Abstract
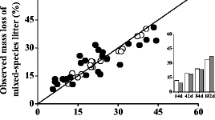
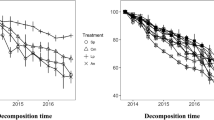
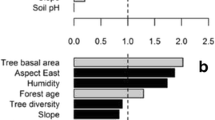
Many studies across a range of ecosystems have shown that decomposition in mixed litter is not predictable from single-species results due to synergistic or antagonistic interactions. Some studies also reveal that species composition and relative abundance may be more important than just richness in driving non-additive effects. Most studies on litter decomposition in Mediterranean maquis, an high-diversity shrubby ecosystem, have dealt exclusively with single species. In this study we investigated, at the individual-litter level, as well as at the litter-mixture level, the effect of litter mixing on decomposition of 3-species litter assemblages with different relative abundance of the component litters; we set up two types of litter assemblages that reflected the heterogeneity of bush cover in the inner maquis and at the edge maquis/gaps, as related to the leaf traits, i.e. sclerophylly vs mesophylly. We measured mass loss, decay of lignin, cellulose and ADSS (acid detergent soluble substances) and fungal mycelium ingrowth. The results show that over a 403-day incubation period, the decomposition of individual litters in mixtures deviated from that of monospecific litters and had different directions. In litter mixtures of the sclerophylls Phillyrea angustifolia and Pistacea lentiscus with the mesophyll Cistus, decomposition was lower than expected (antagonistic effect); in the mixtures of litters with similar physical structure (Ph. angustifolia and P. lentiscus with Quercus ilex) decomposition was faster than expected (synergistic effect). When considering the different decomposition phases, both negative and positive effects occurred in Quercus mixtures depending on the phase of decomposition. In both types of 3-species litter assemblages the greatest effect occurred in uneven mixtures rather than in even mixtures. Our results show that species composition drives the direction whilst the decomposability and the relative abundance drive the magnitude of non-additive effects of litter mixing on decomposition.




Similar content being viewed by others
References
Bardgett RD, Shine A (1999) Linkages between plant litter diversity, soil microbial biomass and ecosystem function in temperate grasslands. Soil Biol Biochem 31:317–321
Berg B, Söderström B (1979) Fungal biomass and nitrogen in decomposing Scots pine needle litter. Soil Biol Biochem 11:339–341
Berg B, McClaugherty C (2008) Plant Litter (Decomposition. Humus formation, Carbon Sequestration), Springer
Blair JM, Parmelee RW, Beare MH (1990) Decay rates, nitrogen fluxes and decomposer communities in single and mixed-species foliar litter. Ecology 71:1976–1985
Bonanomi G, Incerti G, Antignani V, Capodilupo M, Mazzoleni S (2010) Decomposition and nutrient dynamics in mixed litter of Mediterranean species. Plant Soil 331:481–496
Canhoto C, Barlocher F, Graca MAS (2002) The effects of Eucalyptus globulus oils on fungal enzymatic activity. Arch Hydrobiol 154:121–132
Chapman SK, Koch GW (2007) What type of diversity yields synergy during mixed litter decomposition in a natural forest ecosystem? Plant Soil 299:153–162
Chapman SK, Newman GS (2010) Biodiversity at the plant-soil interface: microbial abundance and community structure respond to litter mixing. Oecologia 162:763–769
Cornelissen JHC, Lavorel S, Garnier E et al (2003) A handbook of protocols for standardised and easy measurements of plant functional traits worldwide. Aust J Bot 51:335–380
Cox P, Wilkinson SP, Anderson JM (2001) Effects of fungal inocula on the decomposition of lignin and structural polysaccharides in Pinus sylvestris litter. Biol Fertil Soils 33:246–251
de Graaff MA, Classen AT, Castro HF, Schadt CW (2010) Labile soil carbon inputs mediate the soil microbial community composition and plant residue decomposition rates. New Phytol 188:1055–1064
De Marco A, Forte A, Gentile AE, Virzo De Santo A (2004) Elemental composition and litter decomposition of Phillyrea angustifolia L. at burned and unburned sites. In: Arianoutsou M, Papanastasis VP (eds) Ecology, Conservation and Management of Mediterranean Climate Ecosystems. Proceedings 10th MEDECOS Conference, Rodi (Grecia), 25 Aprile-1 Maggio 2004. Mill, Rotterdam. ISBN 90 5966 016 1
De Marco A, Meola A, Esposito F, Virzo De Santo A (2008) Productivity and modifications of ecosystem processes in gaps of a low Macchia in southern Italy. Web Ecology 8: 55-66
FAO (1998) World reference base for soil resources—World Soil Res 84 Rome
Fioretto A, Papa S, Fuggi A (2003) Litter-fall and litter decomposition in a low Mediterranean shrubland. Biol Fertil Soils 39:37–44
