Abstract
Purpose
The purpose of this study was to develop an amorphous solid dispersion (SD) of an extremely water-insoluble and very weakly basic drug, itraconazole (ITZ), by interaction with weak organic acids and then drying that would enhance dissolution rate of drug and physical stability of formulation.
Methods
Aqueous solubility of ITZ in concentrated solutions of weak organic acids, such as glutaric, tartaric, malic and citric acid, was determined. Solutions with high drug solubility were dried using vacuum oven and the resulting SDs having 2 to 20% drug load were characterized by differential scanning calorimetry (DSC), powder X-ray diffractometry (PXRD) and attenuated total reflectance-Fourier transform infrared (ATR-FTIR) spectroscopy. The dissolution of SDs was initially studied in 250 mL of 0.1 N HCl (pH 1.1), and any undissolved solids were collected and analyzed by PXRD. The pH of the dissolution medium was then changed from 1.1 to 5.5, particle size of precipitates were measured, and drug concentrations in solution were determined by filtration through membrane filters of varying pore sizes.
Results
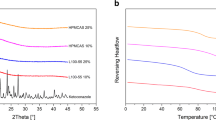
The aqueous solubility of ITZ was greatly enhanced in presence of weak acids. While the solubility of ITZ in water was ~4 ng/ mL, it increased to 25–40 mg per g of solution at 25°C and 200 mg per g of solution at 65°C at a high acid concentration leading to extremely high solubilization. PXRD of SDs indicated that ITZ was present in the amorphous form, wherein the acid formed a partially crystalline matrix. ATR-FTIR results showed possible weak interactions, such as hydrogen bonding, between drug and acid but there was no salt formation. SDs formed highly supersaturated solutions at pH 1.1 and had superior dissolution rate as compared to amorphous drug and physical mixtures of drug and acids. Following the change in pH from 1.1 to 5.5, ITZ precipitated as mostly nanoparticles, providing high surface area for relatively rapid redissolution.
Conclusions
A method of highly solubilizing an extremely water-insoluble drug, ITZ, in aqueous media and converting it into an amorphous form in a physically stable SD was successfully investigated. The dissolution rate and the extent of supersaturation of the drug in dissolution media improved greatly, and any precipitate formed at high pH had very small particle size.







Similar content being viewed by others
Abbreviations
- ATR-FTIR:
-
Attenuated total reflectance-Fourier transform infrared spectroscopy
- DSC:
-
Differential scanning calorimetry
- HPMC:
-
Hydroxypropylmethyl cellulose
- HPMCAS:
-
Hydroxypropylmethyl cellulose acetate succinate
- ITZ:
-
Itraconazole
- PXRD:
-
Powder X-ray diffractometry
- SD:
-
Solid dispersion
- TGA:
-
Thermogravimetric analysis
References
Lipinski CA. Drug-like properties and the causes of poor solubility and poor permeability. J Pharmacol Toxicol Methods. 2000;44(1):235–49.
Huang LF, Tong WQ. Impact of solid state properties on developability assessment of drug candidates. Adv Drug Deliv Rev. 2004;56(3):321–34.
Selen A, Dickinson PA, Müllertz A, Crison JR, Mistry HB, Cruañes MT, et al. The biopharmaceutics risk assessment roadmap for optimizing clinical drug product performance. J Pharm Sci. 2014;103(11):3377–97.
Janssens S, De Zeure A, Paudel A, Van Humbeeck J, Rombaut P, Van den Mooter G. Influence of preparation methods on solid state supersaturation of amorphous SD: a case study with itraconazole and Eudragit E 100. Pharm Res. 2010;27:775–85.
Gilis P, De Conde V, Vandecruys R. Beads having a core coated with an antifungal and a polymer. Patent no. WO-94/05263 (A1). 1994.
O’Neil M.J. (ed.). The Merck index - an encyclopedia of chemicals, drugs, and biologicals. Whitehouse station, NJ: Merck and Co., Inc., 2006., p. 907
Brewster M, Neekens P, Peeters J. Solubilization of itraconazole as a function of cyclodextrin structural space. J Incl Phenom Macrocycl Chem. 2007;57:561–6.
Sporanox capsule product information. https://www.janssen.com.au/files/Products/SPORANOX_CAPSULES_PI.pdf (last accessed: 20 July 2015)
Beule K. Itraconazole: pharmacology, clinical experience and future development. Int J Antimicrob Agents. 1996;6:175–81.
Sporanox oral solution product information. https://www.janssen.com.au/files/Products/SPORANOX_SOLUTION_PI.pdf (last accessed: 20 July 2015)
Tao T, Zhao Y, Wu J, Zhou B. Preparation and evaluation of itraconazole dihydrochloride for the solubility and dissolution rate enhancement. Int J Pharm. 2009;367:109–14.
Namburi R, Kerr J. Oral itraconazole formulations and methods of making the same. Patent no. US 6,663,897 B2. 2003.
