Abstract
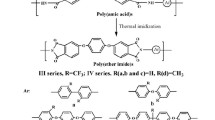
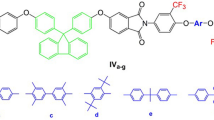
A series of new fluorinated aromatic poly(ether imide)s (PEIs) were prepared from highly contorted 2,2′-bis(3,4-dicarboxyphenoxy)biphenyl dianhydride and various trifluoromethyl-containing bis(ether amine)s via chemical or thermal imidization. All the PEIs showed good solubility in a variety of organic solvents and could afford flexible, colorless, and transparent thin films via solution-casting. They showed cut-off wavelengths of Uv-vis absorptions below 380 nm and very low yellowness index (b* < 7.0). They also showed high thermal stability with the 10% weight loss temperature from 503 to 585 °C of in nitrogen or air atmosphere. The glass transition temperatures (T gs) are recorded in the range of 194–254 °C by DSC. For a comparative study, another series of non-fluorinated PEIs derived from the same biphenyl-2,2′-diol bis(ether anhydride) with various bis(ether amine)s without the trifluoromethyl substituents were also synthesized and characterized.






Similar content being viewed by others
References
Wilson D, Stenzenberger HD, Hergenrother PM (eds) (1990) Polyimides. Blackie, New York
Sroog CE (1991) Prog Polym Sci 16:561
Ghosh MK, Mittal KL (eds) (1996) Polyimides: fundamentals and applications. Marcel Dekker, New York
Liou GS, Yen HJ (2012) Polyimides. In: Matyjaszewski K, Moller M (eds) Polymer science: a comprehensive reference. Vol. 5. pp. 497–535, Elsevier BV, Amsterdam
Liaw DJ, Wang KL, Huang YC, Lee KR, Lai JY, Ha CS (2012) Prog Polym Sci 37:907
Huang SJ, Hoyt AE (1995) Trends Polym Sci 3:262
de Abajo J, de la Campa JG (1999) Adv Polym Sci 140:23
Ando S, Matsuura T, Sasaki S (1997) Polym J 29:69
Hasegawa M, Horie K (2001) Prog Polym Sci 16:259
Takekoshi T (1987) Polym J 19:191
Hsiao SH, Yang CP, Fan JC (1994) J Polym Res 1:345
Tamai S, Yamaguchi A, Ohta M (1996) Polymer 37:3683
Eastmond GC, Paprotny J (1996) React Funct Polym 30:27
Chen G, Pei X, Fang X (2013) J Polym Res 20:159
Wirth JG In: High Performance Polymers: Their Origin and Development. Seymour RB, Krishenbaum GS (eds) (1986) Elesvier, Amsterdam
Yi L, Huang W, Yan D (2017) J Polym Sci Part A: Polym Chem 55:533
Hsiao SH, Yang CP, Chen SH (2000) J Polym Sci Part A: Polym Chem 38:1551
Eastmond GC, Paprotny J, Irwin RS (1996) Macromolecules 29:1382
Ding MX (2007) Prog Polym Sci 32:623
Yi L, Li C, Huang W, Yan D (2014) J Polym Res 21:572
Huang X, Mei M, Liu C, Pei X, Wei C (2015) J Polym Res 22:169
Li C, Yi L, Xu S, Wu X, Huang W, Yan D (2017) J Polym Res 24:7
Yang CP, Hsiao SH, Hsu MF (2002) J Polym Sci Part A: Polym Chem 40:524
Yang CP, Hsiao SH, Chen KH (2002) Polymer 43:5095
Chung CL, Yang CP, Hsiao SH (2006) J Polym Sci Part A: Polym Chem 44:3092
Yang CP, Chen YC, Hsiao SH, Guo WJ, Wang HM (2010) J Polym Res 17:779
Hsiao SH, Wang HM, Chen WJ, Lee TM, Leu CM (2011) J Polym Sci Part A: Polym Chem 49:3109
Liou GS (1998) J Polym Sci Part A: Polym Chem 36:1937
Liou GS, Maruyama M, Kakimoto MA, Imai Y (1998) J Polym Sci Part A: Polym Chem 36:2021
**e K, Zhang SY, Liu JG, He MH, Yang SY (2001) J Polym Sci Part A: Polym Chem 39:2581
Yang CP, Su YY, Wu KL (2004) J Polym Sci Part A: Polym Chem 42:5424
Yang CP, Hsiao FZ (2004) J Polym Sci Part A: Polym Chem 42:2272
Yang CP, Chiang HC (2004) Colloid Polym Sci 282:1347
Hsiao SH, Yang CP, Lin CK (1995) J Polym Res 2:1
Yang CP, Lin JH (1993) J Polym Sci Part A: Polym Chem 31:2153
Acknowledgements
Financial support from the Ministry of Science and Technology, Taiwan ROC is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Chen, YC., Hsiao, SH. & Su, YY. Organosoluble and colorless fluorinated poly(ether imide)s derived from a highly contorted biphenyl-2,2′-diol bis(ether anhydride) and aromatic bis(ether amine)s with trifluoromethyl substituents. J Polym Res 24, 87 (2017). https://doi.org/10.1007/s10965-017-1248-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10965-017-1248-x