Abstract
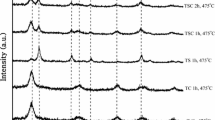
In this study, preparation of 2 mol% Ce and 4 mol% Co doped TiO2 nanopowders via sol–gel process have been investigated. The effects of Co and Ce do** and calcination temperature (475–1000 °C) on the structural and optical properties of titania nanopowders studied by X-ray diffraction (XRD), scanning electron microscope, transmission electron microscope and UV–Vis absorption spectroscope. XRD results showed that, Titania rutile phase formation in ternary system (Ti–Co–Ce) was inhibited by Ce4+ and promoted by Co4+ co-doped TiO2 in high temperatures (500–700 °C) and 61 mol% anatase composition is retained even after calcination at 800 °C. The optical absorption spectrum indicates that the TiO2 nanoparticles have a direct band gap of 3.21 eV. But optical band gap of the doped TiO2 (2 mol% Ce and 4 mol% Co) was found to be 3.14–3.20 eV.



Similar content being viewed by others
References
Bouras, P., Slathatos, E., Lianos, P.: Pure versus metal-ion-doped nanocrystalline titania for photocatalysis. Appl. Catal. B Environ. 73, 51–59 (2007)
Caimei, F., Peng, X., Yan**, S.: Preparation of nano-TiO2 doped with cerium and its photocatalytic activity. J. Rare Earth 24, 309–313 (2006)
Cao, Y., He, T., Zhao, L., Wang, E., Yang, W., Cao, Y.: Structure and phase transition behavior of Sn4+-doped TiO2 nanoparticles. J. Phys. Chem. C 113, 18121–18124 (2009)
Chen, X., Mao, S.S.: Titanium dioxide nanomaterials: synthesis, properties, modifications, and applications. Chem. Rev. 107, 2891–2959 (2007)
Fujishima, A., Honda, K.: Photolysis-decomposition of water at the surface of an irradiated semiconductor. Nature 238, 37–38 (1972)
Fukumura, T., Yamada, Y., Toyosaky, H., Hasegawa, T., Koinuma, H., Kawasaki, M.: Exploration of oxide-based diluted magnetic semiconductors toward transparent spintronics. Appl. Surf. Sci. 223, 62 (2004)
Gopal, M., Chan, W., Jonghe, L.: Room temperature synthesis of crystalline metal oxides. J. Mater. Sci. 32, 6001–6008 (1997)
Hoffmann, M.R., Martin, S.T., Choi, W., Bahnemann, D.W.: Environmental applications of semiconductor photocatalysis. Chem. Rev. 95, 69–96 (1995)
Janisch, R., Gopal, P., Spaldin, N.A.: Transition metal-doped TiO2 and ZnO—present status of the field. J. Phys.: Condens. Matter 17, R657 (2005)
**, Z., Zhang, X., Li, Y., Li, S., Lu, G.: 5.1 % Apparent quantum efficiency for stable hydrogen generation over eosin-sensitized CuO/TiO2 photocatalyst under visible light irradiation. Catal. Commun. 8, 1267–1273 (2007)
Klug, P., Alexander, L.E.: X-Ray Diffraction Procedures. Wiley, New York (1974)
Kostov, I.: The mineralogical knowledge of the ancient bulgarians according to some medieval sources. Annual vol. 46, part І, рр.87–92. Geology and Geophysics, Sofia, 2003 (1973)
Kubacka, A., Fuerte, A., Martinez-Arias, A., Fernandez-Garcia, M.: Nanosized Ti–V mixed oxides: effect of do** level in the photo-catalytic degradation of toluene using sunlight-type excitation. Appl. Catal. B Environ. 74, 26–33 (2007)
Kudo, A.: Recent progress in the development of visible light-driven powdered photocatalysts for water splitting. Int. J. Hydrogen Energy 32, 2673–2678 (2007)
Li, F.B., Li, X.Z., Hou, M.F., Cheah, K.W., Choy, W.C.H.: Enhanced photocatalytic activity of Ce3+–TiO2 for 2-mercaptobenzothiazole degradation in aqueous suspension for odour control. Appl. Catal. A Gen. 285, 181–189 (2005)
Mark H.F., Othmer D.F., Overberger C.G., Seaberg, G.T.: Encyclopedia of Chemical Technology, vol. 23, Wiley, New York (1983)
Ohko, Y., Fujishima, A., Hashimoto, K.: Kinetic analysis of the photocatalytic degradation of gas-phase 2-propanol under mass transport-limited conditions with a TiO2 film photocatalyst. J. Phys. Chem. B 102, 1724–1729 (1998)
Palmer, F.L., Eggins, B.R.: The effect of operational parameters on the photocatalytic degradation of humic acid. J. Photochem. Photobiol. 148, 137–143 (2002)
Paola, A.D., Garcı´a-Lo´ pez, E., Ikeda, S., Marcı`, G., Ohtani, B., Palmisano, L.: Photocatalytic degradation of organic compounds in aqueous systems by transition metal doped polycrystalline TiO2. Catal. Today 75, 87–93 (2002)
Rajeshwar, K., Tacconi, N.R., Chenthamarakshan, C.R.: Semiconductor-based composite materials: preparation, properties, and performance. Chem. Mater. 13, 2765–2782 (2001)
Shinde, S.R., Ogale, S.B., Das Sarma, S., Simpson, J.R., Drew, H.D., Lofland, S.E., Lanci, C., Buban, J.P., Browning, N.D., Kulkarmi, V.N., Higgins, J., Sharma, R.P., Green, R.L., Venkatesan, T.: Ferromagnetism in laser deposited anatase Ti1−x Cox O2−δ films. Phys. Rev. B 67, 115211 (2003)
Stengl, V., Bakardjieva, S., Murafa, N.: Preparation and photocatalytic activity of rare earth doped TiO2 nanoparticles. Mater. Chem. Phys. 114, 217–226 (2009)
Wang, C.Y., Liu, C.Y., Shen, T.: The photocatalytic oxidation of phenylmercaptotetrazole in TiO2 dispersions. J. Photochem. Photobiol. A Chem. 109, 65–70 (1997)
Weast, R.C.: Handbook of Chemistry and Physics, pp. B–154–B–155. CRC Press, Boca Raton (1984)
**e, Y., Yuan, C.: Photocatalysis of neodymium ion modified TiO2 sol under visible light irradiation. Appl. Surf. Sci. 221, 17–24 (2004)
Yan, Q.Z., Su, X.T., Huang, Z.Y., Ge, C.C.: Sol–gel auto-igniting synthesis and structural property of cerium-doped titanium dioxide nanosized powders. J. Eur. Ceram. Soc. 26, 915–921 (2006)
Zhang, X., Liu, Q.Q.: Preparation and characterization of titania photocatalyst co-doped with boron, nickel, and cerium. Mater. Lett. 62, 2589–2592 (2008)
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Ilkhechi, N.N., Alijani, M. & Kaleji, B.K. Optical and structural properties of TiO2 nanopowders with Co/Ce do** at various temperature. Opt Quant Electron 48, 148 (2016). https://doi.org/10.1007/s11082-016-0435-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11082-016-0435-z