Abstract
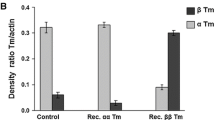
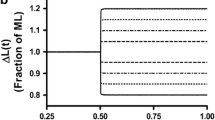
The thin filament extraction and reconstitution protocol was used to investigate the functional roles of tropomyosin (Tm) isoforms and phosphorylation in bovine myocardium. The thin filament was extracted by gelsolin, reconstituted with G-actin, and further reconstituted with cardiac troponin together with one of three Tm varieties: phosphorylated αTm (αTm.P), dephosphorylated αTm (αTm.deP), and dephosphorylated βTm (βTm.deP). The effects of Ca, phosphate, MgATP and MgADP concentrations were examined in the reconstituted fibres at pH 7.0 and 25°C. Our data show that Ca2+ sensitivity (pCa50: half saturation point) was increased by 0.19 ± 0.07 units when βTm.deP was used instead of αTm.deP (P < 0.05), and by 0.27 ± 0.06 units when phosphorylated αTm was used (P < 0.005). The cooperativity (Hill factor) decreased (but insignificantly) from 3.2 ± 0.3 (5) to 2.8 ± 0.2 (7) with phosphorylation. The cooperativity decreased significantly from 3.2 ± 0.3 (5) to 2.1 ± 0.2 (9) with isoform change from αTm.deP to βTm.deP. There was no significant difference in isometric tension or stiffness between αTm.P, αTm.deP, and βTm.deP muscle fibres at saturating [Ca2+] or after rigor induction. Based on the six-state cross-bridge model, sinusoidal analysis indicated that the equilibrium constants of elementary steps differed up to 1.7× between αTm.deP and βTm.deP, and up to 2.0× between αTm.deP and αTm.P. The rate constants differed up to 1.5× between αTm.deP and βTm.deP, and up to 2.4× between αTm.deP and αTm.P. We conclude that tension and stiffness per cross-bridge are not significantly different among the three muscle models.









Similar content being viewed by others
References
Amphlett GM, Syska H, Perry SV (1976) The polymorphic forms of tropomyosin and troponin I in develo** rabbit skeletal muscle. FEBS Lett 63:22–26
Boussouf SE, Maytum R, Jaquet K, Geeves MA (2007) Role of tropomosin isoforms in the calcium sensitivity of striated muscle thin filaments. J Muscle Res Cell Motil 28:49–58
Bowater R, Sleep J (1988) Demembranated muscle fibers catalyze a more rapid exchange between phosphate and adenosine triphosphate than actomyosin subfragment 1. Biochemistry 27:5314–5323
Brandt PW, Cox RN, Kawai M, Robinson T (1982) Effect of cross-bridge kinetics on apparent Ca2+ sensitivity. J Gen Physiol 79:997–1016
Briggs MM, McGinnis HD, Schachat F (1990) Transitions from fetal to fast troponin T isoforms are coordinated with changes in tropomyosin and α-actinin isoforms in develo** rabbit skeletal muscle. Dev Biol 140:253–260
Brisson JR, Golosinska K, Smillie LB, Sykes BD (1986) Interaction of tropomyosin and troponin T: a proton nuclear magnetic resonance study. Biochemistry 25:4548–4555
Bronson DD, Schachat FH (1982) Heterogeneity of contractile proteins: differences in tropomyosin in a fast, mixed and a slow skeletal muscles of the rabbit. J Biol Chem 257:3937–3944
Brown HR, Schachat FH (1985) Renaturation of skeletal muscle tropomyosin: implications for in vivo assembly. Proc Nat Acad Sci USA 82:2359–2363
Brown JH, Zhou Z, Reshetnikova L, Robinson H, Yammani RD, Tobacman LS, Cohen C (2005) Structure of the mid-region of tropomyosin: bending and binding sites for actin. Proc Nat Acad Sci USA 102:18878–18883
Cabral-Lilly D, Tobacman LS, Mehegan JP, Cohen C (1997) Molecular polarity in tropomyosin–troponin T co-crystals. Biophys J 73:1763–1770
Chalovich JM (1992) Actin mediated regulation of muscle contraction. Pharmaco Ther 55:95–148
