Abstract
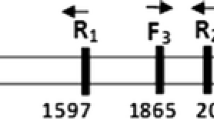
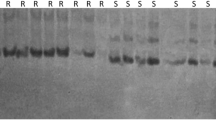
Three single nucleotide polymorphism (SNP) sites in which amino acids had changed were detected by sequence analysis within the leucine-rich repeat (LRR) region of the Fom-2 gene. Cleaved amplified polymorphic sequence (CAPS) and allele-specific PCR (AS-PCR) methods were employed to explore the SNP validation linked to fusarium wilt resistance in the F1 and F2 generations simultaneously. Homozygous- and heterozygous-resistant genotypes and homozygous-susceptible genotype could be clearly distinguished using the CAPS method, and three detected SNP sites were observed to be linked to fusarium wilt resistance, with a segregation ratio of 1:2:1 in the F2 generation. In addition, heterozygous-resistant and homozygous-susceptible genotypes could be clearly distinguished in the F1 generation using the AS-PCR method, showing a 3:1 segregation in terms of resistant and susceptible genotypes in the F2 generation. We therefore developed SNP-based functional markers (FMs) and identified some melon germplasm resistant to fusarium wilt by FM analysis within melon species. In conclusion, the SNP-based FMs originating from the SNP site of the Fom-2 LRR region were determined to be linked to fusarium wilt resistance and showed promise in the enhancement of breeding in melon.



Similar content being viewed by others
References
Andersen JR, Lubberstedt T (2003) Functional markers in plants. Trends Plant Sci 8:554–560
Arnholdt-Schmitt B, Costa JH, de Melo DF (2006) AOX—a functional marker for efficient cell reprogramming under stress? Trends Plant Sci 11:281–287
Bagge M, Lubberstedt T (2008) Functional markers in wheat: technical and economic aspects. Mol Breed 22:319–328
Bagge M, **a XC, Lubberstedt T (2007) Functional markers in wheat—commentary. Curr Opin Plant Biol 10:211–216
Bang H, Kim S, Leskovar D, King S (2007) Development of a codominant CAPS marker for allelic selection between canary yellow and red watermelon based on SNP in lycopene beta-cyclase (LCYB) gene. Mol Breed 20:63–72
Brunner S, Keller B, Feuillet C (2000) Molecular map** of the Rph7.g leaf rust resistance gene in barley (Hordeum vulgare L.). Theor Appl Genet 101:783–788
Chiapparino E, Lee D, Donini P (2004) Genoty** single nucleotide polymorphisms in barley by tetra-primer ARMS-PCR. Genome 47:414–420
Drenkard E, Richter BG, Rozen S, Stutius LM, Angell NA, Mindrinos M, Cho RJ, Oefner PJ, Davis RW, Ausubel FM (2000) A simple procedure for the analysis of single nucleotide polymorphisms facilitates map-based cloning in Arabidopsis. Plant Physiol 124:1483–1492
Gao ZS, van de Weg WE, Schaart JG, Schouten HJ, Tran DH, Kodde LP, van der Meer IM, van der Geest AHM, Kodde J, Breiteneder H, Hoffmann-Sommergruber K, Bosch D, Gilissen LJWJ (2005a) Genomic cloning and linkage map** of the Mal d 1 (PR-10) gene family in apple (Malus domestica). Theor Appl Genet 111:171–183
Gao ZS, van de Weg WE, Schaart JG, van der Meer IM, Kodde L, Laimer M, Breiteneder H, Hoffmann-Sommergruber K, Gilissen LJWJ (2005b) Linkage map positions and allelic diversity of two Mal d 3 (non-specific lipid transfer protein) genes in the cultivated apple (Malus domestica). Theor Appl Genet 110:479–491
Garces-Claver A, Fellman SM, Gil-Ortega R, Jahn M, Arnedo-Andres MS (2007) Identification, validation and survey of a single nucleotide polymorphism (SNP) associated with pungency in Capsicum spp. Theor Appl Genet 115:907–916
Grant MR, Godiard L, Straube E, Ashfield T, Lewald J, Sattler A, Innes RW, Dangl JL (1995) Structure of the Arabidopsis RPM1 gene enabling dual specificity disease resistance. Science 269:843–846
Grant MR, McDowell JM, Sharpe AG, de Torres Zabala M, Lydiate DJ, Dangl JL (1998) Independent deletions of a pathogen-resistance gene in Brassica and Arabidopsis. Proc Natl Acad Sci USA 95:15843–15848
HammondKosack KE, Jones JDG (1997) Plant disease resistance genes. Annu Rev Plant Physiol Plant Mol Biol 48:575–607
Iyer-Pascuzzi AS, McCouch SR (2007) Recessive resistance genes and the Oryza sativa-Xanthomonas oryzae pv. oryzae pathosystem. Mol Plant-Microbe Interact 20:731–739
Jones DA, Jones JDG (1997) The role of leucine-rich repeat proteins in plant defences. Adv Bot Res Inc Adv Plant Pathol 24:89–167
Joobeur T, King JJ, Nolin SJ, Thomas CE, Dean RA (2004) The fusarium wilt resistance locus Fom-2 of melon contains a single resistance gene with complex features. Plant J 39:283–297
