Abstract
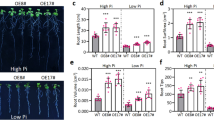
The DREB transcription factors comprise conserved ERF/AP2 DNA-binding domain, bind specifically to DRE/CRT motif and regulate abiotic stress mediated gene expression. In this study we show that PgDREB2A from Pennisetum glaucum is a powerful transcription factor to engineer multiple stress tolerance in tobacco plants. The PgDREB2A protein lacks any potential PEST sequence, which is known to act as a signal peptide for protein degradation. Therefore, the transgenic tobacco plants were raised using full-length cDNA without modification. The transgenics exhibited enhanced tolerance to both hyperionic and hyperosmotic stresses. At lower concentration of NaCl and mannitol, seed germination and seedling growth was similar in WT and transgenic, however at higher concentration germination in WT decreased significantly. D15 and D46 lines showed 4-fold higher germination percent at 200 mM NaCl. At 400 mM mannitol seed germination in WT was completely arrested, whereas in transgenic line it was more than 50%. Seedlings of D15 and D46 lines showed better growth like leaf area, root number, root length and fresh weight compared to wild type for both the stresses. The quantitative Real time PCR of transgenic showed higher expression of downstream genes NtERD10B, HSP70-3, Hsp18p, PLC3, AP2 domain TF, THT1, LTP1 and heat shock (NtHSF2) and pathogen-regulated (NtERF5) factors with different stress treatments.




Similar content being viewed by others
References
Hasegawa PM, Bressan RA, Zhu JK, Bohnert HJ (2000) Plant cellular and molecular responses to high salinity. Ann Rev Plant Physiol Plant Mol Biol 51:463–499
Cushman JC, Bohnert HJ (2000) Genomic approaches to plant stress tolerance. Curr Opin Plant Biol 3:117–124
Chen WQJ, Zhu T (2004) Networks of transcription factors with roles in environmental stress response. Trends Plant Sci 9:591–596
Agarwal PK, Agarwal P, Reddy MK, Sopory SK (2006) Role of DREB transcription factors in abiotic and biotic stress tolerance in plants. Plant Cell Rep 25:1263–1274
Sakuma Y, Maruyama K, Osakabe Y, Qin F, Seki M, Shinozaki K, Yamaguchi-Shinozaki K (2006) Functional analysis of an Arabidopsis transcription factor, DREB2A, involved in drought-responsive gene expression. Plant Cell 18:1292–1309
Sakuma Y, Maruyama K, Qin F, Osakabe Y, Seki M, Shinozaki K, Yamaguchi-Shinozaki K (2006) Dual function of an Arabidopsis transcription factor DREB2A in water-stress-responsive and heat-stress-responsive gene expression. Proc Natl Acad Sci USA 103:18828–18833
Qin F, Kakimoto M, Sakuma Y, Maruyama K, Osakabe Y, Tran L-S P, Shinozaki K, Yamaguchi-Shinozaki K (2007) Regulation and functional analysis of ZmDREB2A in response to drought and heat stresses in Zea mays L. Plant J 50:54–69
Shinozaki K, Yamaguchi-Shinozaki K (2000) Molecular responses to dehydration and low temperature: differences and cross-talk between two stress signaling pathways. Curr Opin Plant Biol 3:217–223
Maruyama K, Sakuma Y, Kasuga M, Ito Y, Seki M, Goda H, Shimada Y, Yoshida S, Shinozaki K, Yamaguchi-Shinozaki K (2004) Identification of cold-inducible downstream genes of the Arabidopsis DREB1A/CBF3 transcriptional factor using two microarray systems. Plant J 38:982–993
Zhao TJ, Sun S, Liu Y, Liu JM, Liu Q, Yan YB, Zhou HM (2006) Regulating the drought-responsive element (DRE)-mediated signaling pathway by synergic functions of trans-active and trans-inactive DRE binding factors in Brassica napus. J Biol Chem 281:10752–10759
Nakashima K, Shinwari ZK, Sakuma Y, Seki M, Miura S, Shinozaki K, Yamaguchi-Shinozaki K (2000) Organization and expression of two Arabidopsis DREB2 genes encoding DRE-binding proteins involved in dehydration- and high-salinity-responsive gene expression. Plant Mol Biol 42:657–665
Sakuma Y, Liu Q, Dubouzet JG, Abe H, Shinozaki K, Yamaguchi-Shinozaki K (2002) DNA-binding specificity of the ERF/AP2 domain of Arabidopsis DREB’s transcription factors involved in dehydration- and cold inducible gene expression. Biochem Biophys Res Commun 290:998–1009
Liu Q, Kasuga M, Sakuma Y, Abe H, Miura S, Yamaguchi-Shinozaki K, Shinozaki K (1998) Two transcription factors, DREB1 and DREB2, with an EREBP/AP2 DNA binding domain separate two cellular signal transduction pathways in drought- and low-temperature-responsive gene expression, respectively, in Arabidopsis. Plant Cell 10:1391–1406
