Abstract
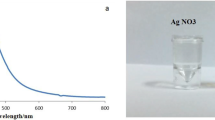
The present study was aimed to evaluate the antifeedant, larvicidal, pupicidal, biochemical effects of Vernonia anthelmintica seeds mediated AgNPs against Spodoptera litura, Helicoverpa armigera, Culex quinquefasciatus and Aedes aegypti. The potential antifeedant activity of synthesized AgNPs were 86.90% and 89.83% and larvicidal activity of (LC50) 56.42 μg/mL and 63.65 μg/mL against S. litura and H. armigera respectively. Furthermore, larval growth duration was increased as 13.66 and 15.85 days on S. litura and H. armigera as compared to control 7.83 and 8.05 days. The AgNPs caused larvicidal activity of (LC50) 45.14 µg/mL and 53.23 µg/mL against Ae. aegypti and Cx. quinquefasciatus. The increased α esterase level of 85.00, 85.33 0.49 and 0.53 μg napthol produced/min/mg larval protein and β esterase level of 92.66, 101.67, 0.53 and 0.48 μg napthol produced/min/mg larval protein and glutathione S-transferase enzyme level of 146.33, 160.00, 0.75 and 0.65 μmol/min/mg larval proteins on S. litura, H. armigera, Ae. aegypti and Cx. quinquefasciatus. The damaged midget cells and peritrophic membrane was observed on treated larvae of S. litura, H. armigera, Ae. aegypti and Cx. quinquefasciatus. Conclusively, biosynthesised of AgNPs using V. anthelmintica seeds were used as potential biopesticide for controlling S. litura and H. armigera, Ae. aegypti and Cx. quinquefasciatus.
Graphic Abstract








Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this manuscript.
References
A. Tuerxuntayi, Y. Q. Liu, A. Tulake, M. Kabas, A. Eblimit, and H. A. Aisa (2014). Kaliziri extract upregulates tyrosinase, TRP-1, TRP-2 and MITF expression in murine B16 melanoma cells. BMC Complement. Altern. Med. 14, 166.
D. Maulina, S. B. Sumitro, M. Amin, and S. R. Lestari (2018). Identification of bioactive compounds from Mirabilis jalapa L. (Caryophyllales: Nyctaginaceae) extract as biopesticides and their activity against the immune response of Spodoptera litura F. (Lepidoptera: Noctudiae). J. Biopestic. 11 (2), 89–97.
J. P. Cunningham and M. P. Zalucki (2014). Understanding heliothine (Lepidoptera: Heliothinae) pests: what is a host plant? J. Econ. Entomol. 107 (3), 881–896.
S. K. Yadav and S. Patel (2017). Bioactivity of some plant extracts against larvae of Spodoptera litura (Fab.) and Athalia proxima lugens (Klug.) under laboratory conditions. J. Entomol. Zool. Stud. 5 (2), 1430–1433.
A. Jeyasankar, T. Chinnamani, V. Chennaiyan, and G. Ramar (2014). Antifeedant activity of Barleria buxifolia (Linn.) (Acanthaceae) against Spodoptera litura Fabricius and Helicoverpa armigera Hübner (Lepidotera: Noctuidae). Int. J. Nat. Sci. Res. 2 (5), 78–84.
B. D. Lade, D. P. Gogle, and S. B. Nandeshwar (2017). Nano Bio Pesticide to constraint plant destructive pests. J. Nanomed. Res. 6 (3), 1–9.
R. Maheswaran, K. Baskar, S. Ignacimuthu, S. Maria Packiam, and K. Rajapandiyan (2019). Bioactivity of Couroupita guianensis Aubl. against filarial and dengue vectors and non-target fish. S. Afr. J. Bot. 125, 46–53.
U. Muthukumaran, M. Govindarajan, and M. Rajeswary (2015). Mosquito larvicidal potential of silver nanoparticles synthesized using Chomelia asiatica (Rubiaceae) against Anopheles stephensi, Aedes aegypti, and Culex quinquefasciatus (Diptera: Culicidae). Parasitol. Res. 114 (3), 989–999.
