Abstract
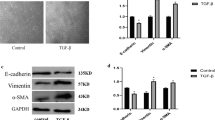
Claudin-1 (CLDN-1) is one of main tight junction components that play an important role in epithelial–mesenchymal transition (EMT). However, the effects of CLDN-1 on the migration and EMT induced by TGF-β1 in primary normal human bronchial epithelial (NHBE) and BEAS-2B cells have not been clear. The expression of CLDN-1 was quantified by Western blotting in NHBE and BEAS-2B cells. Cell migration and invasion were detected using transwell assays. The expression level of E-cadherin, N-cadherin, α-SMA, and Vimentin was evaluated by quantitative real-time PCR and Western blotting. Here we showed that the protein expression of CLDN-1 was increased exposed to TGF-β1 in a dose- and time-dependent manner. Knockdown of CLDN-1 using small interfering CLDN-1 RNA (siCLDN-1) prevented the migration and invasion in NHBE and BEAS-2B cells. Moreover, depletion of CLDN-1 promoted the E-cadherin expression and decreased the mRNA and protein levels of N-cadherin, α-SMA, and Vimentin induced by TGF-β1. Furthermore, CLDN-1 silencing resulted in the reduction of the Notch intracellular domain (NICD) and hairy enhancer of split-1 (Hes-1) in mRNA and protein level. Jagged-1, an activator of Notch signaling pathway, abrogated the protective function of siCLDN-1 in migration and EMT. In conclusion, CLDN-1 promoted the migration and EMT through the Notch signaling pathway.




Similar content being viewed by others
References
Elliot HA, Sampson MD, Roslyn Jaffe Food Allergy Institute (2015) Asthma in teenagers and adults, 10th edn. Wiley, New York
Nelson HS., Davies DE et al (2003) Airway remodeling in asthma: new insights. J Allergy Clin Immunol 111:215–225
Pascual RM., Peters SP (2005) Airway remodeling contributes to the progressive loss of lung function in asthma: An overview. J Allergy Clin Immunol 116:477–486
Pain M, Bermudez O et al (2014) Tissue remodelling in chronic bronchial diseases: from the epithelial to mesenchymal phenotype. Eur Respir Rev 23:118–130
Kalluri R (2009) EMT: when epithelial cells decide to become mesenchymal-like cells. J Clin Invest 119:1417–1419
Lv ZD, Na D et al (2011) Human peritoneal mesothelial cell transformation into myofibroblasts in response to TGF-ß1 in vitro. Int J Mol Med 27:187–193
Zeisberg M, Neilson EG (2009) Biomarkers for epithelial-mesenchymal transitions. J Clin Invest 119:1429–1437
Sohal SS, Mahmood MQ et al (2014) Clinical significance of epithelial mesenchymal transition (EMT) in chronic obstructive pulmonary disease (COPD): potential target for prevention of airway fibrosis and lung cancer. Clin Transl Med 3:1–4
Hei**k IH, Nawijn MC et al (2014) Airway epithelial barrier function regulates the pathogenesis of allergic asthma. Clin Exp Allergy J Br Soc Allergy Clin Immunol 44:620–630
Kudo M, Ishigatsubo Y et al (2013) Pathology of asthma. Front Microbiol 4:263
Yang Z-C et al (2013) Transforming growth factor-beta 1 induces bronchial epithelial cells to;mesenchymal transition by activating the Snail pathway and promotes airway remodeling in asthma. Mol Med Report 8:1663–1668
Horowitz JC, Thannickal VJ (2010) Epithelial-mesenchymal interactions in pulmonary fibrosis. Annu Rev Physiol 73:413–435
Furuse M, Hata M et al (2002) Claudin-based tight junctions are crucial for the mammalian epidermal barrier. J Cell Biol 156:1099–1111
Coyne CB, Gambling TM et al (2003) Role of claudin interactions in airway tight junctional permeability. Ajp Lung Cell Mol Physiol 285:6779–6785
Kaarteenaho R, Merikallio H et al (2010) Divergent expression of claudin -1, -3, -4, -5 and -7 in develo** human lung. Respir Res 11:1–10
Fujita H, Chalubinski M et al (2011) Claudin-1 expression in airway smooth muscle exacerbates airway remodeling in asthmatic subjects. J Allergy Clin Immunol 127:1612–1621(e1618)
Suh Y, Yoon CH et al (2013) Claudin-1 induces epithelial-mesenchymal transition through activation of the c-Abl-ERK signaling pathway in human liver cells. Oncogene 32:4873–4882
Kamitani S, Yamauchi Y et al (2010) Simultaneous stimulation with TGF-β1 and TNF-α induces epithelial mesenchymal transition in bronchial epithelial cells. Int Arch Allergy Immunol 155:119–128
Tatsuya I, Kedes L et al (2003) HES and HERP families: Multiple effectors of the notch signaling pathway. J Cell Physiol 194:237–255
Chen J, Hui C et al (2016) 3,6-Dihydroxyflavone suppresses the epithelial-mesenchymal transition in breast cancer cells by inhibiting the Notch signaling pathway. Sci Rep 6:28858
