Abstract
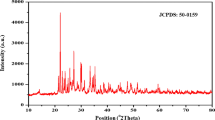
2-benzyl-amino-4-P-tolyl-6,7-di-hydro 5H-cyclo-penta –[b]pyridine–3 carbo–nitrile—BAPTDHCPCAN crystal is grown by solution growth evaporation method and analysed for XRD, Unit Cell, Dielectric analysis, UV, Tauc’s plot and fluorescence and mechano studies. The single crystalline XRD data portrays the monoclinic nature of BAPTDHCPCAN with space group P21/c and a, b, c as 8.6828 Å, 17.7283 Å, 12.0403 Å with β as 94.254°. Chemical formula of crystal as C23H21N3. The crystalline dimension is 14 × 12x7 mm3. The dielectric properties of the BAPTDHCPCAN have larger value at low range of frequencies. The phase matching effectiveness is analyzed and found to be 66 mV. The influx value of macro and thin film of BAPTDHCPCAN crystal is 4.2125 microns and 4.4336 microns as mid value before nano shaped influx. Absorption spectral two peaks centered at 248 nm at 325 nm, the band gap of crystal is calculated as 5.0 eV by photonic enhancement and the fluorescence represented emission for 399 nm. The BAPTDHCPCAN crystal is found to have represented –ve photoconductive nature. The BAPTDHCPCAN crystal specimen in of RISE [Reverse Indentation Size Effect] consequence with n greater than 2.














Similar content being viewed by others
Data availability
No specific menu for data; all data are presented in manuscript.
References
R. Krishnaveni et al., Preamble to Biomaterials and its Applications in Science and Technology (Lulu Publications, Morrisville, 2019)
S. Gunasekaran, K. SenthilKannan, S. Loganathan et al., Indian J Phys. 87, 1189 (2013)
R. Aarthi, S. Ramalingam, S. Periandy, K. SenthilKannan, J. Taibah Uni. Sci. 12, 104 (2018)
K. SenthilKannan, S. Gunasekaran, K.A. Seethalakshmi, Int. J. Sci. Eng. Res. 4, 2 (2013)
N. Kishore, K. SenthilKannan, T. Periyanayagi et al., Appl. Phys. A 123, 706 (2017)
P. Saravanan, K. SenthilKannan, R. Divya et al., J Mater Sci: Mater Electron 31, 4301 (2020)
M. Jothibas, A. Muthuvel et al., AIP Conf. Proc. 2162, 020151 (2019)
S. Gnanam et al., IOP Conf. Series: Mater. Sci. Eng. 561, 012086 (2019)
R. Krishnaveni et al., IJAEMA 11, 8 (2019)
K. SenthilKannan, Int. J. Eng. Sci. Math. 7, 1 (2018)
K. SenthilKannan et al., Int. J. Chemtech Res. 6, 3187 (2014)
K. SenthilKannan, S. Gunasekaran, Int. J. Fron. Sci. Tech. 3, 29 (2013)
T. Malathi Rekha et al., Int. Res. J. Nat. Appl. Sci. 5, 133 (2018)
K. Senthilkannan et al., Sch. Res. Lib. Arch. Appl. Sci. Res. 5, 100 (2013)
G. Flora et al., Mater. Today: Proc. 33, 4233 (2020)
R. Senthilkumar et al., Mater. Today: Proc. 33, 4167 (2020)
K. Senthilkannan et al., Mater. Today: Proc. 33, 4145 (2020)
K. Senthilkannan et al., Mater. Today: Proc. 33, 4163 (2020)
K. Senthilkannan et al., Mater. Today: Proc. 33, 4148 (2020)
G. Wilcox, Clin. Biochem. Rev. 26, 19 (2005)
C.Y. Yang, Y.Y. Yen, K.C. Hung et al., Nutr. Diabetes 9, 1 (2019)
R. Tundis, M.R. Loizzo, F. Menichini, Mini Rev. Med. Chem. 10, 315 (2010)
S.O. Oyedemi, B.O. Oyedemi, I.I. Ijeh et al., Sci. World J. 3, 1 (2017)
K. Khadayat, B.P. Marasini, H. Gautam et al., Clin. Phytosci. 6, 1 (2020)
K. Kumar, K. Senthilkannan, R. Hariharasuthan, M. Jothibas, M. Vimalan, P. Baskaran, M. Iyanar, M. Kolan**athan, J. Mater. Sci. Mater. Electron. 31, 1 (2020)