World Health Organization, Global Strategy for Dengue Prevention and Control 2012–2020. (World Health Organization, Geneva, 2012)
P. A. C. Kamatchi, R. Maheswaran, and S. Ignacimuthu (2016). Evaluation of larval toxicity of Lantana camara L. and Catharanthus roseus L. against Culex quinquefasciatus Say and Aedes Aegypti L. Entomol. Ornithol. Herpetol. 2016, 5–1.
G. B. Hosamani, R. R. Patil, V. I. Benagi, S. S. Chandrashekhar, and B. S. Nandihali (2019). Synthesis of green silver nanoparticles from soybean seed and its bioefficacy on Spodoptera litura (F.). Int. J. Curr. Microbiol. Appl. Sci. 8 (9), 610–618.
R. Maheswaran, S. Sathish, and S. Ignacimuthu (2008). Larvicidal activity of Leucas aspera (Willd.) against the larvae of Culex quinquefasciatus Say and Aedes aegypti L. Int. J. Integr. Biol. 2, 214–217.
R. Maheswaran, S. Sukumaran, G. Nattudurai, and S. Ignacimuthu (2016). Bioefficacy of essential oil from Toddalia asiatica (L.) Lam. against dengue vector mosquitoes Aedes aegypti L. and Aedes albopictus Skuse. Indian J. Nat. Prod. Resour. 7 (3), 245–251.
R. Maheswaran and S. Ignacimuthu (2014). Effect of Polygonum hydropiper L. against dengue vector mosquito Aedes albopictus L. Parasitol. Res. 113, 3143–3150.
S. Sukumaran and R. Maheswaran (2020). Larvicidal activity of Elytraria acaulis against Culex quinquefasciatus and Aedes aegypti (Diptera: Culicidae). J. Arthropod Borne Dis. 14 (3), 293–301.
R. Maheswaran and S. Ignacimuthu (2012). A novel herbal formulation against dengue vector mosquitoes Aedes aegypti and Aedes albopictus. Parasitol. Res. 110 (5), 1801–1013.
R. Maheswaran and S. Ignacimuthu (2013). Bioefficacy of essential oil from Polygonum hydropiper L. against mosquitoes, Anopheles stephensi and Culex quinquefasciatus. J. Ecotoxicol. Environ. Saf. 97, 26–31.
A. Bhattacharyya, A. Bhaumik, P. U. Rani, S. Mandal, and T. T. Epidi (2010). Nano-particles—a recent approach to insect pest control. Afr. J. Biotechnol. 9 (24), 3489–3493.
K. Shahzad and F. Manzoor (2019). Nanoformulations and their mode of action in insects: a review of biological interactions. Drug Chem. Toxicol. 44 (1), 1–11.
J. L. Elechiguerra, J. L. Burt, J. R. Morones, A. Camacho-Bragado, X. Gao, H. H. Lara, and M. J. Yacaman (2005). Interaction of silver nanoparticles with HIV-1. J. Nanobiotechnol. 3 (1), 1–10.
M. Rouhani, M. AminSamih, and S. Kalantari (2012). Efecto insecticida de nanopartículas de plata y zinc contra Aphis nerii Boyer de Fonscolombe (Hemiptera: Aphididae). Chil. J. Agric. Res. 72 (4), 590–594.
P. Kuppusamy, M. M. Yusoff, G. P. Maniam, and N. Govindan (2016). Biosynthesis of metallic nanoparticles using plant derivatives and their new avenues in pharmacological applications—an updated report. Saudi Pharm. J. 24 (4), 473–484.
T. Ito, S. Aimaiti, N. N. Win, T. Kodama, and H. Morita (2016). New sesquiterpene lactones, vernonilides A and B, from the seeds of Vernonia anthelmintica in Uyghur and their antiproliferative activities. Bioorg. Med. Chem. Lett. 26 (15), 3608–3611.
