Abstract
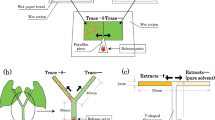
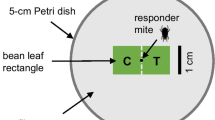
The western flower thrips is an omnivorous insect that consumes both leaf tissue and spider mite eggs. For this reason, these thrips are often described as ‘opportunistic predators’ of spider mites. Several studies have shown that western flower thrips are often found in association with spider mites, and the development time of thrips decreases and their survivorship increases when they consume spider mite eggs. We tested the hypothesis that thrips larvae may respond to chemical cues from spider mites, and that they may modify their prey-searching behavior when these spider mite-induced cues are present. We prepared hexane extracts from: 1) webbing of spider mites isolated from maize leaves, 2) webbing produced by spider mites inside an empty glass tube to exclude any plant-derived cues from the extract, and 3) spider mite cuticle extracts. These three extracts were subsequently applied in droplets to one-half of filter papers, and hexane alone was applied in droplets to the other half. We showed that residence time of thrips larvae was higher on filter paper with spider mite webbing extract, especially when the extract originated from spider mites isolated in glass tubes. In the presence of webbing extracts, we also observed: 1) a decrease in velocity, 2) an increase of angular velocity and 3) an increase in time immobile. Extracts from spider mite cuticle only increased velocity and proportion of time immobile of the thrips larvae. Our results suggest that chemical cues from spider mite webbing induce an arrestment response and play an important role in the non-random search behavior of thrips larvae searching for eggs on spider mite infested plant leaves.



Similar content being viewed by others
References
Agrawal AA, Kobayashi C, Thaler JS (1999) Influence of prey availability and induced plant resistance on omnivory by western flower thrips. Ecology 80:518–523. doi:10.1890/0012-9658(1999)080[0518%3AIOPAAI]2.0.CO%3B
Auger P, Migeon A, Ueckermann EA, Tiedt L, Navarro MN (2013) Evidence for synonymy between Tetranychus urticae and Tetranychus cinnabarinus (Acari, Prostigmata, Tetranychidae): review and new data. Acarologia 53:383–415. doi:10.1051/acarologia/2013XXXX
Bond AB (1980) Optimal foraging in a uniform habitat: the search mechanism of the green lacewing. Anim Behav 28:10–19. doi:10.1016/S0003-3472(80)80003-0
Clotuche G, Mailleux AC, Yano S, Detrain C, Deneubourg J-L, Hance T (2013) Settlement decisions by the two-spotted spider mite Tetranychus urticae. C R Biol 336:93–101. doi:10.1016/j.crvi.2013.02.006
Coll M, Guershon M (2002) Omnivory in terrestrial arthropods: mixing plant and prey diets. Annu Rev Entomol 47:267–297. doi:10.1146/annurev.ento.47.091201.145209
DeGraaf HE, Glenys MW (2009) An improved method for rearing western flower thrips Frankliniella occidentalis. Fla Entomol 92:664–666
Dennis RLH, Dapporto L, Fattorini S, Cook LM (2011) The generalism-specialism debate: the role of generalists in the life and death of species. Biol J Linn Soc 104:725–737. doi:10.1111/j.1095-8312.2011.01789.x
Hazan A, Gertler A, Tahori AS, Gerson U (1975) Spider mite webbing 3. Solubilization and amino-acid composition of silk protein. Comp Biochem Physiol B 51:457–462. doi:10.1016/0305-0491(75)90038-3
Hislop RG, Prokopy RJ (1981) Mite predator responses to prey and predator-emitted stimuli. J Chem Ecol 7:895–904. doi:10.1007/BF00992387
Hoffmann D, Vierheilig H, Schausberger P (2011) Arbuscular mycorrhiza enhances preference of ovipositing predatory mites for direct prey-related cues. Physiol Entomol 36:90–95. doi:10.1111/j.1365-3032.2010.00751.x
Kennedy JS (1978) The concepts of olfactory ‘arrestment’ and ‘attraction’. Physiol Entomol 3:91–98. doi:10.1111/j.1365-3032.1978.tb00138.x
Lincoln C, Williams FJ, Barnes G (1953) Importance of thrips in red spider control. J Econ Entomol 46:899–900
Loxdale HD, Lushai G, Harvey JA (2011) The evolutionary improbability of ‘generalism’ in nature, with special reference to insects. Biol J Linn Soc 103:1–18. doi:10.1111/j.1095-8312.2011.01627.x
