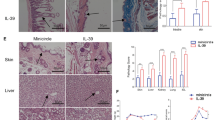
Palifermin (recombinant human keratinocyte growth factor) prevents the development of acute, lethal graft-versus-host disease (GVHD). It does so, at least in part, by protecting cells from injury. Another property of Palifermin is immune regulation. How the latter influences the evolution of GVHD remains uncertain. We explored the effect of Palifermin on GVHD in the DBA/2 → ((DBA/2)×(C57BL/6))F1-hybrid strain combination, a model associated with autoantibody production and glomerulonephritis. Untreated recipients survived until at least day 150 post-induction. Palifermin-treated recipients succumbed between days 50 and 90 with levels of proteinuria of up to 20 g/L, ascites, and rapidly progressive, crescentic glomerulonephritis that was most severe in mice with the greatest levels of proteinuria. Kidney sections from both Palifermin-treated and untreated recipients showed the presence of granular deposits of IgG, IgM, IgA, and C3 in the mesangium and the glomerular basement membrane. Electron microscopy confirmed the extensive glomerular immune complex deposition. Antinuclear and anti-dsDNA antibodies were present in sera from both treated and untreated recipients; however, those in the latter were only detectable if the serum was kept at 37°C, indicating that they were cryoglobulins. IL-4 was detectable only in cultures from Palifermin-treated recipients and the levels of IL-5 and IL-13 were significantly higher in the Palifermin-treated group than in untreated GVHD mice. IFN-γ was only detectable in untreated GVHD mice. These data suggest that although Palifermin can protect mice with acute GVHD, it exacerbates GVHD in a model associated with autoantibody production and a preponderance of Th2 cytokines.




Similar content being viewed by others
REFERENCES
Rus V, Svetic A, Nguyen P, Gause WC, Via CS: Kinetics of Th1 and Th2 cytokine production during the early course of acute and chronic murine graft-versus-host disease. Regulatory role of donor CD8+ T cells. J Immunol 155(5):2396–2406, 1995
Panoskaltsis-Mortari A, Lacey DL, Vallera DA, Blazar BR: Keratinocyte growth factor administered before conditioning ameliorates graft-versus-host disease after allogeneic bone marrow transplantation in mice. Blood 92(10):3960–3967, 1998
Krijanovski OI, Hill GR, Cooke KR, Teshima T, Crawford JM, Brinson YS, et al.: Keratinocyte growth factor separates graft-versus-leukemia effects from graft-versus-host disease. Blood 94(2):825–831, 1999
Panoskaltsis-Mortari A, Taylor PA, Rubin JS, Uren A, Welniak LA, Murphy WJ, et al.: Keratinocyte growth factor facilitates alloengraftment and ameliorates graft-versus-host disease in mice by a mechanism independent of repair of conditioning-induced tissue injury. Blood 96(13):4350–4356, 2000
Min D, Taylor PA, Panoskaltsis-Mortari A, Chung B, Danilenko DM, Farrell C, et al.: Protection from thymic epithelial cell injury by keratinocyte growth factor: A new approach to improve thymic and peripheral T-cell reconstitution after bone marrow transplantation. Blood 99(12):4592–4600, 2002
Rossi S, Blazar BR, Farrell CL, Danilenko DM, Lacey DL, Weinberg KI, et al.: Keratinocyte growth factor preserves normal thymopoiesis and thymic microenvironment during experimental graft-versus-host disease. Blood 100(2):682–691, 2002
Ellison CA, Natuik SA, Fischer JM, McIntosh AR, Scully SA, Bow EJ, et al.: Effect of recombinant human keratinocyte growth factor (rHuKGF) on the immunopathogenesis of intestinal graft-vs.-host disease induced without a preconditioning regimen. J Clin Immunol 24(2):197–211, 2004
Finch PW, Rubin JS, Miki T, Ron D, Aaronson SA: Human KGF is FGF-related with properties of a paracrine effector of epithelial cell growth. Science 245(4919):752–755, 1989
