Abstract
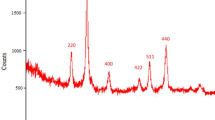
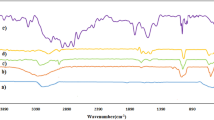
A new 4-aminomethylbenzoic acid-functionalized Fe3O4 magnetic nanoparticles as a hybrid heterogeneous catalyst was synthesised and characterized by FT-IR, XRD, TGA, TEM, SEM and VSM techniques. The catalytic activity of this nanocatalyst was probed through one-pot synthesis of 2-amino-4H-chromene derivatives from three component reactions of various aldehydes, malononitrile and dimedone. The chemically and thermally stable catalyst was easily recovered using an external magnet and reused for at least five successive runs without significant loss of its activity. The simplicity of the method, high yields of the products, mild reaction conditions and low reaction times are the other advantages of this procedure.









Similar content being viewed by others
References
E. Karaoğlu, A. Baykal, H. Erdemi, L. Alpsoy, H. Sozeri, J. Alloys Compd. 509, 9218 (2011)
Q.M. Kainz, O. Reiser, Acc. Chem. Res. 47, 667 (2014)
R.B. Nasir Baig, M.N. Nadagouda, R.S. Varma, Coord. Chem. Rev. 287, 137 (2014)
W. Teunissen, A.A. Bol, J.W. Geus, Catal. Today 48, 329 (1999)
T.-J. Yoon, W. Lee, Y.-S. Oh, J.-K. Lee, N. J. Chem. 27, 227 (2003)
H. Yoon, S. Ko, J. Jang, Chem. Commun. 1468 (2007)
D. Lee, J. Lee, H. Lee, S. **, T. Hyeon, B.M. Kim, Adv. Synth. Catal. 348, 41 (2006)
C.Ó. Dálaigh, S.A. Corr, Y. Gun’ko, S.J. Connon, Angew. Chem. Int. Ed. 46, 4329 (2007)
K.K. Senapati, S. Roy, C. Borgohain, P. Phukan, J. Mol. Catal. A Chem. 352, 128–134 (2012)
D. Lee, J. Lee, H. Lee, S. **, T. Hyeon, B.M. Kim, Adv. Synth. Catal. 348, 41 (2006)
R. Abu-Rezig, H. Alper, D. Wang, M.L. Post, J. Am. Chem. Soc. 128, 5279 (2006)
G.R. Green, J.M. Evans, A.K. Vong, in Pyrans and their benzo derivatives synthesis, ed. by A.R. Katritzky, C.W. Rees, E.F.V. Scriven. Comprehensive Heterocyclic Chemistry II, vol 5 (Pergamon Press, Oxford, 1995), p. 469
L.L. Andreani, E. Lapi, Boll. Chim. Farm. 99, 583 (1960)
Y.L. Zhang, B.Z. Chen, K.Q. Zheng, M.L. Xu, X.H. Lei, Chem. Abstr. 96, 135383e (1982)
L. Bonsignore, G. Loy, D. Secci, A. Calignano, Eur. J. Med. Chem. 28, 517 (1993)
E.C. Witte, P. Neubert, A. Roesch, Chem. Abstr. 104, 224915f (1986)
I.A. Shuklov, N.V. Dubrovina, A. Børner, Synthesis 2925 (2007)
J.P. Bégué, D. Bonnet-Delpon, B. Crousse, Synlett 18 (2004)
T. Wejrzanowski, R. Pielaszek, A. Opalińska, H. Matysiak, W. Lojkowski, K.J. Kurzydlowski, Appl. Surf. Sci. 253, 204 (2006)
R. Pielaszek, J. Appl. Crystallogr. 1, 43 (2003)
F. Noorizadeh, M.T. Maghsoodlou, N. Hazeri, M. Kangani, Res. Chem. Intermed. (2014). doi:10.1007/s11164-014-1710-x
S. Rostamnia, A. Morsali, Inorg. Chim. Acta 411, 113 (2014)
N. Hazeri, M.T. Maghsoodlou, F. Mir, M. Kangani, H. Saravani, E. Molashahi, Chin. J. Catal. 35, 391 (2014)
A. Khazaei, F. Gholami, V. Khakyzadeh, A.R. Moosavi-Zare, J. Afsar, RSC Adv. 5, 14305 (2015)
L.M. Wang, J.H. Shao, H. Tian, Y.H. Wang, B. Liu, J. Fluor. Chem. 127, 97–100 (2006)
A. Patra, T. Mahapatra, J. Chem. Res. Synop. 34, 689–693 (2010)
G. Brahmachari, B. Banerjee, ACS Sustain. Chem. Eng. 2, 411 (2014)
D. Tahmassebi, J.A. Bryson, S.I. Binz, Synth. Commun. 41, 2701 (2011)
M. Abdollahi-Alibeik, F. Nezampour, React. Kinet. Mech. Catal. 108, 213 (2013)
N. Montazeri, T. Noghani, M. Ghorchibeigy, R. Zoghi, J. Chem. Article ID 596171 (2014)
K. Aswin, S.S. Mansoor, K. Logaiya, S.P.N. Sudhan, V.S. Malik, H. Ramadoss, Res. Chem. Intermed. 40, 2583 (2014)
Acknowledgements
This research was supported by the Research Council of the Razi University in Iran.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Khodaei, M.M., Alizadeh, A. & Haghipour, M. A simple synthesis of magnetic nanoparticles-supported 4-aminomethylbenzoic acid as a highly efficient and reusable catalyst for synthesis of 2-amino-4H-chromene derivatives. Res Chem Intermed 46, 1033–1045 (2020). https://doi.org/10.1007/s11164-017-3008-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-017-3008-2