Weinmaster G (1998) Notch signaling: direct or what? Curr Opin Genet Dev 8:436–442
Matsuda M, Kubo A et al (2004) A peculiar internalization of claudins, tight junction-specific adhesion molecules, during the intercellular movement of epithelial cells. J Cell Sci 117:1247–1257
Swisshelm K, Macek R et al (2005) Role of claudins in tumorigenesis. Adv Drug Deliv Rev 57:919–928
Weber CR., Nalle SC et al (2008) Claudin-1 and claudin-2 expression are elevated in inflammatory bowel disease and may contribute to early neoplastic transformation. Lab Investig 88:1110–1120
Kaarteenahowiik R, Soini Y (2009) Claudin-1, -2, -3, -4, -5, and -7 in usual interstitial pneumonia and sarcoidosis. J Histochem Cytochem Off J Histochem Soc 57:187–195
Willis BC, Borok Z (2007) TGF-β-induced EMT: mechanisms and implications for fibrotic lung disease. Ajp Lung Cell Mol Physiol 293:L525–534
Gao J, Zhu Y et al (2014) TGF-β isoforms induce EMT independent migration of ovarian cancer cells. Cancer Cell Int 14:1–10
Zhu Y, Nilsson M et al (2010) Phenotypic plasticity of the ovarian surface epithelium: TGF-beta 1 induction of epithelial to mesenchymal transition (EMT) in vitro. Endocrinology 151:5497–5505
Holgate ST, Lackie P et al (2000) Bronchial epithelium as a key regulator of airway allergen sensitization and remodeling in asthma. Am J Respir Crit Care Med 162:113–117
Kalluri R, Weinberg RA (2009) The basics of epithelial-mesenchymal transition. J Clin Invest 119:1420–1428
Kiesslich T, Pichler M et al (2013) Epigenetic control of epithelial-mesenchymal-transition in human cancer. Mol Clin Oncol 1:3–11
Yi JM (2013) Claudin-1 induces epithelial-mesenchymal transition through activation of the c-Abl-ERK signaling pathway in human liver cells. Oncogene 32:4873–4882
Shiozaki A, Shimizu H et al (2014) Claudin 1 mediates tumor necrosis factor alpha-induced cell migration in human gastric cancer cells. World J Gastroenterol 20:17863–17876
Yoda S, Soejima K et al (2014) Claudin-1 is a novel target of miR-375 in non-small-cell lung cancer. Lung Cancer 85:366–372
Zhao X, Zou Y et al (2015) Lentiviral vector mediated claudin1 silencing inhibits epithelial to mesenchymal transition in breast cancer cells. Viruses 7:2965–2979
Lee JM, Dedhar S et al (2006) The epithelial-mesenchymal transition: new insights in signaling, development, and disease. J Cell Biol 172:973–981
Wang Z, Li Y et al (2010) The role of Notch signaling pathway in epithelial-mesenchymal transition (EMT) during development and tumor aggressiveness. Curr Drug Targets 11:745–751
Katoh M (2007) Notch signaling in gastrointestinal tract (review). Int J Oncol 30:247
Huang F, Zhu X et al (2012) Mesenchymal stem cells modified with miR-126 release angiogenic factors and activate Notch ligand Delta-like-4, enhancing ischemic angiogenesis and cell survival. Int J Mol Med 31:484–492
Miyamoto A, Weinmaster G (2009) Notch signal transduction: molecular and cellular mechanisms. Encycl Neurosci 9:1259–1268
Cheng S (2008) Notch signaling mediates hypoxia-induced tumor cell migration and invasion. Proc Natl Acad Sci 105:6392–6397
Wang Z, Li Y et al (2010) Down-regulation of Notch-1 and Jagged-1 inhibits prostate cancer cell growth, migration and invasion, and induces apoptosis via inactivation of Akt, mTOR, and NF-κB signaling pathways. J Cell Biochem 109:726–736
Ozasa Y, Akazawa H et al (2013) Notch activation mediates angiotensin II-induced vascular remodeling by promoting the proliferation and migration of vascular smooth muscle cells. Hypertens Res 36:859–865
Pope JL (2013) Claudin-1 regulates intestinal epithelial homeostasis through the modulation of Notch-signalling. Gut 63:622–634
Pope JL, Ahmad R et al (2014) Claudin-1 Overexpression in intestinal epithelial cells enhances susceptibility to adenamatous polyposis coli-mediated colon tumorigenesis. Mol Cancer 13:1–13
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no financial conflicts of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Rights and permissions
About this article
Cite this article
Lv, J., Sun, B., Mai, Z. et al. CLDN-1 promoted the epithelial to migration and mesenchymal transition (EMT) in human bronchial epithelial cells via Notch pathway. Mol Cell Biochem 432, 91–98 (2017). https://doi.org/10.1007/s11010-017-3000-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-017-3000-6