Abstract
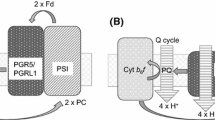
Light-driven photosynthetic electron transport is coupled to the movement of protons from the chloroplast stroma to the thylakoid lumen. The resulting proton motive force that is generated is used to drive the conformational rotation of the transmembrane thylakoid ATPase enzyme which converts ADP (adenosine diphosphate) and Pi (inorganic phosphate) into ATP (adenosine triphosphate), the energy currency of the plant cell required for carbon fixation and other metabolic processes. According to Mitchell’s chemiosmotic hypothesis, the proton motive force can be parsed into the transmembrane proton gradient (ΔpH) and the electric field gradient (Δψ), which are thermodynamically equivalent. In chloroplasts, the proton motive force has been suggested to be split almost equally between Δψ and ΔpH (Kramer et al., Photosynth Res 60:151–163, 1999). One of the central pieces of evidence for this theory is the existence of a steady-state electrochromic shift (ECS) absorption signal detected ~515 nm in plant leaves during illumination. The interpretation of this signal is complicated, however, by a heavily overlap** absorption change ~535 nm associated with the formation of photoprotective energy dissipation (qE) during illumination. In this study, we present new evidence that dissects the overlap** contributions of the ECS and qE-related absorption changes in wild-type Arabidopsis leaves using specific inhibitors of the ΔpH (nigericin) and Δψ (valinomycin) and separately using leaves of the Arabidopsis lut2npq1 mutant that lacks qE. In both cases, our data show that no steady-state ECS signal persists in the light longer than ~60 s. The consequences of our observations for the suggesting parsing of steady-state thylakoid proton motive force between (ΔpH) and the electric field gradient (Δψ) are discussed.






Similar content being viewed by others
References
Avenson TJ, Cruz JA, Kramer DM (2004) Modulation of energy-dependent quenching of excitons in antennae of higher plants. Proc Natl Acad Sci USA 101:5530–5535
Barber J (1976) Ionic regulation in intact chloroplasts and its effect on primary photosynthetic processes. In: Barber J (ed) The intact chloroplast, vol 1. Elsevier North-Holland Biomedical Press, Amsterdam, pp 89–134
Barber J (1982) Influence of surface charges on thylakoid structure and function. Annu Rev Plant Physiol 33:261–295
Barber J, Mills J, Nicolson J (1974) Studies with cation specific ionophores show that within the intact chloroplast Mg2+ acts as the main exchange cation for H+ pum**. FEBS Lett 49:106–110
Bendall DS, Davenport HE, Hill R (1971) Cytochrome components in chloroplasts of the higher plants. Methods Enzymol 23A:327–344
Bennoun P (1994) Chlororespiration revisited: mitochondrial–plastid interactions in Chlamydomonas. Biochim Biophys Acta 1186:59–66
Berry S, Rumberg B (1996) H+/ATP coupling ratio at the unmodulated CF0CF1-ATP synthase determined by proton flux measurements. Biochim Biophys Acta 1276:51–56
Bilger W, Björkman O, Thayer SS (1989) Light-induced spectral absorbance changes in relation to photosynthesis and the epoxidation state of xanthophyll cycle components in cotton leaves. Plant Physiol 91:542–551
Bulychev AA (1984) Different kinetics of membrane potential formation in dark adapted and pre-illuminated chloroplasts. Biochim Biophys Acta 766:647–652
Bulychev AA, Andrianov VK, Kurella GA, Litvin FF (1972) Micro-electrode measurements of the transmembrane potential of chloroplasts and its photoinduced changes. Nature 236:175–177
Checchetto V, Segalla A, Allorent G, La Rocca N, Leanza L, Giacometti GM, Uozumi N, Finazzi G, Bergantino E, Szabo I (2012) Thylakoid potassium channel is required for efficient photosynthesis in cyanobacteria. Proc Natl Acad Sci USA 109:11043–11048
