Abstract
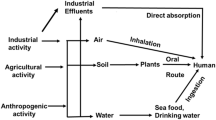
Heavy metal exposure has soared due to the twentieth century’s industrial activity. The most common heavy metals that lead to human poisoning are mercury, cadmium, and arsenic. Acute or chronic poisoning may develop following exposure to water, air, or food, so the bioaccumulation of these heavy metals causes harmful consequences in various human tissues and organs. Heavy metals interfere with biological functions such as growth, proliferation, differentiation, damage repair, and apoptosis. The mechanisms of action for these metals to cause toxicity are similar, including forming reactive oxygen species (ROS), weakening antioxidant defenses, enzyme inactivation, and oxidative stress. Heavy metal exposure is mainly associated with skin, liver, prostate, lung, urinary bladder, thyroid, and kidney cancers, as well as causing gastrointestinal malignancies. Several microRNAs (miRNAs or miRs) have been involved in various human cancers due to the dysregulation of miRNA function. Recent investigations have confirmed that microRNA dysregulation plays a role in the carcinogenesis of many tissues. This review presents the data concerning arsenic, cadmium, and mercury metals and their contamination sources, human exposure, toxicity, and inducing malignant transformations such as carcinogenicity in in-vitro or in-vivo specimens or dysregulated expression of microRNAs.
Graphical abstract






Similar content being viewed by others
References
Balali-Mood M, Naseri K, Tahergorabi Z, Khazdair MR, Sadeghi M (2021) Toxic mechanisms of five heavy metals: mercury, lead, chromium, cadmium, and arsenic. Front Pharmacol 12:643972
Durkalec M, Martínez-Haro M, Nawrocka A, Pareja-Carrera J, Smitsa JE, Mateo R (2022) Factors influencing lead, mercury and other trace element exposure in birds from metal mining areas.Environmental Research. :113575
Sharma S, Meena M, Swapnil P, Marwal A, Gupta AK (2022) Removal of volatile organic compounds and heavy metals through the biological-based process. An Innovative Role of Biofiltration in Wastewater Treatment Plants (WWTPs). Elsevier, pp 45–64
Wang S, Shi X (2001) Molecular mechanisms of metal toxicity and carcinogenesis. Mol Cell Biochem 222(1–2):3–9
Paithankar JG, Saini S, Dwivedi S, Sharma A, Chowdhuri DK (2021) Heavy metal associated health hazards: An interplay of oxidative stress and signal transduction. Chemosphere 262:128350
Rezayian M, Niknam V, Ebrahimzadeh H (2019) Oxidative damage and antioxidative system in algae. Toxicol Rep 6:1309–1313
Danes JM, Palma FR, Bonini MG (eds) (2021) Arsenic and other metals as phenotype driving electrophiles in carcinogenesis. Seminars in Cancer Biology; : Elsevier
Aalami A, Pouresmaeil V, Amirabadi A, Mojahed F, Rad M, Sahebkar A (2020) Evaluation of the Diagnostic Properties of Serum hsa-miR-223-5p in Detection of Gastric Cancer: A Case-Control Study. Anti-cancer agents in medicinal chemistry.
Leitão AL, Enguita FJ (2022) A Structural View of miRNA Biogenesis and Function. Non-coding RNA 8(1):10
Aalami AH, Abdeahad H, Mesgari M, Sahebkar A (2021) MicroRNA-223 in gastrointestinal cancers: a systematic review and diagnostic meta‐analysis. Eur J Clin Invest 51(2):e13448
Aalami AH, Abdeahad H, Shoghi A, Mesgari M, Amirabadi A, Sahebkar A (2022) Brain tumors and circulating micrornas: a systematic review and diagnostic meta-analysis. Expert Rev Mol Diagn 22(2):201–211
Qasemi Rad M, Pouresmaeil V, Hosseini Mojahed F, Amirabadi A, Aalami AH (2022) Clinicopathological utility of miR-203a-3p in diagnosing colorectal cancer.Molecular Biology Reports. :1–11
Aalami AH, Mesgari M, Sahebkar A Synthesis and characterization of green zinc oxide nanoparticles with antiproliferative effects through apoptosis induction and microRNA modulation in breast cancer cells. Bioinorganic chemistry and applications.2020.