Remenar J, Morissette S, Peterson M, Moulton B, MacPhee JM, Guzmán HR, et al. Crystal engineering of novel cocrystals of a triazole drug with 1,4- dicarboxylic acids. J Am Chem Soc. 2003;125:8456–7.
Wei S, Mao S, Shi Y, Li L, Fang L. Nanoization of itraconazole by high pressure homogenization: stabilizer optimization and effect of particle size on oral absorption. J Pharm Sci. 2011;24:297–303.
Mou D, Chen H, Wan J, Xu H, Yang X. Potent dried drug nanosuspensions for oral bioavailability enhancement of poorly soluble drugs with pH-dependent solubility. Int J Pharm. 2011;413:237–44.
Vasanthavada M, Tong WQ, Serajuddin ATM. Development of solid dispersions for poorly water-soluble drug. In: Liu R, editor. Water-insoluble drug formulations. 2nd ed. New York: Informa healthcare; 2008. p. 149–84.
Janssens S, de Armas HN, D’ Autry W, Van Schepdael A, Van den Mooter G. Characterization of ternary solid dispersions of itraconazole in polyethylene glycol 6000/ polyvidone- vinylacetate 64 blends. Eur J Pharm Biopharm. 2008;69:1114–20.
Baert L, Peeters J, Verreck G. Solid mixtures of cyclodextrins prepared via melt extrusion. Patent no. US 6,365,188 B1, 2002.
Mellaerts R, Mole R, Jammaer J, Aerts CA, Paudel A, Van Humbeeck J, et al. Increasing the oral bioavailability of the poorly water soluble drug itraconazole with ordered mesoporous silica. Eur J Pharm Biopharm. 2008;69:223–30.
Miller D, DiNunzio J, Yang W, McGinity J, Williams R. Enhanced in vivo absorption of itraconazole via stabilization of supersaturation following acidic to neutral pH transition. Drug Dev Ind Pharm. 2008;34:890–902.
Bhardwaj S, Arora K, Kwong E, Templeton A, Clas S, Suryanarayanan R. Correlation between molecular mobility and physical stability of amorphous itraconazole. Mol Pharm. 2013;10:694–700.
Sarode A, Sandhu H, Shah N, Malick W, Zia H. Hot melt extrusion for amorphous solid dispersions: temperature and moisture activated drug – polymer interactions for enhanced stability. Mol Pharm. 2013;10:3665–75.
Holvoet C, Vander Heyden Y, Plaizier-Vercammen J. Influence of preparation method on itraconazole oral solutions using cyclodextrins as complexing agents. Die Pharmazie. 2007;62(7):510–4.
Singh S, Parikh T, Sandhu HK, Shah NH, Malick AW, Singhal D, et al. Supersolubilization and amorphization of a model basic drug, haloperidol, by interaction with weak acids. Pharm Res. 2013;30(6):1561–73.
Li S, Wong S, Sethia S, Almoazen H, Joshi YM, Serajuddin ATM. Investigation of solubility and dissolution of a free base and two different salt forms as a function of pH. Pharm Res. 2005;22(4):628–35.
Li S, Doyle P, Metz S, Royce AE, Serajuddin ATM. Effect of chloride ion on dissolution of different salt forms of haloperidol, a model basic drug. J Pharm Sci. 2005;94(10):2224–31.
Shah A, Serajuddin ATM. Supersolubilization by using nonsalt-forming acid–base interaction. In: Shah N, Sandhu H, Choi D, Chokshi H, Malick AW, editors. Amorphous solid dispersions theory and practice. New York: Springer-Verlag; 2014. p. 595–611.
Shah A, Serajuddin ATM. Conversion of solid dispersion prepared by acid–base interaction into free-flowing and tabletable powder by using Neusilin® US2. Int J Pharm. 2015;484(1–2):172–80.
Serajuddin ATM, Pudipeddi M. Salt selection strategies. Handbook of pharmaceutical salts - properties, selection and use. Switzerland: Verlag Helvetica Chemica Acta/Wiley–VCH; 2002. p. 158–9.
Serajuddin ATM. Salt formation to improve drug solubility. Adv Drug Del Rev. 2007;59(7):603–16.
Desiraju GR. Crystal and co-crystal. Cryst Eng Comm. 2003;5:466–7.
Li ZJ, Abramov Y, Bordner J, Leonard J, Medek A, Trask AV. Solid-state acid–base interactions in complexes of heterocyclic bases with dicarboxylic acids: crystallography, hydrogen bond analysis, and 15 N NMR spectroscopy. J Am Chem Soc. 2006;128(25):8199–210.
Vishweshwar P, Nangia A, Lynch VM. Molecular complexes of homologous alkanedicarboxylic acids with isonicotinamide: X-ray crystal structures, hydrogen bond synthons, and melting point alternation. Cryst Growth Des. 2003;3:783–90.
Walsh RDB, Bradner MW, Fleischman S, Morales LA, Moulton B, Rodriguez-Hornedo N, et al. Crystal engineering of the composition of pharmaceutical phases. Chem Commun. 2003;2:186–7.