Chow WS, Wagner G, Hope AB (1976) Light-dependent redistribution of ions in isolated spinach chloroplasts. Aust J Plant Physiol 3:853–861
Crouchman S, Ruban A, Horton P (2006) PsbS enhances nonphotochemical fluorescence quenching in the absence of zeaxanthin. FEBS Lett 580:2053–2058
Cruz JA, Sacksteder CA, Kanazawa A, Kramer DM (2001) Contribution of electric field (Δψ) to steady-state transthylakoid proton motive force (pmf) in vitro and in vivo. Control of pmf parsing into Δψ and ΔpH by ionic strength. Biochemistry 40:1226–1237
Deamer DW, Crofts AR, Packer L (1966) Mechanisms of light-induced structural changes in chloroplasts. I. Light-scattering increments and ultrastructural changes mediated by proton transport. Biochim Biophys Acta 131:81–96
Demmig-Adams B (1990) Carotenoids and photoprotection in plants: a role for the xanthophyll zeaxanthin. Biochim Biophys Acta 1020:1–24
Dilley RA, Vernon LP (1965) Ion and water transport processes related to the light dependent shrinkage of spinach chloroplasts. Arch Biochem Biophys 111:365–375
Duysens LNM (1954) Reversible changes in the absorption spectrum of Chlorella upon irradiation. Science 120:353–354
Giersch C, Heber U, Kobayashi Y, Inoue Y, Shibata K, Heldt HW (1980) Energy charge, phosphorylation potential and proton motive force in chloroplasts. Biochim Biophys Acta 590:59–73
Heber U (1969) Conformational changes of chloroplasts induced by illumination of leaves in vivo. Biochim Biophys Acta 180:302–319
Hind G, Nakatani HY, Izawa S (1974) Light-dependent redistribution of ions in suspensions of chloroplast thylakoid membranes. Proc Natl Acad Sci USA 71:1484–1488
Horton P, Ruban AV, Rees D, Pascal AA, Noctor G, Young AJ (1991) Control of light harvesting function in chloroplast membranes by aggregation of the LHCII chlorophyll–protein complex. FEBS Lett 292:1–2
Ilioaia C, Johnson MP, Duffy CDP, Pascal AA, van Grondelle R, Robert B, Ruban AV (2011) Origin of absorption changes associated with photoprotective energy dissipation in the absence of zeaxanthin. J Biol Chem 286:91–98
Johnson MP, Ruban AV (2010) Arabidopsis plants lacking PsbS protein possess photoprotective energy dissipation. Plant J 61:283–289
Johnson MP, Perez-Bueno ML, Zia A, Horton P, Ruban AV (2009) The zeaxanthin-independent and zeaxanthin-dependent qE components of nonphotochemical quenching involve common conformational changes within the photosystem II antenna in Arabidopsis. Plant Physiol 149:1061–1075
Junesch U, Gräber P (1985) The rate of ATP synthesis as a function of ΔpH in normal and dithiothreitol-modified chloroplasts. Biochim Biophys Acta 809:429–434
Junge W (1977) Membrane potentials in photosynthesis. Ann Rev Plant Physiol 28:503–536
Junge W, Witt HT (1968) On the ion transport system in photosynthesis: investigations on a molecular level. Z Naturforsch B 23:244–254
Kaim G, Dimroth P (1998) ATP synthesis by F-type ATP synthase is obligatorily dependent on the transmembrane voltage. EMBO J 18:4118–4127
Kramer DM, Crofts AR (1989) Activation of the chloroplast ATPase measured by the electrochromic change in leaves of intact plants. Biochim Biophys Acta 976:28–41
Kramer DM, Sacksteder CA (1998) A diffused-optics flash kinetic spectrophotometer (DOFS) for measurements of absorbance changes in intact plants in the steady-state. Photosynth Res 56:103–112
Kramer DM, Wise RR, Frederick JR, Alm DM, Hesketh JD, Ort DR, Crofts AR (1990) Regulation of coupling factor in field-grown sunflower: a Redox model relating coupling factor activity to the activities of other thioredoxin-dependent chloroplast enzymes. Photosynth Res 26:213–222
Kramer DM, Sacksteder CA, Cruz JA (1999) How acidic is the lumen? Photosynth Res 60:151–163