R. Srivastava, A. Verma, A. Mukerjee, and N. Soni (2014). Phytochemical, pharmacological and pharmacognostical profile of Vernonia anthelmintica: an overview. Res. Rev.: J. Pharm. Phytochem. 2 (2), 22–28.
K. V. Otari, R. V. Shete, C. D. Upasani, V. S. Adak, M. Y. Bagade, and A. N. Harpalani (2010). Evaluation of anti-inflammatory and anti-arthritic activities of ethanolic extract of Vernonia anthelmintica seeds. J. Cell Tissue Res. 10 (2), 2269–3228.
J. Parekh and S. Chanda (2008). Antibacterial activity of aqueous and alcoholic extracts of 34 Indian medicinal plants against some Staphylococcus species. Turk. J. Biol. 32, 63–71.
S. F. Shaik, D. R. Maddirala, V. K. Kondeti, T. S. K. Mekala, R. B. Kasetti, and C. A. Rao (2010). Antidiabetic and anti-hyperlipidemic activity of ethyl acetate: isopropanol (1:1) fraction of Vernonia anthelmintica seeds in Streptozotocin induced diabetic rats. Food Chem. Toxicol. 48, 495–501.
N. K. Dogra, S. Kumar, and D. Kumar (2020). Vernonia anthelmintica (L.) Willd.: an ethnomedicinal, phytochemical, pharmacological and toxicological review. J. Ethnopharmacol. 28, 112777.
H. Lei, L. Ya, W. Fei, F. L. Dan, and G. Kun (2012). Biologically active steroids from the aerial parts of Vernonia anthelmintica Will. Fitoterapia 02459, 1–6.
L. Yongqiang, E. N. Alfarius, Y. Hirasawa, A. Nakata, T. Kaneda, N. Uchiyama, Y. Goda, O. Shirota, H. Morita, and H. A. Aisa (2010). Vernodalidimers A and B, novel orthoester elemanolide dimers from seeds of Vernonia anthelmintica. Tetrahedron Lett. 51, 6584–6587.
N. Jahan, M. Ahmad, F. Mehjabeen Saeed, A. B. Amber Rehman, and S. Muhammad (2014). Anti-nociceptive activity of seed extract of Vernonia anthelmintica willd. Pak. J. Pharm. Sci. 27 (6), 2177–2181.
T. Guilian, Z. Ubin, Z. Tianyou, Y. Fuquan, and I. Yoichiro (2004). Separation of flavonoids from the seeds of Vernonia anthelmintica Willd. by high-speed counter-current chromatography. J. Chromatogr. A 1049, 219–222.
K. Baskar, R. Maheshwaran, S. Kingsley, and S. Ignacimuthu (2011). Bioefficacy of plant extracts against Asian army worm Spodoptera litura Fab. (Lepidoptera: Noctuidae). J. Agric. Technol. 7 (1), 123–131.
E. S. Vanderzant, C. D. Richardson, and S. W. Fort Jr. (1962). Rearing of the bollworm on artificial diet. J. Econ. Entomol. 55, 140–140.
R. Maheswaran and S. Ignacimuthu (2015). Effect of confertifolin from Polygonum hydropiper L. against dengue vector mosquitoes Aedes aegypti L. Environ. Sci. Pollut. Res. 22 (11), 8280–8287.
R. Maheswaran and S. Ignacimuthu (2015). A novel biopesticide Ponneem to control human vector mosquitoes Anopheles stephensi L. and Culex quinquefasciatus Say. Environ. Sci. Pollut. Res. 22 (17), 13153–13166.
R. P. Singh and N. C. Pant (1980). Lycorine—a resistance factor in the plant subfamily Amarylloidiodeae against desert locust. Experientia 36, 552–555.
