Abstract
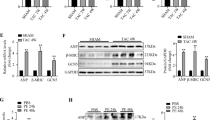
Cardiomyocyte hypertrophy is an adaptive response of the heart to various types of stress. During the period of stress accumulation, the transition from physiological hypertrophy to pathological hypertrophy results in the promotion of heart failure. Gelsolin (GSN) is a member of the actin-binding proteins, which regulate dynamic actin filament organization by severing and cap**. Moreover, GSN also regulates cell morphology, differentiation, movement, and apoptosis. In this study, we used H9c2 and H9c2-GSN stable clones in an attempt to understand the mechanisms of GSN overexpression in cardiomyocytes. These data showed that the overexpression of GSN in H9c2-induced cardiac hypertrophy and increased the pathological hypertrophy markers atrial natriuretic peptide brain natriuretic peptide. Furthermore, we found that E-cadherin expression decreased with the overexpression of GSN in H9c2, but β-catenin expression increased. These data presume that the cytoskeleton is loose. Further, previous studies show that the mitogen-activated protein kinase pathway can induce cardiac hypertrophy. Our data showed that p-p38 expression increased with the overexpression of GSN in H9c2, and the transcription factor p-GATA4 expression also increased, suggesting that the overexpression of GSN in H9c2-induced cardiac hypertrophy seemed to be regulated by the p38/GATA4 pathway. Moreover, we used both the p38 inhibitor (SB203580) and GSN siRNA to confirm our conjecture. We found that both of these factors significantly suppressed gelsolin-induced cardiac hypertrophy through p38/GATA4 signaling pathway. Therefore, we predict that the gene silencing of GSN and/or the downstream blocking of GSN along the p38 pathway could be applied to ameliorate pathological cardiac hypertrophy in the future.





Similar content being viewed by others
References
Cartwright EJ, Oceandy D, Austin C, Neyses L (2011) Ca2+ signalling in cardiovascular disease: the role of the plasma membrane calcium pumps. Sci China Life Sci 54(8):691–698
Heineke J, Molkentin JD (2006) Regulation of cardiac hypertrophy by intracellular signalling pathways. Nat Rev Mol Cell Biol 7(8):589–600
Lorell BH, Carabello BA (2000) Left ventricular hypertrophy: pathogenesis, detection, and prognosis. Circulation 102(4):470–479
Yamamoto S, Kita S, Iyoda T, Yamada T, Iwamoto T (2011) New molecular mechanisms for cardiovascular disease: cardiac hypertrophy and cell-volume regulation. J Pharmacol Sci 116(4):343–349
Tezuka F, Takahashi T (1976) Pathology of cardiac hypertrophy in pressure overload. Jpn Circ J 40(10):1111–1118
Johnson GL, Lapadat R (2002) Mitogen-activated protein kinase pathways mediated by ERK, JNK, and p38 protein kinases. Science 298(5600):1911–1912
Sugden PH, Clerk A (1998) “Stress-responsive” mitogen-activated protein kinases (c-Jun N-terminal kinases and p38 mitogen-activated protein kinases) in the myocardium. Circ Res 83(4):345–352
Liang F, Lu S, Gardner DG (2000) Endothelin-dependent and -independent components of strain-activated brain natriuretic peptide gene transcription require extracellular signal regulated kinase and p38 mitogen-activated protein kinase. Hypertension 35(1 Pt 2):188–192
Garrington TP, Johnson GL (1999) Organization and regulation of mitogen-activated protein kinase signaling pathways. Curr Opin Cell Biol 11(2):211–218
Tenhunen O, Sarman B, Kerkela R, Szokodi I, Papp L, Toth M, Ruskoaho H (2004) Mitogen-activated protein kinases p38 and ERK 1/2 mediate the wall stress-induced activation of GATA-4 binding in adult heart. J Biol Chem 279(23):24852–24860
Kerkela R, Pikkarainen S, Majalahti-Palviainen T, Tokola H, Ruskoaho H (2002) Distinct roles of mitogen-activated protein kinase pathways in GATA-4 transcription factor-mediated regulation of B-type natriuretic peptide gene. J Biol Chem 277(16):13752–13760
