Abstract
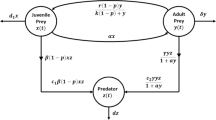
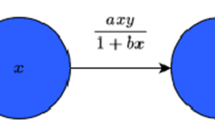
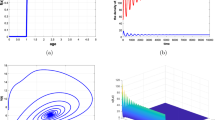
Many prey organisms have developed defense mechanisms against predation. If the predator can encounter in a limited spatial domain, the feeding rates reflect interference between predators and the corresponding functional response depends on both predator and prey densities. In this manuscript, an attempt has been made to understand the role of top predator interference and gestation delays on the dynamics of a three-species food chain model involving intermediate and top predators population. Interaction between the prey and an intermediate predator follows the Monod–Haldane functional response, while that between the top predator and its prey depends on Beddington–DeAngelis-type functional response. Analytically, we study the essential mathematical features such as boundedness, stability and direction of bifurcating periodic solution around the coexisting equilibrium for the model system. Numerically, we study the Hopf and transcritical bifurcations scenarios with respect to inhibitory effect of phytoplankton against zooplankton and death rate of fish population for the non-delayed system. Further, we study the stability behavior of the delayed model system. Model system exhibits irregular behavior when the interference is high or gestation period is larger than its critical value. Further, the system shows extinction of predators with the increase in inhibitory effect. Stability domain plots with respect to different important system parameters for non-delayed system and delay parameters for delayed system give a significant impact to study the stability of the different equilibrium points and bifurcation scenarios of both non-delayed and delayed systems. Our results point out the complexity of three-way interactions between phytoplankton, zooplankton and fish population in marine environment and highlight the role of predator interference and gestation delays in exhibiting the chaotic dynamics and extinction of predator population.















Similar content being viewed by others
References
Smayda,TJ. and Shimizu,Y. (eds) Toxic Phytoplankton Blooms in the Sea. Developments in marine biology, vol. 3. Elsevier, Amsterdam , pp. 1–952 (1993).
Scheffer, M.: Fish and nutrients interplay determines algal biomass: a minimal model. Oikos 62, 271–282 (1991a)
Vilar, J.M.G., Solé, R.V., Rubí, J.M.: On the origin of plankton patchiness. Phys. A Stat. Mech. Appl. 317(1–2), 239–246 (2003)
Chattopadhyay, J., Chatterjee, S., Venturino, E.: Patchy agglomeration as a transition from monospecies to recurrent plankton blooms. J. Theor. Biol. 253(2), 289–295 (2008)
Malchow, H.: Motion instabilities in predator–prey systems. J. Theor. Biol. 204(4), 639–647 (2000)
Levi, S.A., Segel, L.A.: Hypothesis for origin of planktonic patchness. Nature 259(5545), 659 (1976)
Greene, C.H., Widder, E.A., Youngbluth, M.J., Tamse, A., Johnson, G.E.: The migration behavior, fine structure and bioluminescent activity of krill sound-scattering layers. Limnol. Oceanogr. 37(3), 650–658 (1992)
Abbott, M.: Phytoplankton patchiness: ecological implications and observation methods. Patch Dyn. 96, 37–49 (1993)
Blaxter, J.H.S., Southward, A.J. (eds.): Advances in Marine Biology, vol. 83(3), pp. 264–265. Academic Press, San Diego (1997)
Hallegraeff, G.M.: A review of harmful algal blooms and their apparent global increase. Phycologia 32(2), 79–99 (1993)
Roy, S., Chattopadhyay, J.: Toxin-allelopathy among phytoplankton species prevents competitive exclusion. J. Biol. Syst. 15(01), 73–93 (2007)
Roy, S., Alam, S., Chattopadhyay, J.: Competing effects of toxin-producing phytoplankton on overall plankton populations in the Bay of Bengal. Bull. Math. Biol. 68(8), 2303–2320 (2006)
Chakraborty, S., Chattopadhyay, J.: Nutrient-phytoplankton–zooplankton dynamics in the presence of additional food source—a mathematical study. J. Biol. Syst. 16(04), 547–564 (2008)
Chakraborty, S., Bhattacharya, S., Feudel, U., Chattopadhyay, J.: The role of avoidance by zooplankton for survival and dominance of toxic phytoplankton. Ecol. Complex. 11, 144–153 (2012)