Kramer DM, Cruz JA, Kanazawa A (2003) Balancing the central roles of the thylakoid proton gradient. Trends Plant Sci 8:27–32
Krause GH (1973) The high-energy state of the thylakoid system as indicated by chlorophyll fluorescence and shrinkage. Biochim Biphys Acta 292:715–728
Marmagne A, Vinauger-Douard M, Monachello D, de Longevialle AF, Charon C, Allot M, Rappaport F, Wollman FA, Barbier-Brygoo H, Ephritikhine G (2007) Two members of the Arabidopsis CLC (chloride channel) family, AtCLCe and AtCLCf, are associated with thylakoid and Golgi membranes, respectively. J Exp Bot 58:3385–3393
Mitchell P (1961) Coupling of phosphorylation to electron and hydrogen transfer by a chemi-osmotic type of mechanism. Nature 191:144–148
Mitchell P (1966) Chemiosmotic coupling in oxidative and photosynthetic phosphorylation. Biol Rev 41:445–502
Morita S, Itoh S, Nishimura M (1982) Correlation between the activity of membrane bound ATPase and the decay rate of flash-induced 515 nm absorbance change inc chloroplasts in intact leaves, assayed by means of rapid isolation of chloroplasts. Biochim Biophys Acta 679:125–130
Murakami S, Packer L (1970a) Light-induced changes in the conformation and configuration of the thylakoid membrane of Ulva and Porphyra chloroplasts in vivo. Plant Physiol 45:289–299
Murakami S, Packer L (1970b) Protonation and chloroplast membrane structure. J Cell Biol 47:332–351
Neumann J, Jagendorf AT (1964) Light-induced pH changes related to phosphorylation by chloroplasts. Arch Biochem Biophys 107:109–119
Niyogi KK, Grossman AR, Bjorkman O (1998) Arabidopsis mutants define a central role for the xanthophyll cycle in the regulation of photosynthetic energy conversion. Plant Cell 10:1121–1134
Niyogi KK, Shih C, Chow WS, Pogson BJ, Dellapenna D, Björkman O (2001) Photoprotection in a zeaxanthin- and lutein-deficient double mutant of Arabidopsis. Photosynth Res 67:139–145
Noctor G, Ruban AV, Horton P (1993) Modulation of ∆pH-dependent nonphotochemical quenching of chlorophyll fluorescence in spinach chloroplasts. Bicohim Biophys Acta 1183:339–344
Ort DR, Dilley RA (1976) Photophosphorylation as a function of illumination time. I. Effects of permeant cations and permeant anions. Biochim Biophys Acta 449:95–107
Pick U, Rottenberg H, Avron M (1974) The dependence of photophosphorylation in chloroplasts on ΔpH and external pH. FEBS Lett 48:32–36
Pottosin II, Schoneckt G (1996) Ion channel permeable for divalent and monovalent cations in native spinach thylakoid membranes. J Mem Biol 152:223–233
Reed PW (1979) Ionophores. Methods Enzymol 55:435–454
Remiš D, Bulychev AA, Kurella GA (1986) The electrical and chemical components of the protonmotive force in chloroplasts as measured with capillary and pH-sensitive microelectrodes. Biochim Biophys Acta 852:68–73
Rottenberg H, Grunwald T, Avron M (1972) Determination of ΔpH in chloroplasts. Eur J Biochem 25:71–74
Ruban AV, Horton P, Young AJ (1993a) Aggregation of higher plant xanthophylls: differences in absorption spectra and in the dependency on solvent polarity. J Photochem Photobiol 21:229–234
Ruban AV, Young AJ, Horton P (1993b) Induction of non-photochemical energy dissipation and absorbance changes in leaves. Plant Physiol 102:741–750
Ruban AV, Pascal AA, Robert B, Horton P (2002) Activation of zeaxanthin is an obligatory event in the regulation of photosynthetic light harvesting. J Biol Chem 277:7785–7789
Ruban AV, Johnson MP, Duffy CDP (2012) The photoprotective molecular switch in the Photosystem II antenna. Biochim Biophys Acta 1817:167–181
Schönfeld M, Neumann J (1977) Proton conductance of the thylakoid membrane: modulation by light. FEBS Lett 73:51–54