Kasuga M, Liu Q, Miura S, Yamaguchi-Shinozaki K, Shinozaki K (1999) Improving plant drought, salt, and freezing tolerance by gene transfer of a single stress-inducible transcription factor. Nat Biotechnol 17:287–291
Dubouzet JG, Sakuma Y, Ito Y, Kasuga M, Dubouzet EG, Miura S, Seki M, Shinozaki K, Yamaguchi-Shinozaki K (2003) OsDREB genes in rice, Oryza sativa L., encode transcription activators that function in drought-, high-salt- and cold-responsive gene expression. Plant J 33:751–763
Chen M, Wang Q-Y, Cheng X-G, Xu Z-S, Li L-C, Ye X-G, **a L-Q, Ma Y–Z (2007) GmDREB2, a soybean DRE-binding transcription factor, conferred drought and high-salt tolerance in transgenic plants. Biochem Biophys Res Commun 353:299–305
Agarwal P, Agarwal PK, Nair S, Sopory SK, Reddy MK (2007) Stress inducible DREB2A transcription factor from Pennisetum glaucum is a phosphoprotein and phosphorylation negatively regulates its DNA binding activity. Mol Genet Genomics 277:189–198
Töpfer R, Matzeit V, Gronenborn B, Schell J, Steinbiss H–H (1987) A set of plant expression vectors for transcriptional and translational fusions. Nucl Acids Res 15:5890
Horsch RB, Fry JE, Hoffmann NL, Eichholtz D, Rogers SG, Farley RT (1985) A simple and general method for transferring genes into plants. Science 227:1229–1231
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:431–497
Jefferson RA (1987) Assaying chimeric genes in plants: the GUS fusion system. Plant Mol Biol Rep 5:387–405
Doyle JJ, Doyle JL (1987) A rapid DNA isolation procedure from small quantities of fresh leaf tissue. Phytochem Bull 19:11–15
Chomczynski P, Sacchi N (1987) Single-step method of RNA isolation by acid guanidinium thiocyanate–phenol–chloroform extraction. Anal Biochem 162:156–159
Reddy MK, Nair S, Tewari KK (1998) Cloning, expression and characterization of a gene which encodes a topoisomerase I with positive supercoiling activity in pea. Plant Mol Biol 37:773–784
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25:402–408
Steponkus PL, Uemura M, Joseph RA, Gilmour SJ, Thomashow MF (1998) Mode of action of the COR15a gene on the freezing tolerance of Arabidopsis thaliana. Proc Natl Acad Sci USA 95:14570–14575
Shimamura C, Ohno R, Nakamura C, Takumi S (2006) Improvement of freezing tolerance in tobacco plants expressing a cold-responsive and chloroplast-targeting protein WCOR15 of wheat. J Plant Physiol 163:213–219
Rogers S, Wells R, Rechsteiner M (1986) Amino acid sequences common to rapidly degraded proteins: the PEST hypothesis. Science 234:364–368
Salmeron J, Janzen J, Soneji Y, Bump N, Kamens J, Allen H, Ley SC (2001) Direct phosphorylation of NF-kappaB1 p105 by the IkapaB kinase complex on serine 927 is essential for signal induced p105 proteolysis. J Biol Chem 276:22215–22222
Mishra NS, Pham XH, Sopory SK, Tuteja N (2004) Pea DNA helicase 45 overexpression in tobacco confers high salinity tolerance without affecting yield. Proc Natl Acad Sci USA 102:509–514
Garg AK, Kim JK, Owens TG, Ranwala AP, Choi YD, Kochian LV, Wu RJ (2002) Trehalose accumulation in rice plants confers high tolerance levels to different abiotic stresses. Proc Natl Acad Sci USA 99:15898–15903
Singla-Pareek SL, Reddy MK, Sopory SK (2003) Genetic engineering of the glyoxalase pathway in tobacco leads to enhanced salinity tolerance. Proc Natl Acad Sci USA 100:14672–14677
Close TJ (1997) Dehydrins: a commonality in the response of plants to dehydration and low temperature. Physiol Plant 100:291–296
Kasuga M, Miura S, Shinozaki K, Yamaguchi-Shinozaki K (2004) A combination of the Arabidopsis DREB1A gene and stress-inducible rd29A promoter improved drought-and low-temperature stress tolerance in tobacco by gene transfer. Plant Cell Physiol 45:346–350
Schramm F, Larkindale J, Kiehlmann E, Ganguli A, Englich G, Vierling E, von Koskull-Döring P (2008) A cascade of transcription factor DREB2A and heat stress transcription factor HsfA3 regulates the heat stress response of Arabidopsis. Plant J 53:264–274
Chini A, Grant JJ, Seki M, Shinozaki K, Loake GJ (2004) Drought tolerance established by enhanced expression of the CC-NBS-LRR gene, ADR1, requires salicylic acid, EDS1 and ABI1. Plant J 38:810–822