Hosseini Mojahed F, Aalami AH, Pouresmaeil V, Amirabadi A, Qasemi Rad M, Sahebkar A Clinical evaluation of the diagnostic role of MicroRNA-155 in breast cancer.International Journal of Genomics.2020
Aalami AH, Abdeahad H, Mesgari M (2021) Circulating miR-21 as a potential biomarker in human digestive system carcinoma: a systematic review and diagnostic meta-analysis. Biomarkers 26(2):103–113
Sarkar A, Paul B, Darbha GK (2022) The groundwater arsenic contamination in the Bengal Basin-A review in brief.Chemosphere. :134369
De Monteiro EC, Caixeta ES, Santos VSV, Pereira BB (2021) Arsenic exposure from groundwater: environmental contamination, human health effects, and sustainable solutions. J Toxicol Environ Health Part B 24(3):119–135
Hussain MM, Bibi I, Niazi NK, Shahid M, Iqbal J, Shakoor MB et al (2021) Arsenic biogeochemical cycling in paddy soil-rice system: Interaction with various factors, amendments and mineral nutrients. Sci Total Environ 773:145040
Raju NJ (2022) Arsenic in the geo-environment: A review of sources, geochemical processes, toxicity and removal technologies. Environ Res 203:111782
Qamar AS, Qureshi MI (2021) Arsenic Phytotoxicity via the Membrane Transporters and Its Mechanism of Action and Detoxification in Plant Cells. Recent Advances for Sustainable Agriculture: CRC Press;, Plant-Microbe Dynamics, pp 87–102
Soler-Blasco R, Murcia M, Lozano M, Sarzo B, Esplugues A, Vioque J et al (2021) Urinary arsenic species and methylation efficiency during pregnancy: Concentrations and associated factors in Spanish pregnant women. Environ Res 196:110889
Jaishankar M, Tseten T, Anbalagan N, Mathew BB, Beeregowda KN (2014) Toxicity, mechanism and health effects of some heavy metals. Interdisciplinary Toxicol 7(2):60–72
Liu G, Song Y, Li C, Liu R, Chen Y, Yu L et al (2021) Arsenic compounds: The wide application and mechanisms applied in acute promyelocytic leukemia and carcinogenic toxicology. Eur J Med Chem 221:113519
Oh CC, ** A, Koh W-P (2021) Trends of cutaneous basal cell carcinoma, squamous cell carcinoma, and melanoma among the Chinese, Malays, and Indians in Singapore from 1968–2016. JAAD Int 4:39–45
Tchounwou PB, Udensi UK, Isokpehi RD, Yedjou CG, Kumar S (2015) Arsenic and cancer. Handbook of Arsenic Toxicology. Elsevier, pp 533–555
Brunton L, Knollmann B, Hilal-Dandan R (2018) organizadores. Goodman & Gilman’s the pharmacological basis of therapeutics. McGraw Hill Medical, New York
Tchounwou PB, Centeno JA, Patlolla AK (2004) Arsenic toxicity, mutagenesis, and carcinogenesis–a health risk assessment and management approach. Mol Cell Biochem 255(1–2):47–55
Cardoso APF, Al-Eryani L, States JC (2018) Arsenic-induced carcinogenesis: the impact of miRNA dysregulation. Toxicol Sci 165(2):284–290
Feng Z, Zhang C, Wu R, Hu W (2011) Tumor suppressor p53 meets microRNAs. J Mol Cell Biol 3(1):44–50
Cui Y, Han Z, Hu Y, Song G, Hao C, **a H et al (2012) MicroRNA-181b and microRNA‐9 mediate arsenic‐induced angiogenesis via NRP1. J Cell Physiol 227(2):772–783
Beezhold K, Liu J, Kan H, Meighan T, Castranova V, Shi X et al (2011) miR-190-mediated downregulation of PHLPP contributes to arsenic-induced Akt activation and carcinogenesis. Toxicol Sci 123(2):411–420
Sharma D, Tiwari M, Lakhwani D, Tripathi RD, Trivedi PK (2015) Differential expression of microRNAs by arsenate and arsenite stress in natural accessions of rice. Metallomics 7(1):174–187
Wang Z, Humphries B, **ao H, Jiang Y, Yang C (2014) MicroRNA-200b suppresses arsenic-transformed cell migration by targeting protein kinase Cα and Wnt5b-protein kinase Cα positive feedback loop and inhibiting Rac1 activation. J Biol Chem 289(26):18373–18386
Luo F, Ji J, Liu Y, Xu Y, Zheng G, **g J et al (2015) MicroRNA-21, up-regulated by arsenite, directs the epithelial–mesenchymal transition and enhances the invasive potential of transformed human bronchial epithelial cells by targeting PDCD4. Toxicol Lett 232(1):301–309
Hubaux R, Becker-Santos DD, Enfield KS, Rowbotham D, Lam S, Lam WL et al (2013) Molecular features in arsenic-induced lung tumors. Mol Cancer 12(1):20
Falchi L, Verstovsek S, Ravandi-Kashani F, Kantarjian HM (2016) The evolution of arsenic in the treatment of acute promyelocytic leukemia and other myeloid neoplasms: Moving toward an effective oral, outpatient therapy. Cancer 122(8):1160–1168
