ABSTRACT
Purpose
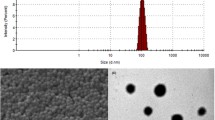
To develop a novel brain drug delivery system based on self-assembled poly(ethyleneglycol)-poly (D,L-lactic-co-glycolic acid) (PEG-PLGA) polymersomes conjugated with lactoferrin (Lf-POS). The brain delivery properties of Lf-POS were investigated and optimized.
Method
Three formulations of Lf-POS, with different densities of lactoferrin on the surface of polymersomes, were prepared and characterized. The brain delivery properties in mice were investigated using 6-coumarin as a fluorescent probe loaded in Lf-POS (6-coumarin-Lf-POS). A neuroprotective peptide, S14G-humanin, was incorporated into Lf-POS (SHN-Lf-POS); a protective effect on the hippocampuses of rats treated by Amyloid-β25-35 was investigated by immunohistochemical analysis.
Results
The results of brain delivery in mice demonstrated that the optimized number of lactoferrin conjugated per polymersome was 101. This obtains the greatest blood–brain barrier (BBB) permeability surface area(PS) product and percentage of injected dose per gram brain (%ID/g brain). Immunohistochemistry revealed the SHN-Lf-POS had a protective effect on neurons of rats by attenuating the expression of Bax and caspase-3 positive cells. Meanwhile, the activity of choline acetyltransferase (ChAT) had been increased compared with negative controls.
Conclusion
These results suggest that lactoferrin functionalized self-assembled PEG-PLGA polymersomes could be a promising brain-targeting peptide drug delivery system via intravenous administration.








Similar content being viewed by others
REFERENCES
Huynh GH, Deen DF, Szoka Jr FC. Barriers to carrier mediated drug and gene delivery to brain tumors. J Control Release. 2006;110:236–59.
Cornford EM, Cornford ME. New systems for delivery of drugs to the brain in neurological disease. Lancet Neurol. 2002;1:306–15.
Pardridge WM. Drug and gene targeting to the brain via blood–brain barrier receptor-mediated transport systems. International Congress Series. 2005;1277:49–62.
Smith QR. Carrier-mediated transport to enhance drug delivery to brain. International Congress Series. 2005;1277:63–74.
Spencer BJ, Verma IM. Targeted delivery of proteins across the blood–brain barrier. PNAS. 2007;104:7594–9.
Witt KA, Davis TP. CNS drug delivery: opioid peptides and the blood-brain barrier. AAPS J. 2006;8:76–88.
Visser CC, Voorwinden H, Crommelin DJA, Danhof M, Boer AG. Characterization and modulation of the transferrin receptor on brain capillary endothelial cells. Pharm Res. 2004;21:761–9.
Sarzehi S, Chamani J. Investigation on the interaction between tamoxifen and human holo-transferrin: determination of the binding mechanism by fluorescence quenching, resonance light scattering and circular dichroism methods. Int J Biol Macromol. 2010;47:558–69.
Chamani J, Vahedian-Movahed H, Saberi MR. Lomefloxacin promotes the interaction between human serum albumin and transferrin: a mechanistic insight into the emergence of the antibiotic’s side effects. J Pharm Biomed Anal. 2011;55:114–24.
Boado RJ, Zhang YF, Zhang Y, Pardridge WM. Humanization of anti-human insulin receptor antibody for drug targeting across the human blood-brain barrier. Biotechnol Bioeng. 2007;96:381–91.
Suzuki YA, Lopez V, Lonnerdal B. Mammalian lactoferrin receptors: structure and function. Cell Mol Life Sci. 2005;22:2560–75.
Ji B, Maeda J, Makoto H, Higuchi M, Inoue K, Akita H, et al. Pharmacokinetics and brain uptake of lactoferrin in rats. Life Sci. 2006;78:851–5.
Kabanov AV, Gendelman HE. Nanomedicine in the diagnosis and therapy of neurodegenerative disorders. Prog Polym Sci. 2007;32:1054–82.
Modi G, Pillay V, Choonara YE, Ndesendo VM, du Toit LC, Naidoo D. Nanotechnological applications for the treatment of neurodegenerative disorders. Prog Neurobiol. 2009;88:272–85.
Pardridge WM. Molecular Trojan horses for blood–brain barrier drug delivery. Curr Opin Pharmacol. 2006;6:494–500.
Discher DE, Eisenberg A. Polymer vesicles. Science. 2002;297:967–73.
Meng FH, Engbers HMG, Feigen J. Biodegradable polymer vesicles as a basis for artificial cells: encapsulation, release and targeting. J Control Release. 2005;101:187–98.
Ahmed F, Discher DE. Self-porating polymersomes of PEG-PLA and PEG-PCl: hydrolysis triggered controlled release vesicles. J Control Release. 2004;96:37–53.
Ahmed F, Pakunlu RI, Brannan A, Bates F, Minko T, Discher DE. Biodegradable polymer vesicles loaded with both paclitaxel and doxorubicin permeate and shrink tumors, inducing apoptosis in proportion to accumulated drug. J Control Release. 2006;116:150–8.
Mamiya T, Ukai M. [Gly14]-Humanin improved the learning and memory impairment induced by scopolamine in vivo. Br J Pharmacol. 2001;134:1597–9.
Avgoustakis K. Pegylated poly(Lactide) and poly(Lactide-Co-Glycolide) nanoparticles: preparation, properties and possible applications in drug delivery. Curr Drug Deliv. 2004;1:321–33.