Rubin JS, Osada H, Finch PW, Taylor WG, Rudikoff S, Aaronson SA: Purification and characterization of a newly identified growth factor specific for epithelial cells. Proc Natl Acad Sci USA 86(3):802–806, 1989
Boismenu R, Havran WL: Modulation of epithelial cell growth by intraepithelial gamma delta T cells. Science 266(5188):1253–1255, 1994
Yi ES, Shabaik AS, Lacey DL, Bedoya AA, Yin S, Housley RM, et al.: Keratinocyte growth factor causes proliferation of urothelium in vivo. J Urol 154(4):1566–1570, 1995
Staiano-Coico L, Krueger JG, Rubin JS, D’Limi S, Vallat VP, Valentino L, et al.: Human keratinocyte growth factor effects in a porcine model of epidermal wound healing. J Exp Med 178(3):865–878, 1993
Bottaro DP, Rubin JS, Ron D, Finch PW, Florio C, Aaronson SA: Characterization of the receptor for keratinocyte growth factor. Evidence for multiple fibroblast growth factor receptors. J Biol Chem 265(22):12767–12770, 1990
Housley RM, Morris CF, Boyle W, Ring B, Biltz R, Tarpley JE, et al.: Keratinocyte growth factor induces proliferation of hepatocytes and epithelial cells throughout the rat gastrointestinal tract. J Clin Invest 94(5):1764–1777, 1994
Pierce GF, Yanagihara D, Klopchin K, Danilenko DM, Hsu E, Kenney WC, et al.: Stimulation of all epithelial elements during skin regeneration by keratinocyte growth factor. J Exp Med 179(3):831–840, 1994
Panos RJ, Rubin JS, Csaky KG, Aaronson SA, Mason RJ: Keratinocyte growth factor and hepatocyte growth factor/scatter factor are heparin-binding growth factors for alveolar type II cells in fibroblast-conditioned medium. J Clin Invest 92(2):969–977, 1993
Ulich TR, Yi ES, Cardiff R, Yin S, Bikhazi N, Biltz R, et al.: Keratinocyte growth factor is a growth factor for mammary epithelium in vivo. The mammary epithelium of lactating rats is resistant to the proliferative action of keratinocyte growth factor. Am J Pathol 144(5):862–868, 1994
Parrott JA, Kim G, Mosher R, Skinner MK: Expression and action of keratinocyte growth factor (KGF) in normal ovarian surface epithelium and ovarian cancer. Mol Cell Endocrinol 167(1/2):77–87, 2000
Farrell CL, Bready JV, Rex KL, Chen JN, DiPalma CR, Whitcomb KL, et al.: Keratinocyte growth factor protects mice from chemotherapy and radiation-induced gastrointestinal injury and mortality. Cancer Res 58(5):933–939, 1998
Yi ES, Williams ST, Lee H, Malicki DM, Chin EM, Yin S, et al.: Keratinocyte growth factor ameliorates radiation- and bleomycin-induced lung injury and mortality. Am J Pathol 149(6):1963–1970, 1996
Ulich TR, Whitcomb L, Tang W, O’Conner Tressel P, Tarpley J, Yi ES, et al.: Keratinocyte growth factor ameliorates cyclophosphamide-induced ulcerative hemorrhagic cystitis. Cancer Res 57(3):472–475, 1997
Frank S, Munz B, Werner S: The human homologue of a bovine non-selenium glutathione peroxidase is a novel keratinocyte growth factor-regulated gene. Oncogene 14(8):915–921, 1997
Takeoka M, Ward WF, Pollack H, Kamp DW, Panos RJ: KGF facilitates repair of radiation-induced DNA damage in alveolar epithelial cells. Am J Physiol 272(6 Pt 1):L1174–L1180, 1997
Williamson E, Garside P, Bradley JA, More IA, Mowat AM: Neutralizing IL-12 during induction of murine acute graft-versus-host disease polarizes the cytokine profile toward a Th2-type alloimmune response and confers long term protection from disease. J Immunol 159(3):1208–1215, 1997
Gartner JG, Merry AC, Smith CI: An analysis of pulmonary natural killer cell activity in F1-hybrid mice with acute graft-versus-host reactions. Transplantation 46(6):879–886, 1988
Ellison CA, Fischer JM, HayGlass KT, Gartner JG: Murine graft-versus-host disease in an F1-hybrid model using IFN-gamma gene knockout donors. J Immunol 161(2):631–640, 1998
Yang X, Gieni RS, Mosmann TR, HayGlass KT: Chemically modified antigen preferentially elicits induction of Th1-like cytokine synthesis patterns in vivo. J Exp Med 78(1):349–353, 1993