W. S. Abbott (1925). A method of computing the effectiveness of insecticides. J. Econ. Entomol. 18, 265–267.
WHO, Guidelines for Laboratory and Field Testing of Mosquito Larvicides, WHO/CDS/WHOPES/GCDPP/2005.13 (WHO, Geneva, 2005)
J. Matowo, M. A. Kulkarni, F. W. Mosha, R. M. Oxborough, J. A. Kitau, F. Tenu, and M. Rowland (2010). Biochemical basis of permethrin resistance in Anopheles arabiensis from Lower Moshi, north-eastern Tanzania. Malar. J. 9, 193.
W. H. Habig, M. J. Pabst, and W. B. Jacoby (1974). Glutathione S-transferases: the first enzymatic step in mercapturic acid formation. J. Biol. Chem. 249, 7130–7139.
A. Dinesh Kumar, E. Srimaan, M. Chellappandian, P. Vasantha Srinivasan, S. Karthi, A. Thanigaivel, and K. Kalaivani (2018). Target and non-target response of Swietenia mahagoni Jacq. chemical constituents against tobacco cutworm Spodoptera litura Fab. and earthworm Eudrilus eugeniae Kinb. Chemosphere 199, 35–43.
SPSS, IBM SPSS Statistics Version 21. (International Business Machines Corp, Boston, 2012), p. 126.
D. J. Finney, Probit Analysis (Cambridge University Press, Cambridge, 1971).
S. B. Santhosh, R. Yuvarajan, and D. Natarajan (2015). Annona muricata leaf extract-mediated silver nanoparticles synthesis and its larvicidal potential against dengue, malaria and filariasis vector. Parasitol. Res. 114 (8), 3087–3096.
M. Q. Nasar, T. Zohra, A. T. Khalil, S. Saqib, M. Ayaz, A. Ahmad, and Z. K. Shinwari (2019). Seripheidium quettense mediated green synthesis of biogenic silver nanoparticles and their theranostic applications. Green Chem. Lett. Rev. 12 (3), 310–322.
R. S. Bhat, J. Almusallam, S. Al Daihan, and A. Al-Dbass (2019). Biosynthesis of silver nanoparticles using Azadirachta indica leaves: characterisation and impact on Staphylococcus aureus growth and glutathione-S-transferase activity IET. Nanobiotechnology 13 (5), 498–502.
J. P. Ruparelia, A. K. Chatterjee, S. P. Duttagupta, and S. Mukherji (2008). Strain specificity in antimicrobial activity of silver and copper nanoparticles. Acta Biomater. 4 (3), 707–716.
P. U. Rani, K. P. Laxmi, V. Vadlapudi, and B. Sreedhar (2016). Phytofabrication of silver nanoparticles using the mangrove associate, Hibiscus tiliaceus plant and its biological activity against certain insect and microbial pests. J. Biopestic. 9 (2), 167.
F. Jalilian, A. Chahardoli, K. Sadrjavadi, A. Fattahi, and Y. Shokoohinia (2020). Green synthesized silver nanoparticle from Allium ampeloprasum aqueous extract: characterization, antioxidant activities, antibacterial and cytotoxicity effects. Adv. Powder Technol. 31 (3), 1323–1332.
P. U. Rani, J. Yasur, K. S. Loke, and D. Dutta (2016). Effect of synthetic and biosynthesized silver nanoparticles on growth, physiology and oxidative stress of water hyacinth: Eichhornia crassipes (Mart) Solms. Acta Physiol. Plant. 38 (2), 58.
T. Rajkumar, A. Sapi, G. Das, T. Debnath, A. Ansari, and J. K. Patra (2019). Biosynthesis of silver nanoparticle using extract of Zea mays (corn flour) and investigation of its cytotoxicity effect and radical scavenging potential. J. Photochem. Photobiol. B 193, 1–7.
K. Murugan, M. Roni, C. Panneerselvam, U. Suresh, R. Rajaganesh, R. Aruliah, and S. Kumar (2017). Sargassum wightii-synthesized ZnO nanoparticles reduce the fitness and reproduction of the malaria vector Anopheles stephensi and cotton bollworm Helicoverpa armigera. Physiol. Mol. Plant Pathol. 101, 202–213.
