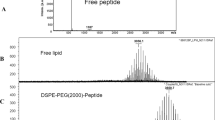
ABSTRACT
Purpose
Both combretastatin A-4 (CA-4) and doxorubicin (DOX) was loaded in different form in a targeted nanomedicine in order to achieve the active delivery of these two drugs followed by sequentially suppressing tumor vasculature and tumor cells.
Methods
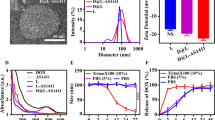
Octreotide-modified stealth liposomes loaded with CA-4 and DOX (Oct-L[CD]) were prepared and characterized. Then in vitro release, cellular uptake, in vitro antitumor effect, pharmacokinetics, in vivo sequential killing effect, in vivo antitumor efficacy against somatostatin receptor (SSTR) positive cells, as well as the action mechanism of such system, were studied.
Results
A rapid release of CA-4 followed by a slow release of DOX was observed in vitro. The active targeted liposomes Oct-L[CD] showed a specific cellular uptake through ligand-receptor interaction and a higher antitumor effect in vitro against SSTR-positive cell line. The in vivo sequential killing effect of such system was found as evidenced by the fast inhibition of blood vessels and slow apoptosis-inducing of tumor cells. Oct-L[CD] also exhibited the strongest antitumor effect in MCF-7 subcutaneous xenograft models.
Conclusions
Oct-modified co-delivery system may have great potential as an effective carrier for cancer therapy.










Similar content being viewed by others
Abbreviations
- AUC:
-
area under the plasma concentration-time curve
- CA-4:
-
combretastatin A-4
- CA-4P:
-
combretastatin A-4 phosphate
- DDS:
-
drug delivery system
- DOX:
-
doxorubicin
- DSPE-PEG (PEG Mw 2000):
-
1.2-distearoyl-sn-glycero-3-phosphoethanola mine-N-[poly(ethylene-glycol)]
- DSPE-PEG-Oct:
-
conjugate of octreotide with DSPE-PEG
- EPC:
-
egg phosphatidylcholine
- FBS:
-
fetal bovine serum
- IC50 :
-
50% inhibitory concentration
- K :
-
elimination rate constant
- L[C]:
-
liposomes encapsulating CA-4
- L[CD]:
-
liposomes encapsulating both CA-4 and DOX
- L[D]:
-
liposomes encapsulating DOX
- Oct:
-
octreotide
- Oct-L[CD]:
-
octreotide-targeted liposomes encapsulating both CA-4 and DOX
- PBS:
-
phosphate buffered saline
- PD:
-
pharmacodynamics
- PDI:
-
polydispersity index
- PK:
-
pharmacokinetics
- SRB:
-
sulforhodamine B
- SSTR2:
-
somatostatin receptor subtype 2
- SSTRs:
-
somatostatin receptors
- T1/2 :
-
plasma half-life
- TEM:
-
transmission electron microscope
- VDAs:
-
vascular disrupting agents
- VEGF:
-
vascular endothelial growth factor
REFERENCES
Lisa BP, James OB. Nanoparticle and targeted systems for cancer therapy. Adv Drug Deliv Rev. 2004;56:1649–59.
Zhang L, Radovic-Moreno AF, Alexis F, Gu FX, Basto PA, Bagalkot V, Jon S, Langer RS, Farokhzad OC. Co-delivery of hydrophobic and hydrophilic drugs from nanoparticle-aptamer bioconjugates. Chem Med Chem. 2007;2:1268–71.
Mitragotris S. Synergistic effect of enhancers for transdermal drug delivery. Pharm Res. 2000;17:1354–59.
Folkman J. Role of angiogenesis in tumor growth and metastasis. Semin Oncol. 2002;29:15–8.
Siemann DW, Chaplin DJ, Horsman MR. Vascular-targeting therapies for treatment of malignant disease. Cancer. 2004;100:2491–97.
Thorpe PE. Vascular targeting agents as cancer therapeutics. Clin Cancer Res. 2004;10:415–27.