L.J. Farrugia, J. Appl. Cryst. 45, 849 (2012)
M. Kolan**athan, K. Senthilkannan, S. Paramasivam, P. Baskaran, M. Iyanar, Mater. Today: Proc. 33, 2750 (2020)
P. Periyathambi et al., Mater. Today: Proc. 33, 3766 (2020)
L. Selvaraj et al., Mater. Today: proc. 33, 3763 (2020)
G. Flora et al., Mater. Today: Proc. 33, 3937 (2020)
G. Flora et al., Mater. Today: Proc. 33, 3578 (2020)
K. Senthilkannan et al., Mater. Today: Proc. 33, 3201 (2020)
P. Saravanan et al., Mater. Today: Proc. 33, 3160 (2020)
K. SenthilKannan et al., Mater. Today: Proc. 33, 3053 (2020)
K. Senthilkannan et al., Mater. Today: Proc. 33, 3056 (2020)
P. Baskaran et al., Mater. Today: Proc. 33, 3058 (2020)
P. Baskaran et al., Mater. Today: Proc. 33, 3051 (2020)
K. SenthilKannan et al., Mater. Today: Proc. 33, 3005 (2020)
K. SenthilKannan et al., Mater. Today: Proc. 33, 2779 (2020)
K. SenthilKannan et al., Mater. Today: Proc. 33, 2776 (2020)
K. SenthilKannan et al., Mater. Today: Proc. 33, 2753 (2020)
V. Kalaipoonguzhali et al., Mater. Today: Proc. 33, 2759 (2020)
M. Kolan**athan et al, Mater. Today: Proc. 33, 2750 (2020)
K. SenthilKannan et al., Mater. Today: Proc. 33, 2746 (2020)
K. SenthilKannan et al., Mater. Today: Proc. 33, 2738 (2020)
G. Flora, K. Senthilkannan, R. Rengarajan, P. Saravanan, Mater. Today: Proc. 33, 4233 (2020)
M.S. Krishnamurthy, N.S. Begum, Acta Crystallogr. E 70, 760 (2014)
R.P. Patel, K. SenthilKannan, R. Hariharasuthan et al., Braz. J. Phys. 51, 339 (2021)
Z.Y. Zhang, X.Y. Yong, M. **ao, Appl. Phys. Lett. 81, 2076 (2002)
S.K. Kurtz, T.T. Perry, J. Appl. Phys. 39, 3798 (1968)
A. Von Hippel, E.P. Gross, F.G. Telstis, A. Geller, Phys. Rev. 91, 568 (1953)
C. Hrizi, N. Chaari, Y. Abid, N. Chniba-Boudjada, S. Chaabouni, [C10H7NH3]BiI4. Polyhedron 46, 41 (2012)
K. SenthilKannan, S. Gunasekaran, Int. J. Fron. Sci. Tech. Res. 3, 29 (2013)
A.C. Dhieb, A. Valkonen, M. Rzaigui, W. Smirani, J. Mol. Struct. 1102, 50 (2015)
R. Tundis, M.R. Loizzo, F. Menichini Mini, Mini. Rev. Med. Chem. 10, 315 (2010)
S.O. Oyedemi, B.O. Oyedemi, I.I. Ijeh, Sci. World J. 2017, 1 (2017)
K. Khadayat, B.P. Marasini, H. Gautam, Clin. Phytosci. 6, 1 (2020)
L.K. Williams, X. Zhang, S. Caner Nat. Chem. Biol. 11, 691 (2019)
S.K. Burley, H.M. Berman, C. Bhikadiya, Nucleic Acids Res. 47, 464 (2019)
W.L. DeLano, The PyMOL molecular graphics system. (2020)
S. Dallakyan, A.J. Olson, Methods Mol. Biol. 1263, 243–250 (2014)
T.A. Halgren, Merck molecular force field I Basis, form, scope, parameterization, and performance of MMFF94. J Comp Chem 17, 490 (1996)
BIOVIA, Dassault systemes, [Biovia Discovery studio Visualizer], San Diego: Dassault systems, (2020)
A. Daina, O. Michielin, V. Zoete, SwissADME, A free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Sci. Rep. 7, 1 (2016)
A. Selmi, A. Zarei, W. Tachoua, H. Puschmann, H. Teymourinia, A. Ramazani, Chem. Methodol. 6, 463–474 (2022)
Teena Mathew and their co-workers, Mechanical and Dielectric Properties of InTe Crystals. (2012)
K. Krishna Kumar, G. Sathaiah, L. Sirdeshmukh, Int. J. Chem. Sci. 9(1), 239 (2011)
S. Goma, C.M. Padma, C.K. Mahadevan, Mater. Lett. 60(29), 3701 (2006)