Schönknecht G, Hedrich R, Junge W, Raschke K (1988) A voltage dependent chloride channel in the photosynthetic membrane of a higher plant. Nature 336:589–592
Schröppel-Meier G, Kaiser WM (1988) Ion homeostasis in chloroplasts under salinity and mineral deficiency: I. Solute concentrations in leaves and chloroplasts from spinach plants under NaCl or NaNO3 salinity. Plant Physiol 87:822–827
Schuldiner S, Rottenberg H, Avron M (1972) Determination of ΔpH in chloroplasts. 2. Fluorescent amines as a probe for the determination of ΔpH in chloroplasts. Eur J Biochem 25:64–70
Seelert H, Dencher NA, Muller DJ (2003) Fourteen protomers compose the oligomer III of the proton-rotor in spinach chloroplast ATP synthase. J Mol Biol 333:337–344
Shavit N, Dilley RA, San Pietro A (1968) Ion translocation in isolated chloroplasts. Uncoupling of photophosphorylation and translocation of K+ and H+ ions induced by Nigericin. Biochemistry 7:2356–2363
Slovacek RE, Hind G (1981) Correlation between photosynthesis and the transthylakoid proton gradient. Biochim Biophys Acta 635:393–404
Steigmiller S, Turina P, Gräber P (2008) The thermodynamic H+/ATP ratios of the H+-ATPsynthases from chloroplasts and Escherichia coli. Proc Natl Acd Sci USA 105:3745–3750
Takizawa K, Cruz JA, Kanazawa A, Kramer DM (2007) The thylakoid proton motive force in vivo. Quantitative, non-invasive probes, energetics, and regulatory consequences of light-induced pmf. Biochim Biophys Acta 1767:1233–1244
Thorne SW, Horvath G, Kahn A, Boardman NK (1975) Light-dependent absorption and selective scattering changes at 518 nm in chloroplast thylakoid membranes. Proc Natl Acad Sci USA 72:3858–3862
Tikhonov AN (2012) Energetic and regulatory role of proton potential in chloroplasts. Biochemistry (Moscow) 77:956–974
Tikhonov AN, Khomutov GB, Ruuge EK, Blumenfeld LA (1981) Electron transport in chloroplasts effects of photosynthetic control monitored by the intrathylakoid pH. Biochim Biophys Acta 637:321–333
Tikhonov AN, Agafonov RV, Grigor’ev IA, Kirilyuk IA, Ptushenko VV, Trubitsin BV (2007) Spin-probes designed for measuring the intrathylakoid pH in chloroplasts. Biochim Biophys Acta 1777:285–294
van Kooten O, Snel JFH, Vredenberg WJ (1986) Photosynthetic free energy transduction related to electrical potential changes across the thylakoid membrane. Photosynth Res 9:211–227
von Ballmoos C, Wiedenmann A, Dimroth P (2009) Essentials for ATP synthesis by F1F0 ATP synthases. Annu Rev Biochem 78:649–672
Vredenberg WJ, Bulychev AA (1976) Changes in the electrical potential across the thylakoid membranes of illuminated intact chloroplasts in the presence of membrane-modifying agents. Plant Sci Lett 7:101–107
Werdan K, Heldt HW, Milovancev M (1975) The role of pH in the regulation of carbon fixation in the chloroplast stroma. Studies on CO2 fixation in the light and dark. Biochim Biophys Acta 396:276–292
Witt HT (1971) Coupling of quanta, electrons, fields, ions and phosphorylation in the functional membrane of photosynthesis. Quart Rev Biophys 4:365–477
Yamamoto HY, Nakayama TOM, Chichester CO (1962) Studies on the light and dark interconversions of leaf xanthophylls. Arch Biohem Biophys 97:168–173
Acknowledgements
The authors wish to thank the Royal Society, the UK Biotechnology and Biological Sciences Research Council (BBSRC), and the Engineering and Physical Sciences Research council (EPSRC) for funding and Professor Peter Horton (University of Sheffield, UK) for very valuable discussions.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Johnson, M.P., Ruban, A.V. Rethinking the existence of a steady-state Δψ component of the proton motive force across plant thylakoid membranes. Photosynth Res 119, 233–242 (2014). https://doi.org/10.1007/s11120-013-9817-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-013-9817-2