Abstract
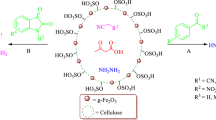
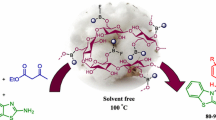
A cellulose-based magnetic nano-composite supported SO3H group was synthesized and characterized using Fourier transform infrared spectroscopy, energy-dispersive X-ray spectroscopy, thermogravimetric analysis, X-ray diffraction, and scanning electron microscopy. Thereafter, its capability to promote the one-pot, three-component synthesis of pyrimido [4,5-b] quinolone and pyrido [2,3-d] pyrimidine derivatives was evaluated. The obtained results are indicative of excellent yields and short reaction times. The biopolymer based catalyst is readily recovered and reused several times without a decrease in yield.
Graphical Abstract













Similar content being viewed by others
References
J. Safari, S. Gandomi-Ravandi, Z. Akbari, J. Adv. Res. 4, 6 (2013)
I.R. Siddiqui, P. Rai, R. Rahila, H. Sagir, P. Singh, RSC Adv. 5, 35 (2015)
R.M. Singh, N. Sharma, R. Kumar, M. Asthana, S. Upadhyay, Tetrahedron Lett. 68, 50 (2012)
J.M. Khurana, A. Chaudhary, B. Nand, A. Lumb, Tetrahedron Lett. 53, 24 (2012)
I. Devi, B.S.D. Kumar, P.J. Bhuyan, Tetrahedron Lett. 44, 45 (2003)
N.A. Hassan, M.I. Hegab, F.M. Abdel-Motti, S.H.A. Hebah, F.M.E. Abdel-Megeid, A.I. Hashem, J. Heterocycl. Chem. 44, 4 (2007)
A. Shaabani, Z. Hezarkhani, M.T. Faroghi, Monatsh. Chem. (2016). doi:10.1007/s00706-016-1717-7
M. Shamsi-Sani, F. Shirini, M. Abedini, M. Seddighi, Res. Chem. Intermed. 42, 2 (2016)
E. Kolvari, N. Koukabi, M.M. Hosseini, J. Mol. Catal. A: Chem. 397, 68 (2015)
A.R. Kiasat, L. Hemat-Alian, Res. Chem. Intermed. 41, 2 (2015)
M. Wu, Y. Wang, D. Wang, M. Tan, P. Li, W. Wu, N. Tsubaki, J. Porous Mater. 23, 1 (2016)
B. Sadeghi, M.H. Sowlat Tafti, J. Iran. Chem. Soc. 13, 8 (2016)
M.A. Zolfigol, Tetrahedron 57, 46 (2001)
F. Nemati, M.M. Heravi, R. Saeedi Rad, Chin. J. Catal. 33, 11–12 (2012)
E. Mehrasbi, Y. Sarrafi, A. Vahid, H. Alinezhad, Res. Chem. Intermed. 41, 7 (2015)
S. Mahdudi, D. Saberi, A. Heydari, J. Iran. Chem. Soc. 12, 5 (2015)
A. Amoozadeh, S. Golian, S. Rahmani, RSC Adv. 5, 57 (2015)
A. Amoozadeh, S. Rahmani, J. Mol. Catal. A: Chem. 396, 96–107 (2015)
E. Kolvari, N. Koukabi, M.M. Hosseini, M. Vahidian, E. Ghobadi, RSC Adv. 6, 9 (2016)
F. Nemati, A. Elhampour, M.B. Natanzi, S. Sabaqian, J. Iran. Chem. Soc. 13, 6 (2016)
R.H. Vekariya, H.D. Patel, RSC Adv. 5, 61 (2015)
A.M. Salgueiro, A.L. Daniel-da-Silva, A.V. Girão, P.C. Pinheiro, T. Trindade, Chem. Eng. J. 229, 276–284 (2013)
C. Esmaeili, M. Ghasemi, L.Y. Heng, S.H.A. Hassan, M.M. Abdi, W.R.W. Daud, H. Ilbeygi, A.F. Ismail, Carbohydr. Polym. 114, 253–259 (2014)
S. Rostamnia, B. Zeynizadeh, E. Doustkhah, A. Baghban, K.O. Aghbash, Catal. Commun. 68, 77–83 (2015)
Y. Habibi, L.A. Lucia, O.J. Rojas, Chem. Rev. 110, 6 (2010)
C. Johansson, J. Bras, I. Mondragon, P. Nechita, D. Plackett, P. Simon, D.G. Svetec, S. Virtanen, M.G. Baschetti, C. Breen, Bioresources. 7, 2 (2012)
Z. Ghasemi, S. Shojaei, A. Shahrisa, RSC Adv. 6, 61 (2016)
A. Shaabani, H. Nosrati, M. Seyyedhamzeh, Res. Chem. Intermed. 41, 6 (2015)