Kuwamura, M., Nakazawa, T., Ogawa, T.: A minimum model of prey-predator system with dormancy of predators and the paradox of enrichment. J. Math. Biol. 58(3), 459–479 (2009)
Kuwamura, M.: Turing instabilities in prey-predator systems with dormancy of predators. J. Math. Biol. 71(1), 125–149 (2015)
May, R.M.: Time-delay versus stability in population models with two and three trophic levels. Ecology 54(2), 315–325 (1973)
Yang, Y.: Hopf bifurcation in a two-competitor, one-prey system with time delay. Appl. Math. Comput. 214(1), 228–235 (2009)
Meng, X.Y., Huo, H.F., Zhang, X.B., **ang, H.: Stability and Hopf bifurcation in a three-species system with feedback delays. Nonlinear Dyn. 64(4), 349–364 (2011)
Rehim, M., Imran, M.: Dynamical analysis of a delay model of phytoplankton–zooplankton interaction. Appl. Math. Model. 36(2), 638–647 (2012)
Kuang, Y. (ed.): Delay Differential Equations: With Applications in Population Dynamics, vol. 191. Academic press, New York (1993)
Freedman, H.I., Ruan, S.: Hopf bifurcation in three-species food chain models with group defense. Math. Biosci. 111(1), 73–87 (1992)
Ma, Z.P., Li, W.T., Yan, X.P.: Stability and Hopf bifurcation for a three-species food chain model with time delay and spatial diffusion. Appl. Math. Comput. 219(5), 2713–2731 (2012)
Erbe, L.H., Freedman, H.I., Rao, V.S.H.: Three-species food-chain models with mutual interference and time delays. Math. Biosci. 80(1), 57–80 (1986)
Naji, R.K., Upadhyay, R.K., Rai, V.: Dynamical consequences of predator interference in a tri-trophic model food chain. Nonlinear Anal. Real World Appl. 11(2), 809–818 (2010)
Sarwardi, S., Haque, M., Mandal, P.K.: Persistence and global stability of Bazykin predator-prey model with Beddington-DeAngelis response function. Commun. Nonlinear Sci. Numer. Simul. 19(1), 189–209 (2014)
Chen, Y.: Multiple periodic solutions of delayed predator-prey systems with type IV functional responses. Nonlinear Anal. Real World Appl. 5(1), 45–53 (2004)
Gakkhar, S., Singh, A.: Complex dynamics in a prey predator system with multiple delays. Commun. Nonlinear Sci. Numer. Simul. 17(2), 914–929 (2012)
Zhang, L.Y.: Hopf bifurcation analysis in a Monod–Haldane predator–prey model with delays and diffusion. Appl. Math. Model. 39(3–4), 1369–1382 (2015)
Batabyal, S., Jana, D., Lyu, J., Parshad, R.D.: Explosive predator and mutualistic preys: a comparative study. Phys. A Stat. Mech. Appl. 541, 123348 (2019)
Jana, D., Upadhyay, R.K., Agrawal, R., Parshad, R.D., Basheer, A.: Explosive tritrophic food chain models with interference: a comparative study. J. Franklin Inst. 357, 385–413 (2019)
Pal, N., Samanta, S., Biswas, S., Alquran, M., Al-Khaled, K., Chattopadhyay, J.: Stability and bifurcation analysis of a three-species food chain model with delay. Int. J. Bifurc. Chaos 25(09), 1550123 (2015)
Anderson, T.W.: Predator responses, prey refuges, and density-dependent mortality of a marine fish. Ecology 82(1), 245–257 (2001)
Bairagi, N., Roy, P.K., Chattopadhyay, J.: Role of infection on the stability of a predator-prey system with several response functions—a comparative study. J. Theor. Biol. 248(1), 10–25 (2007)
Sharma, A., Sharma, A.K., Agnihotri, K.: Analysis of a toxin producing phytoplankton-zooplankton interaction with Holling IV type scheme and time delay. Nonlinear Dyn. 81(1–2), 13–25 (2015)
Zeng, G., Wang, F., Nieto, J.J.: Complexity of a delayed predator-prey model with impulsive harvest and Holling type II functional response. Adv. Complex Syst. 11(01), 77–97 (2008)
Holling, C.S.: The components of predation as revealed by a study of small-mammal predation of the European pine sawfly. Can. Entomol. 91(5), 293–320 (1959)
Holling, C.S.: Some characteristics of simple types of predation and parasitism. Can. Entomol. 91(7), 385–398 (1959)
Cosner, C., DeAngelis, D.L., Ault, J.S., Olson, D.B.: Effects of spatial grou** on the functional response of predators. Theor. Popul. Biol. 56(1), 65–75 (1999)
Hassell, M.P., Varley, G.C.: New inductive population model for insect parasites and its bearing on biological control. Nature 223(5211), 1133 (1969)
Beddington, J.R.: Mutual interference between parasites or predators and its effect on searching efficiency. J. Animal Ecol. 44(1), 331–340 (1975)