A. A. Kajani, A. K. Bordbar, S. H. Z. Esfahani, A. R. Khosropour, and A. Razmjou (2014). Green synthesis of anisotropic silver nanoparticles with potent anticancer activity using Taxus baccata extract. RSC Adv. 4 (106), 61394–61403.
S. Mukherji, J. Ruparelia, and S. Agnihotri, Antimicrobial activity of silver and copper nanoparticles: variation in sensitivity across various strains of bacteria and fungi, in Nano-antimicrobials (Springer, Berlin, 2012), pp. 225–251.
P. Das, K. Ghosal, N. K. Jana, A. Mukherjee, and P. Basak (2019). Green synthesis and characterization of silver nanoparticles using belladonna mother tincture and its efficacy as a potential antibacterial and anti-inflammatory agent. Mater. Chem. Phys. 228, 310–317.
M. Anandan, G. Poorani, P. Boomi, K. Varunkumar, K. Anand, A. A. Chuturgoon, and H. G. Prabu (2019). Green synthesis of anisotropic silver nanoparticles from the aqueous leaf extract of Dodonaea viscosa with their antibacterial and anticancer activities. Process Biochem. 80, 80–88.
R. S. A. Bharani and S. K. R. Namasivayam (2017). Biogenic silver nanoparticles mediated stress on developmental period and gut physiology of major lepidopteran pest Spodoptera litura (Fab.) (Lepidoptera: Noctuidae)—an eco-friendly approach of insect pest control. J. Environ. Chem. Eng. 5 (1), 453–467.
G. S. Jadhav, A. A. Devarshi, and S. R. Yankanchi (2016). Efficacy of certain Clerodendrum leaf crude extracts against cutworm, Spodoptera litura Fab. and cotton bollworm Helicoverpa armigera Hub. J. Entomol. Zool. 4 (4), 466–472.
A. Ponsankar, K. Sahayaraj, S. Senthil-Nathan, P. Vasantha-Srinivasan, S. Karthi, A. Thanigaivel, and W. B. Hunter (2019). Toxicity and developmental effect of cucurbitacin E from Citrullus colocynthis L. (Cucurbitales: Cucurbitaceae) against Spodoptera litura Fab. and a non-target earthworm Eisenia fetida Savigny. Environ. Sci. Pollut. Res. 27 (19), 23390–23401.
M. Gabriel Paulraj, N. Shanmugam, and S. Ignacimuthu (2014). Antifeedant activity and toxicity of two alkaloids from Adhatoda vasica Nees leaves against diamondback moth Plutella xylostella (Linn.) (Lepidoptera: Plutellidae) larvae. Arch. Phytopathol. Plant Prot. 47 (15), 1832–1840.
S. Kantrao, M. A. Ravindra, S. M. D. Akbar, P. Jayanthi, and A. Venkataraman (2017). Effect of biosynthesized silver nanoparticles on growth and development of Helicoverpa armigera (Lepidoptera: Noctuidae): interaction with midgut protease. J. Asia Pac. Entomol. 20 (2), 583–589.
J. Ravikumar, P. Samuthiravelu, S. M. H. Qadri, L. Hemanthkumar, and S. Jayaraj (2010). Integrated Pest Management (IPM) module for Tukra mealy bug, Maconellicoccus hirsutus (Green) and leaf webber, Diaphania pulverulentalis (Hamp.) in mulberry. J. Biopestic. 3 (1), 354.
S. Marimuthu, A. A. Rahuman, G. Rajakumar, T. Santhoshkumar, A. V. Kirthi, C. Jayaseelan, and C. Kamaraj (2011). Evaluation of green synthesized silver nanoparticles against parasites. Parasitol. Res. 108 (6), 1541–1549.