Silacci P, Mazzolai L, Gauci C, Stergiopulos N, Yin HL, Hayoz D (2004) Gelsolin superfamily proteins: key regulators of cellular functions. Cell Mol Life Sci 61(19–20):2614–2623
Sun HQ, Yamamoto M, Mejillano M, Yin HL (1999) Gelsolin, a multifunctional actin regulatory protein. J Biol Chem 274(47):33179–33182
Li GH, Shi Y, Chen Y, Sun M, Sader S, Maekawa Y, Arab S, Dawood F, Chen M, De Couto G et al (2009) Gelsolin regulates cardiac remodeling after myocardial infarction through DNase I-mediated apoptosis. Circ Res 104(7):896–904
Yin HL, Stossel TP (1979) Control of cytoplasmic actin gel–sol transformation by gelsolin, a calcium-dependent regulatory protein. Nature 281(5732):583–586
McGough AM, Staiger CJ, Min JK, Simonetti KD (2003) The gelsolin family of actin regulatory proteins: modular structures, versatile functions. FEBS Lett 552(2–3):75–81
Kwiatkowski DJ (1999) Functions of gelsolin: motility, signaling, apoptosis, cancer. Curr Opin Cell Biol 11(1):103–108
Li GH, Arora PD, Chen Y, McCulloch CA, Liu P (2012) Multifunctional roles of gelsolin in health and diseases. Med Res Rev 32(5):999–1025
Yang J, Moravec CS, Sussman MA, DiPaola NR, Fu D, Hawthorn L, Mitchell CA, Young JB, Francis GS, McCarthy PM et al (2000) Decreased SLIM1 expression and increased gelsolin expression in failing human hearts measured by high-density oligonucleotide arrays. Circulation 102(25):3046–3052
Mani SK, Shiraishi H, Balasubramanian S, Yamane K, Chellaiah M, Cooper G, Banik N, Zile, Kuppuswamy D (2008) In vivo administration of calpeptin attenuates calpain activation and cardiomyocyte loss in pressure-overloaded feline myocardium. Am J Physiol Heart Circ Physiol 295(1):H314–H326
Suhler E, Lin W, Yin HL, Lee WM (1997) Decreased plasma gelsolin concentrations in acute liver failure, myocardial infarction, septic shock, and myonecrosis. Crit Care Med 25(4):594–598
Towbin JA, Bowles NE (2002) The failing heart. Nature 415(6868):227–233
Hunter JJ, Chien KR (1999) Signaling pathways for cardiac hypertrophy and failure. N Engl J Med 341(17):1276–1283
Nishio R, Matsumori A (2009) Gelsolin and cardiac myocyte apoptosis: a new target in the treatment of postinfarction remodeling. Circ Res 104(7):829–831
Molkentin JD, Lu JR, Antos CL, Markham B, Richardson J, Robbins J, Grant SR, Olson EN (1998) A calcineurin-dependent transcriptional pathway for cardiac hypertrophy. Cell 93(2):215–228
Huang CY, Lee SD (2012) Possible pathophysiology of heart failure in obesity: cardiac apoptosis. BioMedicine 2(1):36–40
Roux PP, Blenis J (2004) ERK and p38 MAPK-activated protein kinases: a family of protein kinases with diverse biological functions. Microbiol Mol Biol Rev 68(2):320–344
Wright CD, Chen Q, Baye NL, Huang Y, Healy CL, Kasinathan S, O’Connell TD (2008) Nuclear alpha1-adrenergic receptors signal activated ERK localization to caveolae in adult cardiac myocytes. Circ Res 103(9):992–1000
Acknowledgments
This study was supported by grants from Taipei Medical University -Shuang Ho Hospital (102 TMU-SHH-16), CMU 102-S-13 and CMUBH R102-008, also in part by the Taiwan Department of Health Clinical Trial and Research Center of Excellence (DOH102-TD-B-111-004).
Author information
Authors and Affiliations
Corresponding author
Additional information
Y.-M. Liou and C.-Y. Huang share equal contribution.
Rights and permissions
About this article
Cite this article
Hu, WS., Ho, TJ., Pai, P. et al. Gelsolin (GSN) induces cardiomyocyte hypertrophy and BNP expression via p38 signaling and GATA-4 transcriptional factor activation. Mol Cell Biochem 390, 263–270 (2014). https://doi.org/10.1007/s11010-014-1977-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-014-1977-7