–glutathione cycle in Bechmeria nivea (L.) Gaud. Chemosphere, 69, 99–107. doi:10.1016/j.chemosphere.2007.04.040.
Lodeiro, P., Cordeiro, B., Barriada, J. L., Herrero, R., & Sastre de Vicente, M. E. (2005). Biosorption of cadmium by biomass of Brown marine macroalgae. Bioresource Technology, 96, 1796–1803. doi:10.1016/j.biortech.2005.01.002.
Martínez, M., Miralles, N., Hidalgo, S., Fiol, N., Villaescusa, I., & Poch, J. (2006). Removal of lead(II) and cadmium(II) from aqueous solutions using grape stalk waste. Journal of Hazardous Materials, 133, 203–211. doi:10.1016/j.jhazmat.2005.10.030.
Ozcan, A., Ozcan, A. S., Tunali, S., Akar, T., & Kiran, I. (2005). Determination of the equilibrium, kinetic and thermodynamic parameters of adsorption of copper(II) ions onto seeds of Capsicum annuum. Journal of Hazardous Materials, 124, 200–208. doi:10.1016/j.jhazmat.2005.05.007.
Ozsoy, H. D., Kumbur, H., Saha, B., & Van Leeuwen, J. H. (2008). Use of Rhizopus oligosporus produced from food processing wastewater as a biosorbent for Cu(II) ions removal from the aqueous solutions. Bioresource Technology, 99, 4943–4948. doi:10.1016/j.biortech.2007.09.017.
Saygideger, S., Gulnaz, O., Istifli, E. S., & Yucel, N. (2005). Adsorption of Cd(II), Cu(II) and Ni(II) ions by Lemna minor L.: Effect of physicochemical environment. Journal of Hazardous Materials, 126, 96–104. doi:10.1016/j.jhazmat.2005.06.012.
Acknowledgments
We thank the Araucaria Foundation and the CNPq for financial support of this study. We also thank both Scientific and Technological Research Centers located in Cascavel (Fundetec) and in Toledo (Funtec) cities in the Brazilian Paraná State for the technical support in AAS technique.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pietrobelli, J.M.T.d.A., Módenes, A.N., Fagundes-Klen, M.R. et al. Cadmium, Copper and Zinc Biosorption Study by Non-Living Egeria densa Biomass. Water Air Soil Pollut 202, 385–392 (2009). https://doi.org/10.1007/s11270-009-9987-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11270-009-9987-x