T. Manimegalai, K. Raguvaran, M. Kalpana, and R. Maheswaran (2020). Green synthesis of silver nanoparticle using Leonotis nepetifolia and their toxicity against vector mosquitoes of Aedes aegypti and Culex quinquefasciatus and agricultural pests of Spodoptera litura and Helicoverpa armigera. Environ. Sci. Pollut. Res. 27–34, 43103–43116.
S. K. Mirhaghparast, A. Zibaee, J. Hajizadeh, and S. Ramzi (2020). Toxicity and physiological effects of the tea seed saponin on Helicoverpa armigera. Biocatal. Agric. Biotechnol. 25, 101597.
M. Roni, K. Murugan, C. Panneerselvam, J. Subramaniam, M. Nicoletti, P. Madhiyazhagan, D. Dinesh, U. Suresh, H. F. Khater, H. Wei, A. Canale, A. A. Alarfaj, M. A. Munusamy, A. Higuchi, and G. Benelli (2015). Characterization and biotoxicity of Hypnea musciformis-synthesized silver nanoparticles as potential eco-friendly control tool against Aedes aegypti and Plutella xylostella. Ecotoxicol. Environ. Saf. 121, 31–38.
G. Benelli (2016). Plant-mediated biosynthesis of nanoparticles as an emerging tool against mosquitoes of medical and veterinary importance: a review. Parasitol. Res. 115 (1), 23–34.
A. S. Kandagal and M. C. Khetagoudar (2013). Study on larvicidal activity of weed extracts against Spodoptera litura. J. Environ. Biol. 34 (2), 253–257.
S. Kanthammal, A. Jebanesan, K. Kovendan, J. Subramaniam, and M. Vijay (2018). Novel insecticides of Syzygium cumini fabricated silver nanoparticles against filariasis, malaria, and dengue vector mosquitoes. Int. J. Mosq. Res. 5 (5), 95–106.
M. Baranitharan, S. Alarifi, S. Alkahtani, D. Ali, K. Elumalai, J. Pandiyan, K. Krishnappa, R. Mohan, and M. Govindarajan (2020). Phytochemical analysis and fabrication of silver nanoparticles using Acacia catechu: an efficacious and ecofriendly control tool against selected polyphagous insect pests. Saudi J. Biol. Sci. 28 (1), 148–156.
U. Suresh, K. Murugan, C. Panneerselvam, R. Rajaganesh, M. Roni, A. T. Aziz, H. A. N. Al-Aoh, S. Trivedi, H. Rehman, S. Kumar, A. Higuchi, A. Canale, and G. Benelli (2018). Suaeda maritima-based herbal coils and green nanoparticles as potential biopesticides against the dengue vector Aedes aegypti and the tobacco cutworm Spodoptera litura. Biocatal. Agric. Biotechnol. 101, 225–235.
A. A. Buhroo, G. Nisa, S. Asrafuzzaman, R. Prasad, R. Rasheed, and A. Bhattacharyya (2017). Biogenic silver nanoparticles from Trichodesma indicum aqueous leaf extract against Mythimna separata and evaluation of its larvicidal efficacy. J. Plant Prot. Res. 57 (2), 194–200.
J. Yasur and P. U. Rani (2015). Lepidopteran insect susceptibility to silver nanoparticles and measurement of changes in their growth, development and physiology. Chemosphere 124, 92–102.
H. A. Ammar and E. M. Abd-ElAzeem (2020). Novel treatment of gelatin-copper bio-nanoparticles as a management method against the spiny bollworm, Earias insulana, (Boisd.) (Lepidoptera: Noctuidae) in comparison studies with the uncoated nanoparticles. Inorg. Nano-Met. Chem. https://doi.org/10.1080/24701556.2020.1786403.
K. Baskar, R. Maheswaran, and S. Ignacimuthu (2012). Bioefficacy of Ceasalpinia bonduc (L.) Roxb. against Spodoptera litura Fab. (Lepidoptera: Noctuidae). Arch. Phytopathol. Plant Prot. 45 (10), 1127–1137.