Dziba JM, Marcinek R, Venkataraman G, Robinson JA, Ain KB. Combretastatin A4 phosphate has primary antineoplastic activity against human anaplastic thyroid carcinoma cell lines and xenograft tumors. Thyroid. 2002;12:1063–70.
Nelkin BD, Ball DW. Combretastatin A-4 and doxorubicin combination treatment is effective in a preclinical model of human medullary thyroid carcinoma. Oncol Rep. 2001;8:157–60.
Yeung SC, She M, Yang H, Pan J, Sun L, Chaplin D. Combination chemotherapy including combretastatin A4 phosphate and paclitaxel is effective against anaplastic thyroid cancer in a nude mouse xenograft model. J Clin Endocrinol Metab. 2007;92:2902–09.
Verheul HMW, Voest EE, Schlingemann RO. Are tumours angiogenesis-depent? J Pathol. 2004;202:5–13.
Lee SM, Woll PJ, Rudd R, Ferry D, O’Brien M, Middleton G, Spiro S, James L, Ali K, Jitlal M, Hackshaw A. Anti-angiogenic therapy using thalidomide combined with chemotherapy in small cell lung cancer: a randomized, double-blind, placebo-controlled trial. J Natl Cancer Inst. 2009;101:1049–57.
Sapra P, Tyagi P, Allen TM. Ligand-targeted liposomes for cancer treatment. Curr Drug Deliv. 2005;2:368–81.
Bauer W, Briner U, Doepfner W, Haller R, Huguenin R, Marbach P, Petcher TJ, Pless J. SMS 201-995: a very potent and selective octapeptide analogue of somatostatin with prolonged action. Life Sci. 1982;31:1133–40.
Reubi JC, Kvols L, Krenning E, Lamberts SW. Distribution of somatotatin receptors in normal and tumor tissue. Metabolism. 1990;39:78–81.
Volante M, Rosas R, Allia E, Granata R, Baragli A, Muccioli G, Papotti M. Somatostatin, cortistatin and their receptors in tumours. Mol Cell Endocrinol. 2008;286:219–29.
Wild D, Schmitt JS, Ginj M, Mäcke HR, Bernard BF, Krenning E, De Jong M, Wenger S, Reubi JC. DOTA-NOC, a high-affinity ligand of somatostatin receptor subtypes 2, 3 and 5 for labelling with various radiometals. Eur J Nucl Med Mol Imaging. 2003;30:1338–47.
Ginj M, Schmitt JS, Chen J, Waser B, Reubi JC, de Jong M, Schulz S, Maecke HR. Design, synthesis, and biological evaluation of somatostatin-based radiopeptides. Chem Biol. 2006;13:1081–90.
Shen H, Hu D, Du J, Wang X, Liu Y, Wang Y, Wei JM, Ma D, Wang P, Li L. Paclitaxel–octreotide conjugates in tumor growth inhibition of A549 human non-small cell lung cancer xenografted into nude mice. Eur J Pharmacol. 2008;601:23–9.
Lasfer M, Vadrot N, Schally AV, Nagy A, Halmos G, Pessayre D, Feldmann G, Reyl-Desmars FJ. Potent induction of apoptosis in human hepatoma cell lines by targeted cytotoxic somatostatin analogue AN-238. J Hepatol. 2005;42:230–37.
Moody TW, Fuselier J, Coy DH, Mantey S, Pradhan T, Nakagawa T, Jensen RT. Camptothecin-somatostatin conjugates inhibit the growth of small cell lung cancer cells. Peptides. 2005;26:1560–66.
Zhang YF, Wang JC, Bian DY, Zhang X, Zhang Q. Targeted delivery of RGD-modified liposomes encapsulating both combretastatin A-4 and doxorubicin for tumor therapy: In vitro and in vivo studies. Eur J Pharm Biopharm. 2010;74:467–73.
Sengupta S, Eavarone D, Capila I, Zhao G, Watson N, Kiziltepe T, Sasisekharan R. Temporal targeting of tumour cells and neovasculature with a nanoscale delivery system. Nature. 2005;436:568–72.