DeAngelis, D.L., Goldstein, R.A., O’neill, R.V.: A model for tropic interaction. Ecology 56(4), 881–892 (1975)
Crowley, P.H., Martin, E.K.: Functional responses and interference within and between year classes of a dragonfly population. J. N. Am. Benthol. Soc. 8(3), 211–221 (1989)
Skalski, G.T., Gilliam, J.F.: Functional responses with predator interference: viable alternatives to the Holling type II model. Ecology 82(11), 3083–3092 (2001)
Upadhyay, R.K., Naji, R.K., Raw, S.N., Dubey, B.: The role of top predator interference on the dynamics of a food chain model. Commun. Nonlinear Sci. Numer. Simul. 18(3), 757–768 (2013)
Jana, D., Tripathi, J.P.: Impact of generalist type sexually reproductive top predator interference on the dynamics of a food chain model. Int. J. Dyn. Control 5(4), 999–1009 (2017)
Chakraborty, S., Kooi, B.W., Biswas, B., Chattopadhyay, J.: Revealing the role of predator interference in a predator-prey system with disease in prey population. Ecol. Complex. 21, 100–111 (2015)
Pal, R., Basu, D., Banerjee, M.: Modelling of phytoplankton allelopathy with Monod-Haldane-type functional response—a mathematical study. Biosystems 95(3), 243–253 (2009)
Liu, Z., Tan, R.: Impulsive harvesting and stocking in a Monod-Haldane functional response predator-prey system. Chaos Solitons Fractals 34(2), 454–464 (2007)
Pei, Y., Zeng, G., Chen, L.: Species extinction and permanence in a prey-predator model with two-type functional responses and impulsive biological control. Nonlinear Dyn. 52(1–2), 71–81 (2008)
Maiti, A., Pal, A.K., Samanta, G.P.: Effect of time-delay on a food chain model. Appl. Math. Comput. 200(1), 189–203 (2008)
Do, Y., Baek, H., Lim, Y., Lim, D.: A three-species food chain system with two types of functional responses. In: Abstract and Applied Analysis. Hindawi (2011)
Zhang, Z., Yang, H., Liu, J.: Bifurcation analysis for a delayed food chain system with two functional responses. Electron. J. Qual. Theory Differ. Equ. 2013(53), 1–13 (2013)
Caro, T.: Antipredator Defenses in Birds and Mammals. University of Chicago Press, Chicago (2005)
Jeschke, J.M.: Density-dependent effects of prey defenses and predator offenses. J. Theor. Biol. 242(4), 900–907 (2006)
Mishra, P., Raw, S.N., Tiwari, B.: Study of a Leslie-Gower predator-prey model with prey defense and mutual interference of predators. Chaos Solitons Fractals 120, 1–16 (2019)
Jana, D., Agrawal, R., Upadhyay, R.K.: Top-predator interference and gestation delay as determinants of the dynamics of a realistic model food chain. Chaos Solitons Fractals 69, 50–63 (2014)
Nagumo, M.: Über die Lage der Integralkurven gewönlicher Differentialgleichungen. In: Proceedings of the Physico-Mathematical Society of Japan, 3rd Series, vol. 24, pp. 551–559 (1942)
Song, Y.L., Wei, J.J.: Bifurcation analysis for Chen’s system with delayed feedback and its application to control of chaos. Chaos Solitons Fractals 22(1), 75–91 (2004)
Hale, J.K.: Theory of Functional Differential Equations. Springer, New York (1977)
Hassard, B.D., Kazarinoff, N.D., Wan, Y.H., Wan, Y.W.: Theory and Applications of Hopf Bifurcation, vol. 41. CUP Archive, Cambridge (1981)
Jana, D., Bairagi, N.: Habitat complexity, dispersal and metapopulations: macroscopic study of a predatorprey system. Ecol. Complex. 17, 131–139 (2014)
Jana, D.: Chaotic dynamics of a discrete predator-prey system with prey refuge. Appl. Math. Comput. 224, 848–865 (2013)
Jana, D., Agrawal, R., Upadhyay, R.K.: Dynamics of generalist predator in a stochastic environment: effect of delayed growth and prey refuge. Appl. Math. Comput. 268, 1072–1094 (2015)
Jana, D., Agrawal, R., Upadhyay, R.K., Samanta, G.P.: Ecological dynamics of age selective harvesting of fish population: maximum sustainable yield and its control strategy. Chaos Solitons Fractals 93, 111–122 (2016)
Acknowledgements
This research work is supported by Science and Engineering Research Board (SERB), Government of India, under the Grant No. EMR/2017/000607 to the first author (Nilesh Kumar Thakur).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interests regarding the publication of this article.