Fioretto A, Di Nardo C, Papa S, Fuggi A (2005) Lignin and cellulose degradation and nitrogen dynamics during decomposition of three leaf litter species in a Mediterranean ecosystem. Soil Biol Biochem 37:1083–1091
Gallardo A, Merino J (1993) Leaf decomposition in two Mediterranean ecosystems of southwest Spain. Influence of substrate quality. Ecology 74:152–161
Gartner TB, Cardon ZG (2004) Decomposition dynamics in mixed-species leaf litter a review. Oikos 104:230–246
Gessner MO, Swan CM, Dang CK, McKie BG, Bardgett RD, Wall DH, Hättenschwiler S (2010) Diversity meets decomposition. Trends Ecol Evol 25:372–380
Harrison AF (1971) The inhibitory effect of oak leaf litter tannins on the growth of fungi, in relation to litter decomposition. Soil Biol Biochem 3:167–172
Hättenschwiler S, Tiunov AV, Scheu S (2005) Biodiversity and litter decomposition in terrestrial ecosystems. Ann Rev Ecol Evol Syst 36:191–218
Hector A, Beale AJ, Minns A, Otway SJ, Lawton JH (2000) Consequences of the reduction of plant diversity for litter decomposition: effects through litter quality and microenvironment. Oikos 90:357–371
Kazakou E, Violle C, Roumet C, Pintor C, Gimenez O, Garnier E (2009) Litter quality and decomposability of species from a Mediterranean succession depend on leaf traits but not on nitrogen supply. Ann Bot 104:1151–1161
Kirk TK, Connors WJ, Zeikus JG (1976) Requirement for a growth substrate during lignin decomposition by two wood-rotting fungi. Appl Environ Microb 32:192–194
Magiatis P, Melliou E, Skaltsounis AL, Chinou IB, Mitaku S (1999) Chemical composition and antimicrobial activity of the essential oils of Pistacia lentiscus var. chia. Planta Med 65:749–752
Maisto G, De Marco A, Meola A, Sessa L, Virzo De Santo A (2011) Nutrient dynamics in litter mixtures of four Mediterranean maquis species decomposing in situ. Soil Bio Biochem 43:520–530
McArthur JW, Aho JM, Rader RB et al (1994) Interspecific leaf interactions during decomposition in aquatic and floodplain ecosystems. J N Am Benthol Soc 13:57–67
McTiernan KB, Ineson P, Coward PA (1997) Respiration and nutrient release from tree leaf litter mixtures. Oikos 78:527–538
Nilsson MC, Gallet C, Wallstedt A (1998) Temporal variability of phenolics and batasin-III in Empetrum hermaphroditum leaves over an eight-year period: interpretation of ecological function. Oikos 81:6–16
Olson FCW (1950) Quantitative estimates of filamenthous algae. Trans Am Microsc Soc 69:272–279
Osono T (2007) Ecology of ligninolytic fungi associated with leaf litter decomposition. Ecol Res 22:955–974
Rayner ADM, Webber JF (1984) Interspecific mycelial interactions—an overview. In: Jennings DH, Rayner ADM (eds) The ecology and physiology of fungal mycelium. Cambridge University Press, Cambridge, pp 383–417
Rotheray TD, Boddy L, Jones TH (2009) Collembola foraging responses to interacting fungi. Ecol Entomol 34:125–132
Smith VC, Bradford MA (2003) Do non-additive effects on decomposition in litter-mix experiments result from differences in resource quality between litters? Oikos 102:235–242
Söderström BE (1977) Vital staining of fungi in pure cultures and in soil with fluorescein diacetate. Soil Biol Biochem 9:59–63
Sundman V, Sivela S (1978) A comment on the membrane filter technique for estimation of length of fungal hyphae in soil. Soil Biol Biochem 10:399–401
Swan CM, Gluth MA, Horne CL (2009) Leaf litter species evenness influences non-additive breakdown in a headwater stream. Ecology 90:1650–1658
Van Soest PJ, Wine RH (1968) Determination of lignin and cellulose in acid–detergent fibre with permanganate. J Assoc Off Agr Chem 51:780–785
Zimmer M (2002) Is decomposition of woodland leaf litter influenced by its species richness? Soil Biol Biochem 34:277–284
Ward SE, Ostle NJ, McNamara NP, Bardgett RD (2010) Litter eveness influences short-term peatland decomposition processes. Oecologia 164:511–520
Wardle DA, Bonner KI, Nicholson KS (1997) Biodiversity and plant litter: experimental evidence which does not support the view that enhanced species richness improves ecosystem function. Oikos 79:247–258
Acknowledgments
We are grateful to the Forest Service of the Castel Volturno Nature Reserve for logistic support and Dr. N. Di Fusco for allowing us to use the field site. We thank Dr. C. Arena for measurement of leaf traits. This study was supported by MIUR - PRIN 2005. Two anonymous reviewers are acknowledged for positive criticism.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Hans Lambers.
Rights and permissions
About this article
Cite this article
De Marco, A., Meola, A., Maisto, G. et al. Non-additive effects of litter mixtures on decomposition of leaf litters in a Mediterranean maquis. Plant Soil 344, 305–317 (2011). https://doi.org/10.1007/s11104-011-0748-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-011-0748-4