M. Meena, C.K. Mahadevan, R.S. Saravanan, V.N. Praveen, Effect of Added Impurities on the Properties of LAHCL Single Crystals. Int. J. Macro Nano Phys. 1(1), 12–18 (2016)
M. Meena, C.K. Mahadevan, Mater. Lett. 62, 3742–3744 (2008)
M. Meena, C.K. Mahadevan, Effect of added impurities on the electrical properties of L-arginine acetate single crystals. Arch Appl. Sci. Res. 2, 185–199 (2010)
S. Mendiratta, M. Usman, T.-T. Luo, S.-F. Lee, Y.-C. Lin, Lu. Kuang-Lieh, Cryst. Eng. Comm. 16(28), 6309 (2014)
S.K. Patri, B.B. Sahu, Ferroelectrics 1(518), 178 (2017)
C. Kettel, Introduction to solid state Physics (Wiley, New York, 1965)
S. Suku, R. Ravindran, Synthesis. J. Mol. Struct. 1226, 129314 (2021)
C.P. Smyth, Dielectric behaviour and structure (McGraw - Hill, New York, 1965)
M.E. Lines, A.M. Glass, Principle and application of ferroelectrics and related material (Oxford University Press, Oxford, 2001)
L. González, L. Yu, S. Hvilsted, A.L. Skov, RSC Adv. 4(68), 36117–36124 (2014)
H. Bouaamlat et al., Adv. Mater. Sci. Eng. (2020). https://doi.org/10.1155/2020/8689150
M. Hamdi, S. Karoui, A. Oueslati, S. Kamoun, F. Hlel, J. Mol. Struc. 1154, 516–523 (2017)
N. Goel, N. Sinha, B. Kumar, Opt. Mater. 35, 479–486 (2013)
W. Mao, J. Wang, H. Liao, B. Zhou, G. Zheng, S. Mo, F. Long, Z. Zou, J. Saudi Chem. Soc. 23, 52 (2019)
J. Balaji, P. Srinivasan, S. Prabu, M. George, D. Sajan, J. Mol. Struct. 1207, 1–27 (2020)
R. Divya, M. Meena, C.K. Mahadevan, C.M. Padma, Int. J. Eng. Res. Appl 4(5), 01 (2014)
R. Casalini, C.M. Roland, J. Polym. Sci. Part B: Polym. Phys. 48, 582 (2010)
Funding
No funding arises from any source and the authors share their own equal contribution for synthesis, studies and, etc.
Author information
Authors and Affiliations
Contributions
SK: Crystal synthesis. MJ: molecular model, Electrical effect study, MR: write up for mechanical. SNT: computational model. PRP: XRD study and write up. TK: Dielectric write up. VM: Photoconductivity and phase matching write up. SK: Influx, Experimental and write up.Overall corrections, submission.
Corresponding authors
Ethics declarations
Conflict of interest
All authors have no conflict of interest and paper is not submitted anywhere and is a novel work.
Ethical approval
The manuscript is not be submitted to more than one journal for simultaneous consideration. The submitted work is original and is not published elsewhere in any form or language (partially or in full), A single study is not be split up into several parts. Results should be presented clearly, honestly, and without fabrication, falsification or inappropriate data manipulation. No data, text, or theories by others are presented as if they were the author’s own (‘plagiarism’).
Consent to participate
All authors willing to participate.
Consent for publication
All authors willing to publish.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Suganya, K., Maalmarugan, J., Manikandan, R. et al. Synthesis, studies of 2-benzyl-amino-4-p-tolyl-6,7-di-hydro 5H-cyclo-penta–[b]pyridine-3 carbo-nitrile (BAPTDHCPCN) crystals for optical, photonic and mechano-electronic uses. J Mater Sci: Mater Electron 33, 19320–19330 (2022). https://doi.org/10.1007/s10854-022-08770-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-022-08770-0