Mumford JL, Wu K, **a Y, Kwok R, Yang Z, Foster J et al (2007) Chronic arsenic exposure and cardiac repolarization abnormalities with QT interval prolongation in a population-based study. Environ Health Perspect 115(5):690–694
Kosnett MJ (ed) (2013) The role of chelation in the treatment of arsenic and mercury poisoning. Journal of Medical Toxicology; : Springer
Cui Z-G, Ahmed K, Zaidi SF, Muhammad JS (2021) Ins and outs of cadmium-induced carcinogenesis: Mechanism and prevention. Cancer Treat Res Commun 27:100372
Naeem A, Zafar M, Khalid H, Zia-ur-Rehman M, Ahmad Z, Ayub MA et al (2019) Cadmium-induced imbalance in nutrient and water uptake by plants. Cadmium toxicity and tolerance in plants. Elsevier, pp 299–326
Harries ML, Lam S, MacAulay C, Qu J, Palcic B (1995) Diagnostic imaging of the larynx: autofluorescence of laryngeal tumours using the helium-cadmium laser. J Laryngology Otology 109(2):108–110
Bernhoft RA (2013) Cadmium toxicity and treatment. The Scientific World Journal. ;2013
Wang P, Chen H, Kopittke PM, Zhao F-J (2019) Cadmium contamination in agricultural soils of China and the impact on food safety. Environ Pollut 249:1038–1048
Järup L, Åkesson A (2009) Current status of cadmium as an environmental health problem. Toxicol Appl Pharmcol 238(3):201–208
Ivari SAR, Darroudi A, Zavar MHA, Zohuri G, Ashraf N (2017) Ion imprinted polymer based potentiometric sensor for the trace determination of Cadmium (II) ions. Arab J Chem 10:S864–S9
Authority EFS (2012) Cadmium dietary exposure in the European population. EFSA J 10(1):2551
Makino T, Murakami M, Ishikawa S, Abe T (2019) Regulations for cadmium in Rice and soil in Japan and countermeasures to reduce the concentrations. Springer, Cadmium Toxicity, pp 103–114
Zou M, Zhou S, Zhou Y, Jia Z, Guo T, Wang J (2021) Cadmium pollution of soil-rice ecosystems in rice cultivation dominated regions in China: A review. Environ Pollut 280:116965
Micali A, Pallio G, Irrera N, Marini H, Trichilo V, Puzzolo D et al (2018) Flavocoxid, a natural antioxidant, protects mouse kidney from cadmium-induced toxicity. Oxidative medicine and cellular longevity. ;2018
Andrews GK (2000) Regulation of metallothionein gene expression by oxidative stress and metal ions. Biochem Pharmacol 59(1):95–104
Hernández-Cruz EY, Amador-Martínez I, Aranda-Rivera AK, Cruz-Gregorio A, Chaverri JP (2022) Renal damage induced by cadmium and its possible therapy by mitochondrial transplantation. Chemico-Biol Interact 361:109961
Ayoub N, Mantash H, Dhaini HR, Mourad A, Hneino M, Daher Z (2021) Serum cadmium levels and risk of metabolic syndrome: a cross-sectional study. Biol Trace Elem Res 199(10):3625–3633
Woolf AD, Itai-Itai, disease—Japan (1955) History of Modern Clinical Toxicology: Elsevier; 2022. p. 109 – 20
Kim K, Pollack AZ, Nobles CJ, Sjaarda LA, Zolton JR, Radoc JG et al (2021) Associations between blood cadmium and endocrine features related to PCOS-phenotypes in healthy women of reproductive age: a prospective cohort study. Environ Health 20(1):1–8
Yang O, Kim HL, Weon J-I, Seo YR (2015) Endocrine-disrupting chemicals: review of toxicological mechanisms using molecular pathway analysis. J cancer Prev 20(1):12
Itoh H, Iwasaki M, Sawada N, Takachi R, Kasuga Y, Yokoyama S et al (2014) Dietary cadmium intake and breast cancer risk in Japanese women: a case–control study. Int J Hyg Environ Health 217(1):70–77
Tchounwou PB, Yedjou CG, Patlolla AK, Sutton DJ (2012) Heavy metal toxicity and the environment. Springer, Molecular, clinical and environmental toxicology, pp 133–164
Hung Y-M, Chung H-M (2004) Acute self-poisoning by ingestion of cadmium and barium. Nephrol Dialysis Transplantation 19(5):1308–1309
Nordberg G (1998) Metal: chemical properties and toxicity. Encyclopedia of occupational health and safety. :63.19–63.24
Park JH, Lee BM, Kim HS (2021) Potential protective roles of curcumin against cadmium-induced toxicity and oxidative stress. J Toxicol Environ Health Part B 24(3):95–118
Rahimzadeh MR, Rahimzadeh MR, Kazemi S, Moghadamnia A-a (2017) Cadmium toxicity and treatment: An update. Caspian J Intern Med 8(3):135
Faroon O, Ashizawa A, Wright S, Tucker P, Jenkins K, Ingerman L et al (2012) Toxicological profile for cadmium.
