Abstract
Background and aims
Amaranth is an important underutilized crop with high nutritional value but due to acidic soils and low nutrient availability, its production is low in Uttarakhand hills. Use of inorganic chemicals is restricted as they may disturb the fragile Himalayan ecosystem. Therefore, native cold tolerant Bacillus spp. isolates were used to assess their effect on growth and yield enhancement in amaranth.
Methods
A repository of 120 isolates of Bacillus spp. was developed and evaluated for their cold tolerance ability and PGP (Plant Growth Promoting) traits. Among these isolates, three potential isolates (BS-27, BS-41, BS-58) were selected for the field experiments to assess their effects on plant growth, soil nutrients (N, P and K) availability, nutrient uptake and yield of amaranth. Effect of different treatments on the nutrient use efficiency of amaranth was also determined.
Results
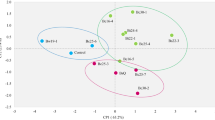
Biopriming of amaranth seeds by the selected three isolates exhibited a significant enhancement in plant growth and yield under field conditions. Among the different treatments, T-8 (BS-27 + BS-58) exhibited a maximum increase in grain yield (36.94%) closely followed by T-4 (BS-27; 33.80%). A significant increase (36.18%, 32.45 and 17.11%) in soil nutrients (NPK) and their uptake (68.01%, 104.16 and 116.46%) was recorded in different treatments. Principal component analysis revealed a positive correlation between soil nutrients and their uptake by host plants. The rpoB gene sequencing and phylogenetic analysis suggested the identity of BS-27 and BS-41 as B. pumilus and BS-58 as B. subtilis.
Conclusion
Our study shows that plant growth promoting bacteria (PGPB) can enhance the nutrient use efficiency of the host plant that further contributes to increase the crop yield. A significant increase in crop yield suggests that regular use of such effective PGPBs by farmers may increase the soil fertility and crop yield. Ultimately, this will lead towards food security under the organic farming framework in general, and sustainable crop production in particular.



Similar content being viewed by others
References
Anderson JM, Ingram JSI (1989) Tropical soil biology and fertility: a handbook of methods. CAB International, Wallingford, p 171
Baetz U, Martinoia E (2014) Root exudates: the hidden part of plant defense. Trends Plant Sci 19:90–98. https://doi.org/10.1016/j.tplants.2013.11.006
Baligar VC, Fageria NK, He ZL (2001) Nutrient use efficiency in plants. Commun Soil Sci Plant Anal 32:921–950. https://doi.org/10.1081/CSS-100104098
Chauhan AK, Maheshwari DK, Kim K, Bajpai VK (2016) Termitarium inhabiting Bacillus endophyticus TSH42 and Bacillus cereus TSH77 colonizing Curcuma longa L.: isolation, characterization and evaluation of their biocontrol and plant growth promoting activities. Can J Microbiol 62:880–892. https://doi.org/10.1139/cjm-2016-0249
Corke H, Cai YZ (2016) Amaranth: overview. In: Wrigley C, Corke H, Seetharaman K, Faubion J (eds) Encyclopedia of food grains. Elsevier, New York, pp 287–296
Das I, Singh AP (2014) Effect of PGPR and organic manures on soil properties of organically cultivated mungbean. Bioscan 9:27–29
Deepa CK, Dastager SG, Pandey A (2010) Plant growth promoting activity in newly isolated Bacillus thioparus (NII-0902) from western ghat forest, India. World J Microbiol Biotechnol 26:2277–2283
Dubey RC, Khare S, Kumar P, Maheshwari DK (2014) Combined effects of chemical fertilizers and rhizosphere-competent Bacillus subtilis BSK17 on yield of Cicer arietinum. Arch Phytopathol Plant Protect 47:2305–2318. https://doi.org/10.1080/03235408.2013.876744
Egamberdieva D, Jabborova D, Berg G (2016) Synergistic interactions between Bradyrhizobium japonicum and the endophyte Stenotrophomonas rhizophila and their effects on growth, and nodulation of soybean under salt stress. Plant Soil 405:35–45. https://doi.org/10.1007/s11104-015-2661-8
Fageria NK, Baligar VC (2005) Enhancing nitrogen use efficiency in crop plants. Adv Agron 88:97–185. https://doi.org/10.1016/S0065-2113(05)88004-6