Ethical approval
The authors state that this research complies with ethical standards. This research does not involve either human participants or animals.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix
Appendix
In order to compute the properties of the Hopf bifurcation, we denote any one of the critical values of \( \tau \) by \( \tau ^* \) without generality loss, and a pair of purely imaginary roots \( \pm i \omega _0 \) exists in Eq. (3.13) by which system undergoes Hopf bifurcation. Let \(N=N^*+x_1\), \(P=P^*+x_2 \), \(Z=Z^*+x_3\), \( \mu =\tau -\tau ^* \), where \( \mu \in \mathrm{Re} \). Rescaling the time by \( t \longrightarrow \frac{t}{\tau } \), system (3.1) can be written into the following continuous real-valued functions as \( C=([-1,0],\mathrm{Re}^3) \)
where \( x(t)=(x_1(t),x_2(t),x_3(t))^T\in \mathrm{Re}^3 \) and \( L_\mu :C\rightarrow \mathrm{Re}^3, \ f:\mathrm{Re}\times C\rightarrow \mathrm{Re}^3 \) are given, respectively,
such that
and
where \( \phi (\theta )=(\phi _1(\theta ),\phi _2(\theta ),\phi _3(\theta ))^T \in C([-1,0],\mathrm{Re}^3) \).
By the Riesz representation theorem, there exist a function \( \eta (\theta ,\mu )\) of bounded variation for \( \theta \in [-1,0] \), such that
In fact, we can take
where \( \delta (\theta ) \) is the Dirac delta function.
For \( \phi \in C_1 ([-1,0],\mathrm{Re}^3) \), define
and
System (7.1) is then equivalent to
where \( x_t(\theta )=x(t+\theta ) \) for \( \theta \in [-1,0]. \)
For \( \psi \in C^1([0,1],(\mathrm{Re}^3)^*) \), define
and a bilinear inner product is given by
where \(\overline{\psi }(0)\) and \(\overline{\psi }(\xi -\theta )\) are the complex conjugate of \(\psi (0)\) and \(\psi (\xi -\theta )\).
For further calculation, we assume that \( i\omega _0\tau ^* \) and \( -i\omega _0\tau ^* \) are eigenvalues of A(0) and \( A^* \), respectively.
Now, let \( q(\theta )=(1,\sigma _2,\sigma _3)^T \mathrm{e}^{i\omega _0\tau ^*\theta } \) and \( q^*(s)=M (1,\sigma _2^*,\sigma _3^*)\)\(\times \mathrm{e}^{i\omega _0\tau ^* s} \) are the eigenvector of A(0) and \( A^*(0)\) corresponding to \( +i\omega _0\tau ^* \) and \( -i\omega _0\tau ^* \), respectively. Then,
and for \(\theta =0\), we obtained
Solving the system of equations, we get
and
Similarly, let
where
and
Under the normalization condition \(\langle q^*(s),q(\theta ) \rangle = 1\), we have
where
and \(\overline{D}\) is the complex conjugate of D.
Now following in the same manner as given in [61], we obtained
which can also be written as
where
From Eq. (7.15), we obtained the value of \(g_{20}\), \(g_{11}\), \(g_{02}\) and \(g_{21}\) which is given in Sect. 4.
To compute \(g_{21}\), we have to compute the values of \(W_{20}^{(l)}(\theta )\) and \(W_{11}^{(l)}(\theta )\), for \(l=1,2,3.\)
Now, we denote \( W_{20}(\theta ) {=} (W_{20}^{(1)}(\theta ){,}W_{20}^{(2)}(\theta ){,}W_{20}^{(3)}(\theta ))^T \) and \( W_{11}(\theta ) {=} (W_{11}^{(1)}(\theta ),W_{11}^{(2)}(\theta ),W_{11}^{(3)}(\theta ))^T \).
By computing, we obtained
and
Here, \( E_1 = (E_1^{(1)},E_1^{(2)},E_1^{(3)}) \) and \( E_2 = (E_2^{(1)},E_2^{(2)},E_2^{(3)}) \in \mathrm{Re}^3\) are the constant vectors, which we have to be determined.
Now using [61], we have
Solve this system for \(E_1\), we obtained
and
where
Similarly, we have
Solve this system for \(E_2\), we obtained
and
where
Consequently, we determine the value of \( W_{20}(\theta ) \) and \( W_{11}(\theta ) \) from Eqs. (7.16) and (7.17). The value of \( g_{21} \) can be expressed by delay and parameters [Eq. (4.1)].
Rights and permissions
About this article
Cite this article
Thakur, N.K., Ojha, A., Jana, D. et al. Modeling the plankton–fish dynamics with top predator interference and multiple gestation delays. Nonlinear Dyn 100, 4003–4029 (2020). https://doi.org/10.1007/s11071-020-05688-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11071-020-05688-2