Kim DS, Kim DH, Yoo JH, Kim BD (2006) Cleaved amplified polymorphic sequence and amplified fragment length polymorphism markers linked to the fertility restorer gene in chili pepper (Capsicum annuum L.). Mol Cell 21:135–140
Konieczny A, Ausubel FM (1993) A procedure for map** arabidopsis mutations using codominant ecotype-specific PCR-based markers. Plant J 4:403–410
Kwok S, Kellogg DE, McKinney N, Spasic D, Goda L, Levenson C, Sninsky JJ (1990) Effects of primer-template mismatches on the polymerase chain reaction: human immunodeficiency virus type 1 model studies. Nucl Acids Res 18:999–1005
Leach JGCT (1938) Fusarium wilt of muskmelons in Minnesota. Minnesota Agric Exp Station Tech Bull 129:32
Martin GB, Bogdanove AJ, Sessa G (2003) Understanding the functions of plant disease resistance proteins. Annu Rev Plant Biol 54:23–61
Martyn RDBL, Amador J (1987) Fusarium wilt (F.oxysporum f. sp. melonis race 0) of muskmelon in Texas. Plant Dis 70:233–236
Martyn RD, Park Y, Lovic BR, Miller ME (1996) The role of double stranded RNA (dsRNA) in the growth and virulence of the melon pathogen, Monosporascus cannonballus. Cucurbits Towards 2000:269–279
McCreight JD, Nerson H, Grumet R (1993) Melon Cucumis melo L. In: Kalloo G, Berch BO (eds) Genetic improvement of vegetable crops. Pergamon Press, Oxford, pp 267–294
Michelmore RW, Meyers BC (1998) Clusters of resistance genes in plants evolve by divergent selection and a birth-and-death process. Genome Res 8:1113–1130
Mondragon-Palomino M, Meyers BC, Michelmore RW, Gaut BS (2002) Patterns of positive selection in the complete NBS-LRR gene family of Arabidopsis thaliana. Genome Res 12:1305–1315
Newton CR, Graham A, Heptinstall LE, Powell SJ, Summers C, Kalsheker N, Smith JC, Markham AF (1989) Analysis of any point mutation in DNA: the amplification refractory mutation system (ARMS). Nucl Acid Res 17:2503–2516
Risser G, Mas P (1965) Putting obviousness of several races of Fusarium oxysporum f melonis. Ann Amelior Plant 15: 405–408
Risser G, Banihashemi Z, Davis DW (1976) Proposed nomenclature of Fusarium-oxysporum-f sp melonis races and resistance genes in Cucumis melo. Phytopathology 66:1105–1106
Robinson RW, Munger HM, Whitaker TW, Bohn GW (1976) Genes of Cucurbitaceae. Hortscience 11:554–568
Sherf AF, MacNab AA (1986) Vegetable diseases and their control, 2nd edn. Wiley, New York
Stahl EA, Dwyer G, Mauricio R, Kreitman M, Bergelson J (1999) Dynamics of disease resistance polymorphism at the Rpm1 locus of Arabidopsis. Nature 400:667–671
Tian D, Traw MB, Chen JQ, Kreitman M, Bergelson J (2003) Fitness costs of R-gene-mediated resistance in Arabidopsis thaliana. Nature 423:74–77
Wang YH, Thomas CE, Dean RA (2000) Genetic map** of a fusarium wilt resistance gene (Fom-2) in melon (Cucumis melo L.). Mol Breed 6:379–389
Wang YH, Choi WB, Thomas CE, Dean RA (2002) Cloning of disease-resistance homologues in end sequence of BAC clones linked to Fom-2, a gene conferring resistance to fusarium wilt in melon (Cucumis melo L.). Genome 45:473–480
Wu DY, Ugozzoli L, Pal BK, Wallace RB (1989) Allele-specific enzymatic amplification of beta-globin genomic DNA for diagnosis of sickle cell anemia. Proc Natl Acad Sci USA 86:2757–2760
Yeam I, Kang BC, Lindeman W, Frantz JD, Faber N, Jahn MM (2005) Allele-specific CAPS markers based on point mutations in resistance alleles at the pvr1 locus encoding eIF4E in Capsicum. Theor Appl Genet 112:178–186
Zheng XY, Wolff DW, Baudracco-Arnas S, Pitrat M (1999) Development and utility of cleaved amplified polymorphic sequences (CAPS) and restriction fragment length polymorphisms (RFLPs) linked to the Fom-2 fusarium wilt resistance gene in melon (Cucumis melo L.). Theor Appl Genet 99:453–463
Zink FW, Gubler WD (1985) Inheritance of resistance in muskmelon to fusarium-wilt. J Am Soc Hort Sci 110:600–604
Acknowledgments
This work was supported by a grant from Science and Technology Department of Zhejiang Province. We thank Prof. Gao for communicating the AS-PCR method.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, S., Yang, J. & Zhang, M. Developments of functional markers for Fom-2-mediated fusarium wilt resistance based on single nucleotide polymorphism in melon (Cucumis melo L.). Mol Breeding 27, 385–393 (2011). https://doi.org/10.1007/s11032-010-9439-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11032-010-9439-8