Cooke R (1997) Actomyosin interaction in striated muscle. Physiol Rev 77:671–697
Cooke R, Pate E (1985) The effects of ADP and phosphate on the contraction of muscle fibers. Biophys J 48:789–798
Cummins P, Perry SV (1973) The subunits and biological activity of polymorphic forms of tropomyosin. Biochem J 133:765–777
Dantzig J, Goldman Y, Millar NC, Lacktis J, Homsher E (1992) Reversal of the cross-bridge force-generating transition by the photogeneration of phosphate in rabbit psoas muscle fibres. J Physiol 451:247–278
de Belle I, Mak AS (1987) Isolation and characterization of tropomyosin kinase from chicken embryo. Biochim Biophys Acta 925:17–26
Ding W, Fujita H, Kawai M (2002) The length of cooperative units on the thin filament in rabbit psoas muscle fibres. Exp Physiol 87:691–697
Eisenberg E, Kielley WW (1974) Troponin–tropomyosin complex. Column chromatographic separation and activity of the three active troponin components with and without tropomyosin present. J Biol Chem 249:4742–4748
Farah CS, Reinach FC (1999) Regulatory properties of recombinant tropomyosins containing 5-hydroxytryptophan: Ca2+-binding to troponin results in a conformational change in a region of tropomyosin outside the troponin binding site. Biochemistry 38:10543–10551
Ferenczi MA, Simmons RM, Sleep JA (1982) General considerations of cross-bridge models in relation to the dependence on MgATP concentration of mechanical parameters of skinned fibers from frog muscles. Soc Gen Physiol Ser 37:91–107
Flicker PF, Phillips GN Jr, Cohen (1982) Troponin and its interactions with tropomyosin. An electron microscope study. J Mol Biol 162:495–501
Fortune NS, Geeves MA, Ranatunga KW (1991) Tension responses to rapid pressure release in glycerinated rabbit muscle fibers. Proc Natl Acad Sci (USA) 88:7323–7327
Fujita H, Kawai M (2002) Temperature effect on isometric tension is mediated by regulatory proteins tropomyosin and troponin in bovine myocardium. J Physiol 539:267–276
Fujita H, Yasuda K, Niitsu S, Funatsu T, Ishiwata S (1996) Structural and functional reconstitution of thin filaments in the contractile apparatus of cardiac muscle. Biophys J 71:2307–2318
Fujita H, Sasaki D, Ishiwata S, Kawai M (2002) Elementary steps of the cross-bridge cycle in bovine myocardium with and without regulatory proteins. Biophys J 82:915–928
Fujita H, Lu X, Suzuki M, Ishiwata S, Kawai M (2004) The effect of tropomyosin on force and elementary steps of the cross-bridge cycle in reconstituted bovine myocardium. J Physiol 556:637–649
Gaffin RD, Gokulan K, Sacchettini JC, Hewett T, Klevitsky R, Robbins J, Muthuchamy M (2004) Charged residue changes in the carboxy-terminus of alpha-tropomyosin alter mouse cardiac muscle contractility. J Physiol 556.2:531–543
Gordon AM, Homsher E, Regnier M (2000) Regulation of contraction in striated muscle. Physiol Rev 80:853–924
Greene LE, Eisenberg E (1988) Relationship between regulated actomyosin ATPase activity and cooperative binding of myosin to regulated actin. Cell Biophys 12:59–71
Greenfield NJ, Huang YJ, Swapna GV, Bhattacharya A, Rapp B, Singh A, Montelione GT, Hitchcock-DeGregori SE (2006) Solution NMR structure of the junction between tropomyosin molecules: implications for actin binding and regulation. J Mol Biol 364:80–96
Güth K, Potter JD (1987) Effect of rigor and cycling cross-bridges on the structure of troponin C and on the Ca2+ affinity of the Ca2+-specific regulatory sites in skinned rabbit psoas fibers. J Biol Chem 262:13627–13635