Waalkes M, Berthan G (1995) Handbook on metal-ligand interactions of biological fluids. Marcel Dekker, New York
Ogunlewe J, Osegbe D (1989) Zinc and cadmium concentrations in indigenous blacks with normal, hypertrophic, and malignant prostate. Cancer 63(7):1388–1392
Lee J-D, Wu S-M, Lu L-Y, Yang Y-T, Jeng S-Y (2009) Cadmium concentration and metallothionein expression in prostate cancer and benign prostatic hyperplasia of humans. J Formos Med Assoc 108(7):554–559
Cheng Y, Zhang J, Wu T, Jiang X, Jia H, Qing S et al (2019) Reproductive toxicity of acute Cd exposure in mouse: Resulting in oocyte defects and decreased female fertility. Toxicol Appl Pharmcol 379:114684
Henson MC, Chedrese PJ (2004) Endocrine disruption by cadmium, a common environmental toxicant with paradoxical effects on reproduction. Experimental biology and medicine 229(5):383–392
Gu S, Dai J, Qu T, He Z (2019) Emerging Roles of MicroRNAs and Long Noncoding RNAs in Cadmium Toxicity.Biological trace element research. :1–10
Torun V, Değerli E, Cansaran-Duman D (2018) A Promising Role of Lichens, Their Secondary Metabolites and miRNAs on Treatment of Cancer Disease After Exposure to Carcinogenic Heavy Metals. Springer, Phytoremediation, pp 203–214
Liu Q, Zheng C, Shen H, Zhou Z, Lei Y (2015) MicroRNAs-mRNAs expression profile and their potential role in malignant transformation of human bronchial epithelial cells induced by cadmium. BioMed research international. ;2015
Kim J-J, Kim Y-S, Kumar V (2019) Heavy metal toxicity: An update of chelating therapeutic strategies. J Trace Elem Med Biol 54:226–231
Nelson LS, Hoffman RS, Howland MA, Lewin NA, Goldfrank LR (2018) Goldfrank’s toxicologic emergencies:McGraw Hill Professional;
Haribabu T, Sudha P (2011) Effect of heavy metals copper and cadmium exposure on the antioxidant properties of the plant Cleome gynandra. Int J Plant Anim Environ Sci 1(2):80–87
Katzung BG, Trevor AJ (2015) Basic & clinical pharmacology. McGraw-Hill Education New York
Sun G-F, Hu W-T, Yuan Z-H, Zhang B-A, Lu H (2017) Characteristics of mercury intoxication induced by skin-lightening products. Chin Med J 130(24):3003
Rai RK, Pati RS, Islam A, Roy G (2022) Detoxification of organomercurials by thiones and selones: A short review.Inorganica Chimica Acta. :120980
Haschek WM, Rousseaux CG, Wallig MA, Bolon B, Ochoa R (2013) Haschek and Rousseaux’s handbook of toxicologic pathology. Academic Press
Wang Y-J, Chen C-Z, Li P, Liu L, Chai Y, Li Z-H (2022) Chronic Toxic Effects of Waterborne Mercury on Silver Carp (Hypophthalmichthys molitrix) Larvae. Water 14(11):1774
Rice KM, Walker EM Jr, Wu M, Gillette C, Blough ER (2014) Environmental mercury and its toxic effects. J Prev Med public health 47(2):74
Tong M, Yu J, Liu M, Li Z, Wang L, Yin C et al (2021) Total mercury concentration in placental tissue, a good biomarker of prenatal mercury exposure, is associated with risk for neural tube defects in offspring. Environ Int 150:106425
Elder A, Nordberg GF, Kleinman M (2022) Exposure, dose, and toxicokinetics of metals. Elsevier, Handbook on the Toxicology of Metals, pp 55–86
Miraj SS, Poojari PG, Thunga G, Rao M (2020) Clinical Toxicology of Mercury: Source, Toxidrome, Mechanism of Toxicity and Management. CRC Press, Metal Toxicology Handbook, pp 393–404
Moszczyński P (1997) Mercury compounds and the immune system: a review. Int J Occup Med Environ Health 10(3):247–258
Briffa J, Sinagra E, Blundell R (2020) Heavy metal pollution in the environment and their toxicological effects on humans. Heliyon 6(9):e04691