Glick BR, Patten CL, Holguin G, Penrose DM (1999) Biochemical and genetic mechanism used by plant growth promoting bacteria. Imperial college press, London, UK
Gonita-Mishra I, Sapre S, Kachare S, Tiwari S (2017) Molecular diversity of 1-aminocyclopropane-1-carboxylate (ACC) deaminase producing PGPR from wheat Triticumaestivum L.) rhizosphere. Plant Soil 414:213–227. https://doi.org/10.1007/s11104-016-3119-3
Holt JG, Krieg NR, Sneath PHA, Staley JT, Williams ST (1994) Aerobic chemolithotrophic bacteria and associated organisms. In: Williams & Williams (eds) Bergey’s manual of determinative bacteriology, 9th ed. Baltimore, USA, pp 427–455
Idriss EE, Makarewicz O, Farouk A, Rosner K, Greiner R, Bochow H, Richter T, Borriss R (2002) Extracellular phytase activity of Bacillus amyloliquefaciens FZB45 contributes to its plant growth promoting effect. Microbiology 148:2097–2109
Kibblewhite MG, Ritz K, Swift MJ (2008) Soil health in agricultural systems. Philos Trans R Soc B 363:685–701. https://doi.org/10.1098/rstb.2007.2178
Ko KS, Kim JM, Kim JW, Jung BY, Kim W, Kim IJ, Kook YH (2003) Identification of Bacillus anthracis by rpoB sequence analysis and multiplex PCR. J Clin Microbiol 41:2908–2914. https://doi.org/10.1128/JCM.41.7.2908-2914.2003
Kumar H, Bajpai VK, Dubey RC, Maheshwari DK, Kang SC (2010) Wilt disease management and enhancement of growth and yield of Cajanus cajan (L) var. manak by bacterial combinations amended with chemical fertilizer. Crop Prot 29:591–598. https://doi.org/10.1016/j.cropro.2010.01.002
Kumar P, Dubey RC, Maheshwari DK (2012) Bacillus strains isolated from rhizosphere showed plant growth promoting and antagonistic activity against phytopathogens. Microbiol Res 167:493–499. https://doi.org/10.1016/j.micres.2012.05.002
Margesin R, Neuner G, Storey KB (2007) Cold-loving microbes, plants, and animals fundamental and applied aspects. Nature 94:77–99. https://doi.org/10.1007/s00114-006-0162-6
Masciarelli O, Urbani L, Reinoso H, Luna V (2013) Alternative mechanism for the evaluation of indole-3-acetic acid (IAA) production by Azospirillum brasilense strains and its effects on the germination and growth of maize seedlings. J Microbiol 51:590–597. https://doi.org/10.1007/s12275-013-3136-3
Mlakar SG, Turinek M, Jakop M, Bavec M, Bavec F (2009) Nutritional value and use of grain amaranth: potential future application in bread making. Agricultura 6:43–53
Negi YK, Prabha D, Garg SK, Kumar J (2011) Genetic diversity among cold-tolerant fluorescent Pseudomonas isolates from Indian Himalayas and their characterization for biocontrol and plant growth promoting activities. J Plant Growth Regul 30:128–143. https://doi.org/10.1007/s00344-010-9175-7
Negi YK, Prabha D, Garg SK, Kumar J (2017) Biological control of ragi blast disease by chitinase producing fluorescent Pseudomonas isolates. Org Agr 7:63–71. https://doi.org/10.1007/s13165-015-0142-2
Olsen SR, Cole CV, Watanabe FS, Dean LA (1954) Estimation of available phosphorus in soils by extraction with sodium bicarbonate. US Dep Agric, Washington, DC Circ No 939, pp 1–19
Panhwar QA, Radziah O, Naher UA, Zaharah AR, Sariah M, Mohd Razi I (2012) Root colonization and association of phosphate solubilizing bacteria at various levels of triple supper phosphate in aerobic rice seedlings. Afr J Microbiol Res 6:2277–2286. https://doi.org/10.5897/AJMR11.916
Para-Cota Fannie I, Pena-Cabriales JJ, de los Santos-Villalobos S, Martinez-Gallardo NA, Delano-Frier JP (2014) Burkholderia ambifaria and B. caribensis promote growth and increase yield in grain amaranth (Amaranthus cruentus and A. hypochondriacus) by improving plant nitrogen uptake. PLoS ONE 9:2. https://doi.org/10.1371/journal.pone.0088094
Parewa HP, Yadav J (2014) Response of fertility levels, FYM and bioinoculants on yield attributes, yield and quality of wheat. Agric Sustainable Dev 2:5–10
Parmar JK, Patel JJ (2009) Effect of organic and inorganic nitrogen and biofertilizer on nutrient content and uptake by amaranth (Amaranthus hypochondriacus L.). An Asian J Soil Sci 4:135–138