Hammell RL, Hitchcock-DeGregori SE (1997) The sequence of the alternatively spliced sixth exon of α-tropomyosin is critical for cooperative actin binding but not for interaction with troponin. J Biol Chem 272:22409–22416
Haselgrove JC (1972) X-ray evidence for a conformational change in the actin-containing filaments of vertebrate strialed muscle. Cold Spring Harb Symp Quant Biol 37:341–352
Hayley M, Chevaldina T, Mudalige WA, Jackman DM, Dobbin AD, Heeley DH (2008) Shark skeletal muscle tropomyosin is a phosphoprotein. J Muscle Res Cell Motil 29:101–107
Heeley DH (1994) Investigation of the effects of phosphorylation of rabbit striated muscle αα-tropomyosin and rabbit skeletal muscle troponin T. Eur J Biochem 221:129–137
Heeley DH, Moir AJ, Perry SV (1982) Phosphorylation of tropomyosin during development in mammalian striated muscle. FEBS Lett 146:115–118
Heeley DH, Watson MH, Mak AS, Dubord P, Smillie LB (1989) Effect of phosphorylation on the interaction and functional properties of rabbit striated muscle αα-tropomyosin. J Biol Chem 264:2424–2430
Herzig JW, Peterson JW, Solaro RJ, Ruegg JC (1982) Phosphate and vanadate reduce the efficiency of the chemo-mechanical energy transformation in cardiac muscle. Adv Exp Med Biol 151:267–281
Hill TL, Eisenberg E, Greene L (1980) Theoretical model for the cooperative equilibrium binding of myosin subfragment 1 to the actin–troponin–tropomyosin complex. Proc Natl Acad Sci USA 77:3186–3190
Huxley HE (1972) Structural changes in actin- and myosin-containing filaments during contraction. Cold Spring Harbor Symp Quant Biol 37:361–376
Johnson P, Smillie LB (1977) Polymerizability of rabbit skeletal tropomyosin: effects of enzymic and chemical modifications. Biochemistry 16(10):2264–2269
Kawai M (1986) The role of orthophosphate in crossbridge kinetics in chemically skinned rabbit psoas fibers as detected with sinusoidal and step length alterations. J Muscle Res Cell Motil 7:421–434
Kawai M, Brandt PW (1976) Two rigor states in skinned crayfish single muscle fibers. J Gen Physiol 68:267–280
Kawai M, Brandt PW (1977) Effectof MgATP on stiffness measured at two frequencies in Ca2+-activated muscle fibers. Proc Natl Acad Sci USA 74:4073–4075
Kawai M, Brandt PW (1980) Sinusoidal analysis: a high resolution method for correlating biochemical reactions with physiological processes in activated skeletal muscles of rabbit, frog and crayfish. J Muscle Res Cell Motil 1:279–303
Kawai M, Halvorson HR (1989) Role of MgATP and MgADP in the cross-bridge kinetics in chemically skinned rabbit psoas fibers. Study of a fast exponential process (C). Biophys J 55:595–603
Kawai M, Halvorson HR (1991) Two step mechanism of phosphate release and the mechanism of force generation in chemically skinned fibers of rabbit psoas. Biophys J 59:329–342
Kawai M, Ishiwata S (2006) Use of thin filament reconstituted muscle fibres to probe the mechanism of force generation. J Muscle Res Cell Motil 27:455–468
Kawai M, Zhao Y (1993) Cross-bridge scheme and force per cross-bridge state in skinned rabbit psoas muscle fibers. Biophys J 65:638–651
Kawai M, Saeki Y, Zhao Y (1993) Cross-bridge scheme and the kinetic constants of elementary steps deduced from chemically skinned papillary and trabecular muscles of the ferret. Circ Res 73:35–50
Kawai M, Lu X, Hitchcock-DeGregori SE, Stanton KJ, Wandling MW (2009) Tropomyosin period 3 is essential for enhancement of isometric tension in thin filament-reconstituted bovine myocardium. J Biophysics 2009:1–17