Abraham SA, Edwards K, Karlsson G, Hudon N, Mayer LD, Bally MB. An evaluation of transmembrane ion gradient-mediated encapsulation of topotecan within liposomes. J Control Release. 2004;96:449–61.
Choucair A, Soo PL, Eisenberg A. Active loading and tunable release of doxorubicin from block copolymer vesicles. Langmuir. 2005;21:9308–13.
Wu J, Eisenberg A. Proton diffusion across membranes of vesicles of poly(styrene-b-acrylic Acid) diblock copolymers. JACS. 2006;128:2880–4.
Huwyler J, Wu DF, Pardridge WM. Brain drug delivery of small molecules using immunoliposomes. PNAS. 1996;93:14164–9.
Lu W, Wan J, She ZJ, Jiang XG. Brain delivery property and accelerated blood clearance of cationic albumin conjugated pegylated nanoparticle. J Control Release. 2007;118:38–53.
Olivier JC, Huertas R, Lee HJ, Calon F, Pardridge WM. Synthesis of pegylated immunonanoparticles. Pharm Res. 2002;19:1137–43.
Taylor S, Brock J, Kruger C. Safety determination for the use of bovine milkderived lactoferrin as a component of an antimicrobial beef carcass spray. Regul Toxicol Pharm. 2004;39:12–24.
Shard AG, Davies MC, Li YX, Volland C, Kissel T. XPS and SSIMS analysis revealing surface segregation and short-range order in solid films of block copolymers of PEO and PLGA. Macromolecules. 1997;30:3051–7.
Novotná K, Havliš J, Havel J. Optimisation of high performance liquid chromatography separation of neuroprotective peptides: fractional experimental designs combined with artificial neural networks. J Chromatogr A. 2005;1096:50–7.
Thom SR, Bhopale VM, Fisher D, Zhang J, Gimotty P. Delayed neuropathology after carbon monoxide poisoning is immune-mediated. Proc Natl Acad Sci. 2004;101:13660–5.
Alric L, Orfila C, Carrere N, Beraud M, Carrera G, Lepert JC, et al. Reactive oxygen intermediates and eicosanoid production by kupffer cells and infiltrated macrophages in acute and chronic liver injury induced in rats by CCl4. Inflamm Res. 2000;49:700–7.
Kim DH, Kim S, Jeon SJ, Son KH, Lee S, Yoon BH, et al. The effects of a-cute and repeated oroxylin a treatments on Aβ25-35 induced memory impairment in mice. Neuropharmacology. 2008;55:639–47.
Tajima H, Kawasumi M, Chiba T, Yamada M, Yamashita K, Nawa M, et al. A humanin derivative, S14G-HN, prevents amyloid–induced memory impairment in mice. J Neurosci Res. 2005;79:714–23.
Pardridge>Pardridge WM. Molecular Trojan horses for blood–brain barrier drug delivery. Curr Opin Pharmacol. 2006;6:494–500.
Pang ZQ, Lu W, Gao HL, Hu KL, Chen J, Zhang CL, et al. Preparation and brain delivery property of biodegradable polymersomes conjugated with OX26. J Control Release. 2008;128:120–7.
Nittaa A, Itoha A, Hasegawaa T, Nabeshima T. β-Amyloid protein-induced Alzheimer’s disease animal model. Neurosci Lett. 1994;170:63–6.
Su JH, Deng G, Cotman CW. Bax protein expression is increased in Alzheimer’s brain: correlations with DNA damage, Bcl-2 expression, and brain pathology. J Neuropath Exp Neur. 1997;56:86–93.
MacGibbon GA, Lawlor PA, Sirimanne ES, Walton MR, Connor B, Young D, et al. Bax expression in mammalian neurons undergoing apoptosis, and in Alzheimer’s disease hippocampus. Brain Res. 1997;750:223–34.
Grossoa CA, Tamb J, Royb S, Xanthoudakisb S, Costaa DD, Nicholsonb DW, et al. Caspase-3 cleaved spectrin colocalizes with neurofilament-immunoreactive neurons in Alzheimer’s disease. Neuroscience. 2006;141:863–74.
Guo B, Zhai DY, Cabezas E, Welsh K, Nouraini S, Satterthwait AC, et al. Humanin peptide suppresses apoptosis by interfering with Bax activation. Nature. 2003;423:456–61.
DeKosky ST, Ikonomovic MD, Styren SD, Beckett L, Wisniewski S, et al. Upregulation of choline acetyltransferase activity in hippocampus and frontal cortex of elderly subjects with mild cognitive impairment. Ann Neurol. 2002;51:145–5.
ACKNOWLEDGMENTS & DISCLOSURES
This work was supported by National Basic Research Program of China (973 Program 2007CB935800), National Science and Technology Major Project (2009ZX09310-006), National Natural Science Foundation of China (30762544), and Postdoctoral Science Foundation of China (20060390144). The authors acknowledge Dr. Kunpeng Li, School of Life Sciences, Zhongshan University, China, for his precious help on Cryo-TEM of polymersomes.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yu, Y., Pang, Z., Lu, W. et al. Self-Assembled Polymersomes Conjugated with Lactoferrin as Novel Drug Carrier for Brain Delivery. Pharm Res 29, 83–96 (2012). https://doi.org/10.1007/s11095-011-0513-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-011-0513-7