Perales-Sanchez JX, Reyes-Moreno C, Gomez-Favela MA, Milan-Carrillo J, Cuevas-Rodriguez EO, Valdez-Ortiz A, Gutierrez-Dorado R (2014) Increasing the antioxidant activity, total phenolic and flavonoid contents by optimizing the germination conditions of amaranth seeds. Plant Food Hum Nutr 69:196–202. https://doi.org/10.1007/s11130-014-0430-0
Pierson EA, Weller DM (1994) Use of mixtures of fluorescent pseudomonads to suppress take all and improve the growth of wheat. Phytopathology 84:940–947
Rahman A, Sitepu IR, Tang SY, Hashidoko Y (2010) Salkowski’s reagent test as a primary screening index for functionalities of rhizobacteria isolated from wild dipterocarp saplings growing naturally on medium-strongly acidic tropical peat soil. Biosci Biotechnol Biochem 74:2202–2208
Raju MS, Verma SC, Ramaiah NV (1991) Effect of phosphorus in relation to FYM vs Rhizobium inoculation on nutrient uptake by chickpea cultivars under rainfed condition. Indian J Agric Res 25:43–48
Rokhzadi A, Toashih V (2011) Nutrient uptake and yield of chickpea (Cicer arietinum L.) inoculated with plant growth promoting rhizobacteria. Aust J Crop Sci 5:44–48
Rooney AP, Price NP, Ehrhardt C, Swezey JL, Bannan JD (2009) Phylogeny and molecular taxonomy of the Bacillus subtilis species complex and description of Bacillus subtilis subsp. inaquosorum subsp. nov. Int J Syst Evol Microbiol 59:2429–2436. https://doi.org/10.1099/ijs.0.009126-0
Schwyn B, Neilands JB (1987) Universal chemical assay for detection and determination of siderophores. Anal Biochem 160:47–56. https://doi.org/10.1016/0003-2697(87)90612-9
Shen J, Yuan L, Zhang J, Li H, Bai Z, Chen X, Zhang W, Zhang F (2011) Phosphorus dynamics: from soil to plant. Plant Physiol 156:997–1005. https://doi.org/10.1104/pp.111.175232
Singh PB, Singh V, Nayak PK (2008) Pesticide residues and reproductive dysfunction in different vertebrates from North India. Food Chem Toxicol 46:2533–2539. https://doi.org/10.1016/j.fct.2008.04.009
Talboys PJ, Owen DW, Healey JR, Withers PJA, Jones DL (2014) Auxin secretion by Bacillus amyloliquifaciens FZB42 both stimulates root exudation and limits phosphorus uptake in Triticum aestivium. BMC Plant Biol 14:51
Yadav AN, Sachan SG, Verma P, Tyagi SP, Kaushik R, Saxena AK (2014) Culturable diversity and functional annotation of psychrotrophic bacteria from cold desert of Leh Ladakh (India). World J Microbiol Biotechnol 31:95–108. https://doi.org/10.1007/s11274-014-1768-z
Yanti Y, Astuti FF, Habazar T, Nasution CR (2017) Screening of rhizobacteria from rhizosphere of healthy chili to control bacterial wilt disease and to promote growth and yield of chili. Biodiversitas 18:1–9. https://doi.org/10.13057/biodiv/d180101
Yu X, Liu X, Zhu TH, Liu GH, Mao C (2011) Isolation and characterization of phosphate-solubilizing bacteria from walnut and their effect on growth and phosphorus mobilization. Biol Fertil Soils 47:437–446. https://doi.org/10.1007/s00374-011-0548-2
Acknowledgements
Authors are thankful to Dean, College of Forestry, Ranichauri for providing necessary facilities to carry out this research and to Department of Crop improvement, for providing necessary facilities for field experiments. Support from Genomics team of Pathcare labs, Greater Hyderabad is gratefully acknowledged. Authors are grateful to Dr. Arun Pant, Associate Professor, Dept. of English, SRT Campus (HNB Garhwal University), Badshahithaul, Tehri Garhwal, Uttarakhand for extending his help in language editing of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Stéphane Compant.
Electronic supplementary material
ESM 1
(DOCX 1666 kb)
Rights and permissions
About this article
Cite this article
Pandey, C., Negi, Y.K., Maheshwari, D.K. et al. Potential of native cold tolerant plant growth promoting bacilli to enhance nutrient use efficiency and yield of Amaranthus hypochondriacus. Plant Soil 428, 307–320 (2018). https://doi.org/10.1007/s11104-018-3681-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-018-3681-y