Kon T, Imamula K, Roberts AJ, Ohkura R, Knight PJ, Gibbons IR, Burgess SA, Sutoh K (2009) Helix sliding in the stalk coiled coil of dynein couples ATPase and microtubule binding. Nature Strct Mol Biol 16(3):326–333
Kondo H, Ishiwata S (1976) Uni-directional growth of F-actin. J Biochem 79:159–171
Kress M, Huxley HE, Faruqi AR, Hendrix J (1986) Structural changes during activation of frog muscle studied by time-resolved X-ray diffraction. J Mol Biol 188:325–342
Kurokawa H, Fujii W, Ohmi K, Sakurai T, Nonomur Y (1990) Simple and rapid purification of brevin. Biochem Biophys Res Commun 168:451–457
Leger J, Bouvert P, Schwartz K, Swynghedauw B (1976) A comparative study of skeletal and cardiac tropomyosins; subunits, thiol group content and biological activities. Pflugers Archiv 362:271–277
Lehrer SS (1975) Intramolecular crosslinking of tropomyosin via disulfide bond formation: evidence for chain register. Proc Nat Acad Sci USA 72:3377–3381
Lewis WG, Smillie LB (1980) The amino acid sequence of rabbit cardiac tropomyosin. J Biol Chem 255:6854–6859
Lu X, Tobacman LS, Kawai M (2003) Effects of tropomyosin internal deletion Δ23Tm on isometric tension and the cross-bridge kinetics in bovine myocardium. J Physiol (Lond) 553:457–471
Lu X, Bryant MK, Bryan KE, Rubenstein PA, Kawai M (2005) Role of the N-terminal negative charges of actin in force generation and cross-bridge kinetics in reconstituted bovine cardiac muscle fibres. J Physiol 564:65–82
Lu X, Tobacman LS, Kawai M (2006) Temperature-dependence of isometric tension and cross-bridge kinetics of cardiac muscle fibers reconstituted with a tropomyosin internal deletion mutant. Biophys J 91:4230–4240
Mak AS, Smillie LB (1981) Structural interpretation of the two-site binding of troponin on the muscle thin filament. J Mol Biol 149:541–550
Mak A, Smillie LB, Bárány M (1978) Specific phosphorylation at serine-283 of alpha tropomyosin from frog skeletal and rabbit skeletal and cardiac muscle. Proc Natl Acad Sci (USA) 75:3588–3592
Mak AS, Smillie LB, Stewart GR (1980) A comparison of the amino acid sequences of rabbit skeletal muscle α-and β-tropomyosins. J Biol Chem 255:3647–3655
Maytum R, Bathe F, Konrad M, Geeves MA (2004) Tropomyosin exon 6b is troponin-specific and required for correct acto-myosin regulation. J Biol Chem 279:18203–18209
McKillop DFA, Geeves MA (1993) Regulation of the interaction between actin and myosin subfragment 1: evidence for three states of the thin filament. Biphys J 65:693–701
Montarras D, Fiszman MY, Gros F (1981) Characterization of the tropomyosin present in various chick embryo muscle types and in muscle cells differentiated in vitro. J Biol Chem 256:4081–4086
Montarras D, Fiszman MY, Gros F (1982) Changes in tropomyosin during development of chick embryonic skeletal muscles in vivo and during differentiation of chick muscle cells in vitro. J Biol Chem 257(1):545–548
Montgomery K, Mak AS (1984) In vitro phosphorylation of tropomyosin by a kinase from chicken embryo. J Biol Chem 259:5555–5560
Murakami K, Stewart M, Nozawa K, Tomii K, Kudou N, Igarashi N, Shirakihara Y, Wakatsuki S, Yasunaga T, Wakabayashi T (2008) Structural basis for tropomyosin overlap in thin (actin) filaments and the generation of a molecular swivel by troponin T. Proc Nat Acad Sci USA 105:7200–7205
Murray JM, Knox MK, Trueblood CE, Weber A (1982) Potentiated state of the tropomyosin actin filament and nucleotide-containing myosin subfragment 1. Biochemistry 21:906–915
Muthuchamy M, Gripp IL, Grupp G, O’Toole BA, Kiert AB, Boivin GP, Neumann J, Wieczorek DF (1995) Molecular and physiological effects of overexpressing striated muscle β-tropomyosin in the adult murine heart. J Biol Chem 270:30593–30603
