Abstract
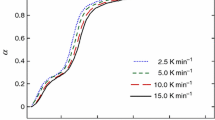
As one 3-D coordination polymer, lead formate was synthesized; calorimetric study and thermal analysis for this compound were performed. The low-temperature heat capacity of lead formate was measured by a precise automated adiabatic calorimeter over the temperature range from 80 to 380 K. No thermal anomaly or phase transition was observed in this temperature range. A four-step sequential thermal decomposition mechanism for the lead formate was found through the DSC and TG-DTG techniques at the temperature range from 500 to 635 K.
Similar content being viewed by others
References
J.L.C. Rowsell and O. M. Yaghi, Micropor. Mesopor. Mater., 73 (2004) 3.
S. Kitagawa, R. Kitaura and S. Noro, Angew. Chem. Int. Ed., 43 (2004) 2334.
P. G. Harrison and A. T. Steel, J. Organomet. Chem., 239 (1982) 105.
R. C. Weast, CRC Handbook of Chemistry and Physics, 62nd Ed. (Chemical Rubber, Boca Raton, FL 1981).
K. Betzler, H. Hesse, R. Jaquet and D. Lammers, J. Appl. Phys., 87 (2000) 2.
P. Baraldi, Spectrochim. Acta A, 37 (1981) 99.
M. N. Ray and N. D. Sinnarkar, J. Inorg. Nucl. Chem., 35 (1973) 1373.
V. G. Bessergenev, Y. A. Kovalevskaya, L. G. Lavrenova and I. E. Paukov, J. Therm. Anal. Cal., 75 (2004) 331.
J. Boerio-Goates, R. Stevens, B. Lang and B. F. Woodfield, J. Therm. Anal. Cal., 69 (2002) 773.
V. A. Drebushchak, E. V. Boldyreva, Y. A. Kovalevskaya, I. E. Paukov and T. N. Drebushchak, J. Therm. Anal. Cal., 79 (2005) 65.
V. Rohac, M. Fulem, H. G. Schmidt, V. Ruzicka, K. Ruzicka and G. Wolf, J. Therm. Anal. Cal., 70 (2002) 455.
L. Wang, Z. C. Tan, S. H. Meng, D. B. Liang, S. T. Ji and Z. K. Hei, J. Therm. Anal. Cal., 66 (2001) 409.
Z. C. Tan, B. Xue, S. W. Lu, S. H. Meng, X. H. Yuan and Y. S. Song, J. Therm. Anal. Cal., 63 (2000) 297.
D. A. Ditmars, S. Ishihara, S. S. Chang, G. Bernstein and B. D. West, J. Res. Natl. Bur. Stand., 87 (1982) 159.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, J., Liu, Y.Y., Zhang, Z.H. et al. Low-temperature heat capacity and thermodynamic properties of crystalline lead formate. J Therm Anal Calorim 89, 643–647 (2007). https://doi.org/10.1007/s10973-006-7510-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-006-7510-9