Ellison CA, HayGlass KT, Fischer JM, Rector ES, MacDonald GC, Gartner JG: Depletion of natural killer cells from the graft reduces interferon-gamma levels and lipopolysaccharide-induced tumor necrosis factor-alpha release in F1 hybrid mice with acute graft-versus-host disease. Transplantation 66(3):284–294, 1998
Ellison CA, Bradley DS, Fischer JM, Hayglass KT, Gartner JG: Murine graft-versus-host disease induced using interferon-gamma-deficient grafts features antibodies to double-stranded DNA, T helper 2-type cytokines and hypereosinophilia. Immunology 105(1):63–72, 2002
Austin HA, III, Muenz LR, Joyce KM, Antonovych TA, Kullick ME, Klippel JH, et al.: Prognostic factors in lupus nephritis. Contribution of renal histologic data. Am J Med 75(3):382–391, 1983
Kimura S, Horie A, Hiki Y, Yamamoto C, Suzuki S, Kuroda J, et al.: Nephrotic syndrome with crescent formation and massive IgA deposition following allogeneic bone marrow transplantation for natural killer cell leukemia/lymphoma. Blood 101(10):4219–4221, 2003
Chan GS-W, Lam MF, Au WY, Tse KC, Chan TM, Lai KN, et al.: IgA nephropathy complicating graft-versus-host disease, another nephropathy causing nephrotic syndrome after bone marrow transplantation. Histopathology 45:642–656, 2004
Nouri-Majelan N, Sanadgol H, Ghafari A, Rahimian M, Najafi F, Mortazavizadeh M, et al.: Antineutrophil cytoplasmic antibody-associated glomerulonephritis in chronic graft-versus-host disease after allogenic hematopoietic stem cell transplantation. Transplant Proc 37(7):3213–3215, 2005
Friend SL, Hosier S, Nelson A, Foxworthe D, Williams DE, Farr A: A thymic stromal cell line supports in vitro development of surface IgM+ B cells and produces a novel growth factor affecting B and T lineage cells. Exp Hematol 22(3):321–328, 1994
Levin SD, Koelling RM, Friend SL, Isaksen DE, Ziegler SF, Perlmutter RM, et al.: Thymic stromal lymphopoietin: A cytokine that promotes the development of IgM+ B cells in vitro and signals via a novel mechanism. J Immunol 162(2):677–683, 1999
Ray RJ, Furlonger C, Williams DE, Paige CJ: Characterization of thymic stromal-derived lymphopoietin (TSLP) in murine B cell development in vitro. Eur J Immunol 26(1):10–16, 1996
Sims JE, Williams DE, Morrissey PJ, Garka K, Foxworthe D, Price V, et al.: Molecular cloning and biological characterization of a novel murine lymphoid growth factor. J Exp Med 192(5):671–680, 2000
Erickson M, Morkowski S, Lehar S, Gillard G, Beers C, Dooley J, et al.: Regulation of thymic epithelium by keratinocyte growth factor. Blood 100(9):3269–3278, 2002
Osborn MJ, Ryan PL, Kirchhof N, Panoskaltsis-Mortari A, Mortari F, Tudor KS: Overexpression of murine TSLP impairs lymphopoiesis and myelopoiesis. Blood 103(3):843–851, 2004
Taneda S, Segerer S, Hudkins KL, Cui Y, Wen M, Segerer M, et al.: Cryoglobulinemic glomerulonephritis in thymic stromal lymphopoietin transgenic mice. Am J Pathol 159(6):2355–2369, 2001
ACKNOWLEDGMENTS
We thank Amgen Inc. for providing us with the Palifermin for the study and Dr. Robert Colvin for his advice with regard to cryoglobulins. We also thank Jacqie Fischer and Bill Stefura for their excellent technical assistance. This project was supported by grants to JGG and CAE from the Canadian Institute of Health Research, the Manitoba Regional Partnership Program (Medical Research Council of Canada and the Manitoba Health Research Council), and the Manitoba Institute of Child Health.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
ELLISON, C., GIBSON, I., HAYGLASS, K. et al. Effect of Palifermin in a Murine Model of Graft-Versus-Host Disease (GVHD) Associated with Th2 Cytokine Production, Autoantibody Production, and Glomerulonephritis. J Clin Immunol 26, 485–494 (2006). https://doi.org/10.1007/s10875-006-9039-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10875-006-9039-x