Muthuchamy M, Boivin GP, Grupp IL, Wieczorek DF (1998) Beta-tropomyosin overexpression induces severe cardiac abnormalities. J Mol Cell Cardiol 30:1545–1557
O’Connor CM, Balzer DR, Lazarides E (1979) Phosphorylation of subunit proteins of intermediate filaments from chicken muscle and nonmuscle cells. Proc Nat Acad Sci USA 76:819–823
Ohtsuki I (1979) Molecular arrangement of troponin-T in the thin filament. J Biochem 86:491–497
Ookubo N, Ueno H, Ooi T (1975) Similarities and differences of the α and β components of tropomyosin. J Biochem 78:739–747
Palmiter KA, Kitada Y, Muthuchamy M, Wieczorek DF, Solaro RJ (1996) Exchange of beta- for alpha-tropomyosin in hearts of transgenic mice induces changes in thin filament response to Ca2+, strong cross-bridge binding, and protein phosphorylation. J Biol Chem 71:11611–11614
Pearlstone JR, Smillie LB (1982) Binding of troponin-T fragments to several types of tropomyosin. Sensitivity to Ca2+ in the presence of troponin-C. J Biol Chem 257:10587–10592
Perry SV (1998) Troponin T: genetics, properties and function. J Muscle Res Cell Motil 19:575–602
Perry SV (2001) Vertebrate tropomyosin: distribution, properties and function. J Muscle Res Cell Motil 22:5–49
Potter JD (1982) Preparation of troponin and its subunits. Methods Enzymol 85(Pt B):241–263
Rao VS, Marongelli EN, Guilford WH (2009) Phosphorylation of tropomyosin extends cooperative binding of myosin beyond a single regulatory unit. Cell Motil Cytoskeleton 66:10–23
Ribolow H, Bárány M (1977) Phosphorylation of tropomyosin in live frog muscle. Arch Biochem Biophys 179:718–720
Rodriguez AG, Flint GV, Regnier M (2008) Effect of tropomyosin isoforms on actomyosin interaction in an in vitro motility assay. Biophys J 94:232A
Rosol M, Lehman W, Craig R, Landis C, Butters C, Tobacman LS (2000) Three-dimensional reconstruction of thin filaments containing mutant tropoyosin. Biophys J 78:908–917
Roy RK, Sreter FA, Sarkar S (1979) Changes in tropomyosin subunits and myosin light chains during development of chicken and rabbit striated muscles. Dev Biol 69:15–30
Sano K, Maeda K, Oda T, Maeda Y (2000) The effect of single residue substitutions of serine 283 on the strength of head to tail interaction and actin binding properties of rabbit skeletal muscle α-tropomyosin. J Biochem (Tokyo) 127:1095–1102
Schachat FH, Bronson DD, McDonald OB (1985) Heterogeneity of contractile proteins. A continuum of troponin-tropomyosin expression in mammalian skeletal muscle. J Biol Chem 260:1108–1113
Singh A, Hitchcock-DeGregori S (2006) Dual requirement for flexibility and specificity for binding of the coiled-coil tropomyosin to its target, actin. Structure 14:43–50
Smillie LB (1982) Preparation and identification of alpha- and beta-tropomyosins. Methods Enzymol 85(Pt B):234–241
Smillie L (1999) Tropomyosins: In: Kreis T, Vale R (eds) Guidebook to the cytoskeletal and motor proteins, 2nd edn. Oxford University Press, Oxford, pp 159–164
Spudich JA, Watt S (1971) The regulation of rabbit skeletal muscle contraction. I. Biochemical studies of the interaction of the tropomyosin–troponin complex with actin and the proteolytic fragments of myosin. J Biol Chem 246:4866–4871
Stapleton MT, Fuchsbauer CM, Allshire AP (1998) BDM drives protein dephosphorylation and inhibits adenine nucleotide exchange in cardiomyocytes. Am J Physiol 275:H1260–H1266