K. Baskar, R. Maheswaran, M. Pavunraj, S. M. Packiam, S. Ignacimuthu, V. Duraipandiyan, and G. Benelli (2018). Toxicity and antifeedant activity of Caesalpinia bonduc (L.) Roxb. (Caesalpiniaceae) extracts and fractions against the cotton bollworm Helicoverpa armigera Hub. (Lepidoptera: Noctuidae). Physiol. Mol. Plant Pathol. 101, 69–74.
K. Balaraju, R. Maheswaran, P. Agastian, and S. Ignacimuthu (2009). Egg hatchability and larvicidal activity of Swertia chirata Buch.-Hams. ex Wall. against Aedes aegypti L. and Culex quinquefasciatus Say. Indian J. Sci. Technol. 2, 46–49.
L. D. Amarasinghe, P. A. S. R. Wickramarachchi, A. A. A. U. Aberathna, W. S. Sithara, and C. R. De Silva (2020). Comparative study on larvicidal activity of green synthesized silver nanoparticles and Annona glabra (Annonaceae) aqueous extract to control Aedes aegypti and Aedes albopictus (Diptera: Culicidae). Heliyon 6 (6), 04322.
R. Ishwarya, B. Vaseeharan, R. Anuradha, R. Rekha, M. Govindarajan, N. S. Alharbi, S. Kadaikunnan, J. M. Khaled, and G. Benelli (2017). Eco-friendly fabrication of Ag nanostructures using the seed extract of Pedalium murex, an ancient Indian medicinal plant: histopathological effects on the Zika virus vector Aedes aegypti and inhibition of biofilm-forming pathogenic bacteria. J. Photochem. Photobiol. B 174, 133–143.
M. Ahamed, M. Karns, M. Goodson, J. Rowe, S. M. Hussain, J. J. Schlager, and Y. Hong (2008). DNA damage response to different surface chemistry of silver nanoparticles in mammalian cells. Toxicol. Appl. Pharmacol. 233 (3), 404–410.
K. Murugan, C. Panneerselvam, C. M. Samidoss, P. Madhiyazhagan, U. Suresh, M. Roni, B. Chandramohan, J. Subramaniam, D. Dinesh, R. Rajaganesh, M. Paulpandi, H. Wei, A. T. Aziz, M. S. Alsalhi, S. Devanesan, M. Nicoletti, R. Pavela, A. Canale, and G. Benelli (2016). In vivo and in vitro effectiveness of Azadirachta indica-synthesized silver nanocrystals against Plasmodium berghei and Plasmodium falciparum, and their potential against malaria mosquitoes. Res. Vet. Sci. 106, 14–22.
B. Morejón, F. Pilaquinga, F. Domenech, D. Ganchala, A. Debut, and M. Neira (2018). Larvicidal activity of silver nanoparticles synthesized using extracts of Ambrosia arborescens (Asteraceae) to control Aedes aegypti L. (Diptera: Culicidae). J. Nanotechnol. https://doi.org/10.1155/2018/6917938.
N. Sutthanont, S. Attrapadung, and S. Nuchprayoon (2019). Larvicidal activity of synthesized silver nanoparticles from Curcuma zedoaria essential oil against Culex quinquefasciatus. Insects 10 (1), 27. https://doi.org/10.3390/insects10010027.
K. Kovendan, S. Arivoli, R. Maheshwaran, K. Baskar, and S. Vincent (2012). Larvicidal efficacy of Sphaeranthus indicus, Cleistanthus collinus and Murraya koenigii leaf extracts against filarial vector, Culex quinquefasciatus Say (Diptera: Culicidae). Parasitol. Res. 111 (3), 1025–1035.
K. Chinnaperumal, B. Govindasamy, D. Paramasivam, A. Dilipkumar, A. Dhayalan, A. Vadivel, K. Sengodan, and P. Pachiappan (2018). Bio-pesticidal effects of Trichoderma viride formulated titanium dioxide nanoparticle and their physiological and biochemical changes on Helicoverpa armigera (Hub.). Pestic. Biochem. Phys. 149, 26–36.