Fischer U, Dröge-Laser W (2004) Overexpression of NtERF5, a new member of the tobacco ethylene response transcription factor family enhances resistance to Tobacco mosaic virus. Mol Plant Microbe Inter 17:1162–1171
Negrel J, Martin C (1984) The biosynthesis of feruloyltyramine in Nicotiana tabacum. Phytochemistry 23:2797–2801
Negrel J, Javelle F (1997) Purification, characterization and partial amino acid sequencing of hydroxy cinnamoyl-CoA:tyramine N-(hydroxycinnamoyl) transferase from tobacco cell-suspension cultures. Eur J Biochem 247:1127–1135
Schmidt A, Grimm R, Schmidt J, Scheel D, Strack D, Rosahl S (1999) Cloning and expression of a potato cDNA encoding hydroxycinnamoyl-CoA:tyramine N-(Hydroxycinnamoyl)transferase. J Biol Chem 274:4273–4280
Ishihara A, Kawata N, Matsukawa T, Iwamura H (2000) Induction of N-hydroxy cinnamoyltyramine synthesis and tyramine N-hydroxycinnamoyltransferase (THT) activity by wounding in maize leaves. Biosci Biotechnol Biochem 64:1025–1031
Sommerville CR, Browse J (1991) Plant lipids: metabolism, mutants, and membranes. Science 252:80–87
Kader JC (1996) Lipid-transfer proteins in plants. Ann Rev Plant Physiol Plant Mol Biol 47:627–654
Wu G, Robertson A, Liu X, Zheng P, Wilen R, Nesbitt N, Gusta L (2004) A lipid transfer protein gene BG-14 is differentially regulated by abiotic stress, ABA, anisomycin, and sphigosine in bromegrass (Bromus inermis). J Plant Physiol 161:449–458
Sapitnitskaya M, Maul P, McCollum GT, Guy CL, Weiss B, Samach A, Porat R (2006) Postharvest heat and conditioning treatments activate different molecular responses and reduce chilling injuries in grapefruit. J Exp Bot 57:2943–2953
Molina A, Diaz I, Vasil IK, Carbonero P, Garcia-Olmedo F (1996) Two cold- inducible genes encoding lipid transfer protein LTP4 from barley show differential responses to bacterial pathogens. Mol Gen Genet 252:162–168
Kalifa Y, Perlson E, Gilad A, Konrad Z, Scolnik PA, Bar-Zvi D (2004) Over- expression of the water and salt stress-regulated Asr1 gene confers an increased salt tolerance. Plant Cell Environ 27:1459–1468
Chen W, Provart NJ, Glazebrook J, Katagiri F, Chang HS, Eulgem T, Mauch F, Luan S, Zou G, Whitham SA, Budworth PR, Toa Y, **e Z, Chen X, Lam S, Kreps JA, Harpe JF, Si-Ammour A, Mauch-Mani B, Heinlein M, Kobayashi K, Hohn T, Dang JL, Wang X, Zhu T (2002) Expression profile matrix of Arabidopsis transcription factor genes suggests their putative functions in response to environmental stresses. Plant Cell 14:559–574
Kreps JA, Wu YJ, Chang HS, Zhu T, Wang X, Harper JF (2002) Transcriptome changes for Arabidopsis in response to salt, osmotic and cold stress. Plant Physiol 130:2129–2141
Seki M, Narusaka M, Ishida J, Nanjo T, Fujita M, Oono Y, Kamiya A, Nakajima M, Enju A, Sakurai T, Satou M, Akiyama K, Taji T, Yamaguchi-Shinozaki K, Carninci P, Kawai J, Hayashizak Y, Shinozaki K (2002) Monitoring expression profile of 7000 Arabidopsis genes under drought, cold-and high-salinity stresses using a full-length cDNA microarray. Plant J 31:279–292
Seki M, Ishida J, Narusaka M, Fujita M, Nanjo T, Umezawa T, Kamiya A, Nakajima M, Enju A, Sakurai T, Satou M, Akiyama K, Yamaguchi-Shinozaki K, Carninci P, Kawai J, Hayashizaki Y, Shinozaki K (2002) Monitoring expression profile of 7000 Arabidopsis genes under ABA treatments using a full-length cDNA microarray. Funct Integr Genomics 2:282–291
Buchanan CD, Lim S, Salzman RA, Kagiampakis I, Morishige DT, Weers BD, Klein RR, Pratt LH, Cordonnier-Pratt MM, Klein PE, Mullet JE (2005) Sorghum bicolor’s transcriptome response to dehydration, high salinity and ABA. Plant Mol Biol 58:699–720
Rizhsky L, Liang H, Mittler R (2002) The combined effect of drought stress and heat shock on gene expression in tobacco. Plant Physiol 130:1143–1151
Acknowledgements
This work was supported by internal grants from ICGEB, NATP (Indian Council and Agriculture Research, New Delhi). P.A. is thankful to Council of Scientific and Industrial Research (CSIR), New Delhi for SRF; and Bhavnagar University for providing facilities.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Agarwal, P., Agarwal, P.K., Joshi, A.J. et al. Overexpression of PgDREB2A transcription factor enhances abiotic stress tolerance and activates downstream stress-responsive genes. Mol Biol Rep 37, 1125 (2010). https://doi.org/10.1007/s11033-009-9885-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11033-009-9885-8