L. Edjlali, R.H. Khanamiri, J. Abolhasani Monatsh. Chem. 146, 8 (2015)
B. Liu, Z. Zhang, K. Huang, Cellulose 20, 4 (2013)
H.R. Shaterian, F. Rigi, Res. Chem. Intermed. 40, 5 (2014)
A. Shaabani, A. Rahmati, Z. Badri, Catal. Commun. 9, 1 (2008)
R.H. Vekariya, H.D. Patel, ARKIVOC 1, 136–159 (2015)
R.K. Sharma, S. Dutta, S. Sharma, R. Zboril, R.S. Varma, M.B. Gawande, Green Chem. 18, 11 (2016)
B. Liu, Z. Zhang, ACS Catal. 6, 326 (2016)
B. Liu, Y. Ren, Z. Zhang, Green Chem. 17, 1610 (2015)
Z. Zhang, J. Zhen, B. Liu, K. Lv, K. Deng, Green Chem. 17, 1308 (2015)
Sh Yin, J. Sun, B. Liu, Z. Zhang, J. Mater. Chem. A 3, 4992 (2015)
Z. Zhang, Z. Yuan, D. Tang, Y. Ren, K. Lv, B. Liu, Chem. Sus. Chem. 7, 3496 (2014)
N. Mei, B. Liu, J. Zheng, K. Lv, D. Tang, Z. Zhang, Catal. Sci. Technol. 5, 3194 (2015)
F. Nemati, A. Elhampour, H. Farrokhi, M. Bagheri, Natanzi. Catal. Commun. 66, 15 (2015)
F. Nemati, M.M. Heravi, A. Elhampour, RSC Adv. 5, 57 (2015)
F. Nemati, S.H. Nikkhah, A. Elhampour, Chin. Chem. Lett. 26, 11 (2015)
F. Nemati, A.G.G. Ghiyaei, B. Notash, M.H. Shayegan, V. Amani, Tetrahedron Lett. 55, 25 (2014)
F. Nemati, A. Elhampour, S. Zulfaghari, Phosphorus, Sulfur Silicon Relat. Elem. 190, 10 (2015)
F. Nemati, A. Elhampour, Res. Chem. Intermed. 42, 7611 (2016)
A. Elhampour, F. Nemati, M. Kaveh, Chem. Lett. 45, 223 (2016)
A. Elhampour, F. Nemati, J. Chin. Chem. Soc. 63, 653 (2016)
L.C. Fidale, M. Nikolajski, T. Rudolph, S. Dutz, F.H. Schacher, T. Heinze, J. Colloid Interface Sci. 390, 1 (2013)
M.H. Beyki, M. Bayat, S. Miri, F. Shemirani, H. Alijani, Ind. Eng. Chem. Res. 53, 39 (2014)
X. Sun, L. Yang, Q. Li, J. Zhao, X. Li, X. Wang, H. Liu, Chem. Eng. J. 241, 174–183 (2014)
P.V. Chavan, K.S. Pandit, U.V. Desai, M.A. Kulkarni, P.P. Wadgaonkar, RSC Adv. 4, 79 (2014)
H.R. Shaterian, M. Ghashang, M. Feyzi, Appl. Catal. A 345, 2 (2008)
S. Peng, H. Meng, Y. Ouyang, J. Chang, Ind. Eng. Chem. Res. 53, 6 (2014)
H.M. Savanur, R.G. Kalkhambkar, G. Aridoss, K.K. Laali, Tetrahedron Lett. 57, 3029 (2016)
J.H. Clark, D.J. Macquarrie, J. Sherwood, Chem. Eur. J. 19, 5174 (2013)
F. Nemati, R. Saeedirad, Chin. Chem. Lett. 24, 5 (2013)
K. Tabatabaeian, A.F. Shojaei, F. Shirini, S.Z. Hejazi, M. Rassa, Chin. Chem. Lett. 25, 2 (2014)
S.-J. Ji, S.-N. Ni, F. Yang, J.-W. Shi, G.-L. Dou, X.-Y. Li, X.-S. Wang, D.-Q. Shi, J. Heterocycl. Chem. 45, 3 (2008)
G.K. Verma, K. Raghuvanshi, R. Kumar, M.S. Singh, Tetrahedron Lett. 53, 4 (2012)
K. Mohammadi, F. Shirini, A. Yahyazadeh, RSC Adv. 5, 23586 (2015)
Acknowledgment
The authors gratefully acknowledge Semnan University Research Council for financial support of this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Osanlou, F., Nemati, F. & Sabaqian, S. An eco-friendly and magnetized biopolymer cellulose-based heterogeneous acid catalyst for facile synthesis of functionalized pyrimido[4,5-b]quinolines and indeno fused pyrido[2,3-d]pyrimidines in water. Res Chem Intermed 43, 2159–2174 (2017). https://doi.org/10.1007/s11164-016-2752-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-016-2752-z