E. Parthiban, M. Ramachandran, M. Jayakumar, and R. Ramanibai (2019). Biocompatible green synthesized silver nanoparticles impact on insecticides resistant develo** enzymes of dengue transmitted mosquito vector. SN Appl. Sci. 1 (10), 1282.
V. Sujitha, K. Murugan, D. Dinesh, A. Pandiyan, R. Aruliah, J. S. Hwang, K. Kalimuthu, C. Panneerselvam, A. Higuchi, S. Kumar, and A. A. Alarfaj (2017). Green-synthesized CdS nano-pesticides: toxicity on young instars of malaria vectors and impact on enzymatic activities of the non-target mud crab Scylla serrata. Aquat. Toxicol. 188, 100–108.
P. Manjula, K. Lalitha, G. Vengateswari, J. Patil, S. S. Nathan, and M. S. Shivakumar (2020). Effect of Manihot esculenta (Crantz) leaf extracts on antioxidant and immune system of Spodoptera litura (Lepidoptera: Noctuidae). Biocatal. Agric. Biotechnol. 23, 101476.
M. Bilal, C. Xu, L. Cao, P. Zhao, C. Cao, F. Li, and Q. Huang (2020). Indoxacarb-loaded fluorescent mesoporous silica nanoparticles for effective control of Plutella xylostella L. with decreased detoxification enzymes activities. Pest Manag. Sci. https://doi.org/10.1002/ps.5924.
D. Stone, P. Jepson, and R. Laskowski (2002). Trends in detoxification enzymes and heavy metal accumulation in ground beetles (Coleoptera: Carabidae) inhabiting a gradient of pollution. Comp. Biochem. Physiol. C 132 (1), 105–112.
E. Zvereva, V. Serebrov, V. Glupov, and I. Dubovskiy (2003). Activity and heavy metal resistance of non-specific esterases in leaf beetle Chrysomela lapponica from polluted and unpolluted habitats. Comp. Biochem. Physiol. C 135, 383–391.
M. Fiaz, L. C. Martínez, Costa M. Da Silva, J. F. S. Cossolin, A. Plata-Rueda, W. G. Gonçalves, A. E. G. Sant’Ana, J. C. Zanuncio, and J. E. Serrão (2018). Squamocin induce histological and ultrastructural changes in the midgut cells of Anticarsia gemmatalis (Lepidoptera: Noctuidae). Ecotoxicol. Environ. Saf. 156, 1–8.
K. Kalimuthu, C. Panneerselvam, C. Chou, L. C. Tseng, K. Murugan, K. H. Tsai, A. A. Alarfaj, A. Higuchi, A. Canale, J. S. Hwang, and G. Benelli (2017). Control of dengue and Zika virus vector Aedes aegypti using the predatory copepod Megacyclops formosanus: synergy with Hedychium coronarium-synthesized silver nanoparticles and related histological changes in targeted mosquitoes. Process Saf. Environ. 109, 82–96.
Acknowledgements
The author (Dr. Rajan Maheswaran/Principal Investigator/UGC Major Research Project, File No. MRP-MAJOR-ZOOL-2013-21921) is thankful to the University Grants Commission (UGC), New Delhi, India for providing financial support and Periyar University, Salem, Tamil Nadu, India for providing laboratory support.
Author information
Authors and Affiliations
Contributions
TM and KR have contributed equally to this work (Investigation, Writing—original draft). MK—Review & editing, RM: Conceptualization, Supervision.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interests regarding the publication of this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Manimegalai, T., Raguvaran, K., Kalpana, M. et al. Facile Synthesis of Silver Nanoparticles Using Vernonia anthelmintica (L.) Willd. and Their Toxicity Against Spodoptera litura (Fab.), Helicoverpa armigera (Hüb.), Aedes aegypti Linn. and Culex quinquefasciatus Say. J Clust Sci 33, 2287–2303 (2022). https://doi.org/10.1007/s10876-021-02151-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-021-02151-z