Grandjean P, Satoh H, Murata K, Eto K (2010) Adverse effects of methylmercury: environmental health research implications. Environ Health Perspect 118(8):1137–1145
Silbernagel SM, Carpenter DO, Gilbert SG, Gochfeld M, Groth E, Hightower JM et al (2011) Recognizing and preventing overexposure to methylmercury from fish and seafood consumption: information for physicians. Journal of toxicology. ;2011
Li X, Pan J, Wei Y, Ni L, Xu B, Deng Y et al (2021) Mechanisms of oxidative stress in methylmercury-induced neurodevelopmental toxicity. Neurotoxicology 85:33–46
Ding E, Zhao Q, Bai Y, Xu M, Pan L, Liu Q et al (2016) Plasma microRNAs expression profile in female workers occupationally exposed to mercury. J Thorac disease 8(5):833
Onwuzuligbo O, Hendricks AR, Hassler J, Domanski K, Goto C, Wolf MT (2018) Mercury intoxication as a rare cause of membranous nephropathy in a child. Am J Kidney Dis 72(4):601–605
Yu H-S, Liao W-T, Chai C-Y (2006) Arsenic carcinogenesis in the skin. J Biomed Sci 13(5):657–666
Yu RC, Hsu K-H, Chen C-J, Froines JR (2000) Arsenic methylation capacity and skin cancer. Cancer Epidemiol Prev Biomarkers 9(11):1259–1262
Lindsay ER, Maathuis FJ (2017) New molecular mechanisms to reduce arsenic in crops. Trends Plant Sci 22(12):1016–1026
Shahid M, Niazi NK, Dumat C, Naidu R, Khalid S, Rahman MM et al (2018) A meta-analysis of the distribution, sources and health risks of arsenic-contaminated groundwater in Pakistan. Environ Pollut 242:307–319
Chi Z, Tan S, Tan X, Zhong L, Li W (2018) The toxicity of cadmium ion (Cd 2+) to phycocyanin: an in vitro spectroscopic study. Environ Sci Pollut Res 25(15):14544–14550
Kim HS, Kim YJ, Seo YR (2015) An overview of carcinogenic heavy metal: molecular toxicity mechanism and prevention. J cancer Prev 20(4):232
Oldani M, Fabbri M, Melchioretto P, Callegaro G, Fusi P, Gribaldo L et al (2020) in vitro and bioinformatics mechanistic-based approach for cadmium carcinogenicity understanding.Toxicology in vitro. :104757
Imura J, Tsuneyama K, Ueda Y (2019) Novel Pathological Study of Cadmium Nephropathy of Itai-itai Disease. Springer, Cadmium Toxicity, pp 39–50
Group MS (2007) Extramural depth of tumor invasion at thin-section MR in patients with rectal cancer: results of the MERCURY study. Radiology 243(1):132
Gómez MG, Boffetta P, Klink J, Espanol S, Quintana JG, Colin D (2007) Cancer mortality in mercury miners. Gac Sanit 21(3):210–217
Pandey G, Madhuri S (2014) Heavy metals causing toxicity in animals and fishes. Res J Anim Veterinary Fish Sci 2(2):17–23
Funding
No external funding was received for this study.
Author information
Authors and Affiliations
Contributions
Amir Hossein Aalami designed the manuscript. Amir Hossein Aalami, Mohammadsaleh Hoseinzadeh, Parsa Hosseini Manesh, Ali Jiryai Sharahi, and Ehsan Kargar Aliabadi have participated in data collection. Amir Hossein Aalami wrote the draft. All authors have the same contribution as the first author, and all names have been categorized based on alphabet order.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
The study has not described any experimentation on animals or humans.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
All authors have the same contribution as the first author. All names have been categorized based on alphabet order.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Aalami, A., Hoseinzadeh, M., Hosseini Manesh, P. et al. Carcinogenic effects of heavy metals by inducing dysregulation of microRNAs: A review. Mol Biol Rep 49, 12227–12238 (2022). https://doi.org/10.1007/s11033-022-07897-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-022-07897-x