Bis JA, McLaughlin OL, Vishweshwar P, Zaworotko MJ. Supramolecular heterocatemers and their role in cocrystal design. Cryst Growth Des. 2006;6:2648–50.
Etter MC. Encoding and decoding hydrogen bonds patterns of organic compounds. Acc Chem Res. 1990;23:120–6.
Bis JA, Vishweshwar P, Weyna D, Zaworotko MJ. Hierarchy of supramolecular synthons: persistent hydroxyl…pyridine hydrogen bonds in cocrystals that contain a cyano acceptor. Mol Pharm. 2007;4(3):401–16.
Shattock T, Arora K, Vishweshwar P, Zaworotko M. Hierarchy of supramolecular synthons: persistent carboxylic acid-pyridine hydrogen bonds in cocrystals that also contain hydroxyl moiety. Cryst Growth Des. 2008;8(12):4533–45.
Qiao N, Li M, Schlindwein W, Malek N, Davies A, Trappitt G. Pharmaceutical cocrystal: an overview. Int J Pharm. 2011;419:1–11.
Izutsu KI, Kadoya S, Yomota C, Kawanishi T, Yonemochi E, Terada K. Freeze-drying of proteins in glass solids formed by basic amino acids and dicarboxylic acids. Chem Pharm Bull. 2009;57(1):43–8.
Jensen KT, Löbmann K, Rades T, Grohganz H. Improving co-amorphous drug formulations by the addition of the highly water soluble amino acid proline. Pharmaceutics. 2014;6(3):416–35.
Lobmann K, Laitinen R, Grohganz H, Strachan C, Rades T, Gordon K. Coamorphous drug systems: enhanced physical stability and dissolution rate of indomethacin and naproxen. Mol Pharm. 2011;8(5):1919–28.
Morrision RT, Boyd RN. Organic chemistry. 6th ed. Michigan: Prentice Hall International; 1992.
Ober C, Gupta R. Formation of itraconazole-succinic acid cocrystals by gas antisolvent cocrystallization. AAPS PharmSciTech. 2012;13(4):1396–406.
Kashchiev D, Van Rosmalen G. Nucleation in solutions revisited. Cryst Res Technol. 2003;38:555–74.
Gebauer D, Coelfen H. Prenucleation clusters and non-classical nucleation. Nano Today. 2011;6:564–84.
Vekilov PG. Nucleation. Cryst Growth Des. 2010;10:5007–19.
Good D, Rodriguez-Hornedo N. Solubility advantage of pharmaceutical cocrystals. Cryst Growth Des. 2009;9:2252–64.
Thakuria R, Delori A, Jones W, Lipert M, Roy L, Rodriguez N. Pharmaceutical cocrystals and poorly soluble drugs. Int J Pharm. 2013;453(1):101–25.
Shevchenko A, Bimbo LM, Miroshnyk I, Haarala J, Jelínková K, Syrjänen K, et al. A new cocrystal and salts of itraconazole: comparison of solid-state properties, stability and dissolution behavior. Int J Pharm. 2012;436(1):403–9.
Hancock BC, Zografi G. The relationship between the glass transition temperature and the water content of amorphous pharmaceutical solids. Pharm Res. 1994;11:471–7.
Andronis V, Zografi G. Crystal nucleation and growth of indomethacin polymorphs from the amorphous state. J Non-cryst Sol. 2000;271:236–48.
Six K, Verreck G, Peeters J, Binnemans K, Berghmans H, Augustijns P, et al. Investigation of thermal properties of glassy itraconazole: identification of a monotropic mesophase. Thermochim Acta. 2001;376(2):175–81.
Parikh T, Gupta SS, Meena AK, Vitez I, Mahajan N, Serajuddin ATM. Application of film casting technique to investigate drug–polymer miscibility in solid dispersion and hot melt extrudate. J Pharm Sci. 2015;104:2142–52.
Stevenson CL, Bennett DB, Lechuga‐Ballesteros D. Pharmaceutical liquid crystals: the relevance of partially ordered systems. J Pharm Sci. 2005;94(9):1861–80.
Murdande S, Pikal M, Shanker R, Bogner R. Solubility advantage of amorphous pharmaceuticals, part 3: is maximum solubility advantage experimentally attainable and sustainable? J Pharm Sci. 2011;100(10):4349–56.
ACKNOWLEDGMENTS AND DISCLOSURES
The authors would like to acknowledge the support of chemistry department at St. John’s University for providing the equipment to conduct FTIR studies.
Disclaimer
The views and opinions expressed in this article are those of the authors and do not reflect the official policy or position of Food and Drug Administration or any other agency of the U.S. government.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Parikh, T., Sandhu, H.K., Talele, T.T. et al. Characterization of Solid Dispersion of Itraconazole Prepared by Solubilization in Concentrated Aqueous Solutions of Weak Organic Acids and Drying. Pharm Res 33, 1456–1471 (2016). https://doi.org/10.1007/s11095-016-1890-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-016-1890-8