Magalhães S, Janssen A, Montserrat M, Sabelis MW (2005) Host-plant species modifies the diet of an omnivore feeding on three trophic levels. Oikos 111:47–56. doi:10.1111/j.0030-1299.2005.13897.x
Martini X, Kincy N, Vaughn K, Dever J, Nansen C (2013) Positive association between thrips and spider mites in seedling cotton. Agric For Entomol 15:197–203. doi:10.1111/afe.12004
Mayland H, Margolies DC, Charlton RE (2000) Local and distant prey-related cues influence when an acarine predator leaves a prey patch. Entomol Exp Appl 96:245–252. doi:10.1046/j.1570-7458.2000.00703.x/full
Milne M, Walter G (1997) The significance of prey in the diet of the phytophagous thrips, Frankliniella schultzei. Ecol Entomol 22:74–81. doi:10.1046/j.1365-2311.1997.00034.x/full
Mound LA (2005) Thysanoptera: diversity and interactions. Annu Rev Entomol 50:247–269. doi:10.1146/annurev.ento.49.061802.123318
Pallini A, Janssen A, Sabelis MW (1998) Predators induce interspecific herbivore competition for food in refuge space. Ecol Lett 1:171–177
Pallini A, Janssen A, Sabelis MW (1999) Do western flower thrips avoid plants infested with spider mites? Interactions between potential competitors. In: Bruin J, van der Geest LPS, Sabelis MW (eds) Ecology and evolution of the Acari. Springer, Berlin, pp 375–380
Penman DR, Cone WW (1974) Role of web, tactile stimuli, and female sex pheromone in attraction of male twospotted spider mites to quiescent female deutonymphs. Ann Entomol Soc Am 67:179–182
Pickett CH, Wilson LT, Gonzalez D (1988) Population dynamic and within plant distribution of the western flower thrips (Thysanoptera, Thripidae), an early season predator of spider mites infesting cotton. Environ Entomol 17:551–559
Roda Nyrop J, English-Loeb G, Dicke M (2001) Leaf pubescence and two-spotted spider mite webbing influence phytoseiid behavior and population density. Oecologia 129:551–560. doi:10.1007/s004420100762
Sabelis MW, Bakker FM (1992) How predatory mites cope with the web of their tetranychid prey – a functional view on dorsal chaetotaxy in the phytoseiidae. Exp Appl Acarol 16:203–225. doi:10.1007/BF01193804
Shimoda T, Takabayashi J, Ashihara W, Takafuji A (1997) Response of predatory insect Scolothrips takahashii toward herbivore-induced volatiles under laboratory and field conditions. J Chem Ecol 23:2033–2048. doi:10.1023/B:JOEC.0000006487.49221.df
Silva R, Furlong MJ, Wilson LJ, Walter GH (2013) How predictable are the behavioural responses of insects to herbivore induced changes in plants? Responses of two congeneric thrips to induced cotton plants. PLoS ONE 8:e63611. doi:10.1371/journal.pone.0063611
Stephen DW, Krebs JR (1986) Foraging theory. Princeton University Press, Princeton
Trichilo PJ, Leigh TF (1986) Predation on spider mite eggs by the western flower thrips, Frankliniella occidentalis (Thysanoptera: Thripidae), an opportunist in a cotton agroecosystem. Environ Entomol 15:821–825
Waage JK (1978) Arrestment responses of parasitoid, Nemeritis canescens, to a contact chemical produced by its host, Plodia interpunctella. Physiol Entomol 3:135–146. doi:10.1111/j.1365-3032.1978.tb00143.x
Wilson LT, Trichilo PJ, Gonzalez D (1991) Natural enemies of spider mites (Acari: Tetranychidae) on cotton: density regulation or casual association? Environ Entomol 20:849–856
Wilson LJ, Bauer LR, Walter GH (1996) Phytophagous thrips are facultative predators of twospotted spider mites (Acari: Teranychidae) on cotton in Australia. Bull Entomol Res 86:297–305. doi:10.1006/anbe.2000.1483
Zhi JR, Margolies DC, Nechols JR, Boyer JE (2006) Host-plant-mediated interaction between populations of a true omnivore and its herbivorous prey. Entomol Exp Appl 121:59–66. doi:10.1111/j.1570-8703.2006.00456.x
Acknowledgments
We thank Texas State Support Committee and Cotton Incorporated for financial support of this project. We thank Wendy Meyer, Sean Michael Prager, and two anonymous reviewers for their valuable comments on earlier versions of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Martini, X., Guvvala, H. & Nansen, C. The Search Behavior of Omnivorous Thrips Larvae is Influenced by Spider Mite Cues. J Insect Behav 28, 593–603 (2015). https://doi.org/10.1007/s10905-015-9527-z
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10905-015-9527-z