Stienen GJ, Roosemalen MC, Wilson MG, Elzinga G (1990) Depression of force by phosphate in skinned skeletal muscle fibers of the frog. Am J Physiol 259:C349–C357
Takagi Y, Shuman H, Goldman YE (2004) Coupling between phosphate release and force generation in muscle actomyosin. Philos Trans R Soc Lond B Biol Sci 359:1913–1920
Tesi C, Colomo F, Piroddi N, Poggesi C (2002) Characterization of the cross-bridge force-generating step using inorganic phosphate and BDM in myofibrils from rabbit skeletal muscles. J Physiol (London) 541:187–199
Tobacman LS (1996) Thin filament-mediated regulation of cardiac contraction. Annu Rev Physiol 58:447–481
Tobacman LS, Butters CA (2000) A new model of cooperative myosin-thin filament binding. J Biol Chem 275:27587–27593
Vahebi S, Ota A, Li M, Warren CM, de Tombe PP, Wang Y, Solaro RJ (2007) p38-MAPK induced dephosphorylation of α-tropomyosin is associated with depression of myocardial sarcomeric tension and ATPase activity. Circ Res 100:408–415
Vibert P, Craig R, Lehman W (1997) Steric-model for activation of muscle thin filaments. J Mol Biol 266:8–14
Wakabayashi K, Tokunaga M, Kohno I, Sugimoto Y, Hamanaka T, Takezawa Y, Wakabayashi T, Amemiya Y (1992) Small-angle synchrotron x-ray scattering reveals distinct shape changes of the myosin head during hydrolysis of ATP. Science 258:443–447
Wang G, Kawai M (2001) Effect of temperature on elementary steps of the cross-bridge cycle in rabbit soleus slow-twitch muscle fibres. J Physiol (Lond) 531:219–234
Wannenburg T, Heijne GH, Geerdink JH, Van Den Dool HW, Janssen PM, De Tombe PP, Wannenburg et al (2000) Cross-bridge kinetics in rat myocardium: effect of sarcomere length and calcium activation. Am J Physiol Heart Circ Physiol 279:H779–H790
Waurick R, Knapp J, Van Aken H, Boknik P, Neumann J, Schmitz W (1999) Effect of 2, 3-butanedione monoxime on force of contraction and protein phosphorylation in bovine smooth muscle. Naunyn Schmiedebergs Arch Pharmacol 359:484–492
Wolska BM, Keller RS, Evans CC, Palmiter KA, Phillips RM, Muthuchamy M, Oehlenschlager J, Wieczorek DF, de Tombe PP, Solaro RJ (1999) Correlation between myofilament response to Ca2+ and altered dynamics of contraction and relaxation in transgenic cardiac cells that express β-tropomyosin. Circ Res 84:745–751
Xu C, Craig R, Tobacman L, Horowitz R, Lehman W (1999) Tropomyosin positions in regulated thin filaments revealed by cryoelectron microscopy. Biophys J 77:985–992
Zhao Y, Kawai M (1996) Inotropic agent EMD-53998 weakens nucleotide and phosphate binding to cross-bridges in porcine myocardium. Am J Physiol 271 (Heart Circ Physiol 40):H1394–H1406
Acknowledgements
The authors would like to thank Ms. Mary K. Bryant for her excellent technical assistance in reconstitution experiments, and Dr. Sathivel Chinathambi for his help in 2D SDS-PAGE. This work was supported by grants from NIH HL70041 and AHA 0850184Z to MK, AHA Postdoctoral Fellowships 0320083Z and 0520084Z to XL, and the Canadian Institutes of Health Research (C.I.H.R.) and the Heart and Stroke Foundation of Canada to LBS. The contents of this work are solely the responsibility of the authors and do not necessarily represent the official view of awarding organizations.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lu, X., Heeley, D.H., Smillie, L.B. et al. The role of tropomyosin isoforms and phosphorylation in force generation in thin-filament reconstituted bovine cardiac muscle fibres. J Muscle Res Cell Motil 31, 93–109 (2010). https://doi.org/10.1007/s10974-010-9213-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10974-010-9213-x