Vichai V, Kirtikara K. Sulforhodamine B colorimetric assay for cytotoxicity screening. Nat Protoc. 2006;1:1112–16.
Zhang J, ** W, Wang X, Wang J, Zhang X, Zhang Q. A novel octreotide modified lipid vesicle improved the anticancer efficacy of doxorubicin in somatostatin receptor 2 positive tumor models. Mol Pharm. 2010;7:1159–68.
Zhang Y, Wang X, Wang J, Zhang X, Zhang Q. Octreotide-modified polymeric micelles as potential carriers for targeted docetaxel delivery to somatostatin receptor overexpressing tumor cells. Pharm Res. 2011;28:1167–78.
Sun M, Wang Y, Shen J, **ao Y, Su Z, ** Q. Octreotide-modification enhances the delivery and targeting of doxorubicin-loaded liposomes to somatostatin receptors expressing tumor in vitro and in vivo. Nanotechnology. 2010;21:475101–12.
Fritze A, Hens F, Kimpfler A, Schubert R, Peschka-Süss R. Remote loading of doxorubicin into nanoliposomes driven by a transmembrane phosphate gradient. Biochim Biophys Acta. 2006;1758:1633–40.
Gu F, Zhang L, Teply BA, Mann N, Wang A, Radovic-Moreno AF, Langer R, Farokhzad OC. Precise engineering of targeted nanoparticles by using self-assembled biointegrated block copolymers. Proc Natl Acad Sci USA. 2008;105:2586–91.
Cairo CW, Gestwicki JE, Kanai M, Kiessling LL. Control of multivalent interactions by binding epitope density. J Am Chem Soc. 2002;124:1615–19.
Jule E, Nagasaki Y, Kataoka K. Lactose-installed poly(ethylene glycol)-poly(d, l-lactide) block copolymer micelles exhibit fast-rate binding and high affinity toward a protein bed simulating a cell surface. A surface plasmon resonance study. Bioconjug Chem. 2003;14:177–86.
Emanuel N, Kedar E, Bolotin E, Smorodinsky NI, Barenholz Y. Targeted delivery of doxorubicin via sterically stabilized immunoliposomes: pharmacokinetics and biodistribution in tumor-bearing mice. Pharm Res. 1996;13:861–68.
Turka MJ, Watersb DJ, Low PS. Folate-conjugated liposomes preferentially target macrophages associated with ovarian carcinoma. Cancer Lett. 2004;213:165–72.
Linuma H, Maruyama K, Okinaga K. Intracellular targeting therapy of cisplatin-encapsulated transferrin-polyethylene glycol liposomes on peritoneal dissemination of gastric cancer. Int J Cancer. 2002;299:130–37.
Mayer LD, Bally MB, Cullis PR, Wilson SL, Emerman JT. Comparison of free and liposome encapsulated doxorubicin tumor drug uptake and antitumor efficacy in the SC115 murine. Cancer Lett. 1990;53(2–3):183–90.
Lu WL, Qi XR, Zhang Q, Li RY, Wang GL, Zhang RJ, Wei SL. A pegylated liposomal platform: pharmacokinetics, pharmacodynamics, and toxicity in mice using doxorubicin as a model drug. J Pharmacol Sci. 2004;95(3):381–9.
Gabizon AA. Pegylated liposomal doxorubicin: metamorphosis of an old drug into a new form of chemotherapy. Cancer Invest. 2001;19(4):424–36.
ACKNOWLEDGMENTS AND DISCLOSURES
This study was supported by the National Nature Science Foundation (No.81130059), National Basic Research Program of China (No. 2009CB930300).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dai, W., **, W., Zhang, J. et al. Spatiotemporally Controlled Co-delivery of Anti-vasculature Agent and Cytotoxic Drug by Octreotide-Modified Stealth Liposomes. Pharm Res 29, 2902–2911 (2012). https://doi.org/10.1007/s11095-012-0797-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-012-0797-2