Abstract
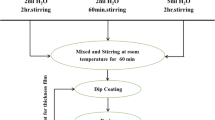
Ag and Sn co doped TiO2 thin films were deposited on glass slides by sol–gel dip coating technique. Morphology, optical properties and wettability of the obtained TiO2 thin films changed by changing the content of dopant. The TiO2 thin films with silver molar ratio from 5 to 20% (molar ratio of tin fixed at 5 mol%) were tested for its photo catalytic property under irradiation of visible light. The influences of different molar ratio of Ag on hydrophilicity of the films were also investigated by measuring the water contact angle. The results showed that the wettability was significantly improved by increasing silver content comparing with pure TiO2 thin film. Accordingly, the thin films showed various optical band gaps. As, the optical band gap of the nanostructured thin films decreased with various content of dopant from 3.15 to 2.13 eV.








Similar content being viewed by others

References
O. Carp, C.L. Huisman, A. Reller, Prog. Solid State Chem. 32, 33–177 (2004)
X.B. Chen, S.M. Samuel, Chem. Rev. 107, 2891–2959 (2007)
M.M. Momeni, I. Ahadzadeh, A. Rahmati, J. Mater. Sci.: Mater. Electron. 27, 8646–8653 (2016)
R. Dastjerdi, M. Montazer, Colloids Surf. B 79, 5–18 (2010)
M.M. Momeni, Y. Ghayeb, M. Davarzadeh, J. Electroanal. Chem. 739, 149–155 (2015)
M.M. Momeni, Y. Ghayeb, Appl. Phys. A 122, 620 (2016)
F. Fujishima, X. Zhang, D.A. Tryk, Surf. Sci. Rep. 63, 515–582 (2008)
Y. Lai, C. Lin, H. Wang, J. Huang, H. Zhuang, L. Sun, Electrochem. Commun. 10, 387–391 (2008)
M.M. Momeni, Y. Ghayeb, J. Alloys Compd. 637, 393–400 (2015)
S. Bauer, J. Park, K. Mark, P. Schmuki, Acta Biomater. 4, 1576–1582 (2008)
M.M. Momeni, M. Hakimian, A. Kazempour, Surf. Eng. 32, 514–519 (2016)
M.M. Momeni, Y. Ghayeb, J. Mol. Catal. A: Chem. 417, 107–115 (2016)
T. Yuranova, R. Mosteco, J. Bandara, D. Laub, J. Kiwi, J. Mol. Catal. A: Chem. 244, 160–167 (2006)
B. Sanchez, J.M. Coronado, R. Candal, B. Appl. Catal. et al., Environ 66, 295–301 (2006)
M.M. Momeni, I. Ahadzadeh, Mater. Res. Innov. 20, 44–50 (2016)
M.M. Momeni, Y. Ghayeb, Ceram. Int. 42, 7014–7022 (2016)
M.C. Hidalgo, S. Sakthivel, D. Bahnemann, Appl. Catal. A Gen. 277, 183–189 (2004)
M.M. Momeni, Z. Nazari, Ceram. Int. 42, 8691–8697 (2016)
K. Hashimoto, H. Irie, A. Fujishima, Jpn. Soc. Appl. Phys. 44, 8269–8285 (2005)
H. **ng-Gang, L. An-Dong, H. Mei-Dong, L. Bin, W. **ao-Ling, Chin. Phys. Lett. 26, 077106 (2009)
N. Najibi Ilkhechi, B. Koozegar Kaleji, Silicon (2015). doi:10.1007/s12633-015-9356-x
M.M. Momeni, Y. Ghayeb, J. Electroanal. Chem. 751, 43–48 (2015)
X.G. Hou, M.D. Huang, X.L. Wu, A.D. Liu, Chem. Eng. J. 146, 42–48 (2009)
M.M. Momeni, Y. Ghayeb, Z. Ghonchegi, Ceram. Int. 41, 8735–8741 (2015)
B. **n, L. **g, Z. Ren, B. Wang, H. Fu, J. The, Phys. Chem. B 109, 2805–2809 (2005)
M.M. Momeni, Mater. Res. Innov. 20, 317–325 (2016)
H.E. Chao, Y.U. Yun, H.U. **ngfang, A. Larbot, J. Eur. Ceram. Soc. 23, 1457–1464 (2003)
M.M. Momeni, Y. Ghayeb, J. Appl. Electrochem. 45, 557–566 (2015)
Q. Liu, X. Wu, B. Wang, Q. Liu, Mater. Res. Bull. 37, 2255–2262 (2002)
J. Martinez, D.R. Acosta, G. Cedillo, Thin Solid Films 490, 118–123 (2005)
K.K. Akurati, A. Vital, R. Hany, B. Bommer, T. Graule, M. Winterer, Int. J. Photoenergy 7, 153–161 (2005)
J. Yang, D. Li, X. Wang, L. Yang, L. Lu, J. Solid State Chem. 165, 193–198 (2002)
M.M. Momeni, Y. Ghayeb, J. Solid State Electrochem. 20, 683–689 (2016)
S. Mahanty, S. Roy, S. Sen, J. Cryst. Growth 261, 77–81 (2004)
Z.M. Shi, L. Yan, L.N. **, X.M. Lu, G. Zhao, J. Non-Cryst. Solids 353, 2171–2178 (2007)
M.M. Momeni, M. Mirhosseini, M. Chavoshi, Ceram. Int. 42, 9133–9138 (2016)
M.M. Momeni, Y. Ghayeb, J. Iran. Chem. Soc. 13, 481–488 (2016)
B. Koozegar Kaleji, R. Sarraf-Mamoory, K. Nakata, A. Fujishima, J. Sol-Gel. Sci. Technol. 60, 99–107 (2011)
X. Wang, X. Hou, W. Luan, D. Li, K. Yao, Appl. Surf. Sci. 258, 8241–8246 (2012)
N. Najibi Ilkjechi, B. Koozegar Kaleji, J. Sol-Gel. Sci. Technol. 69, 351–356 (2014)
N.N. Ilkhechi, B.K. Kaleji, E. Salahi, N. Hosseinabadi, J. Sol-Gel. Sci. Technol. 74(3), 765–773 (2015)
N. Najibi Ilkhechi, Z. Azar, M. Khajeh, M. Mozammel, J. Mater. Sci.: Mater. Electron. 27, 10541–10549 (2016)
R.A. Spurr, H. Myers, Anal. Chem. 29, 760–762 (1957)
A.H. Dorian, C. Charles, J. Mater. Sci. 46, 855–874 (2011)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ilkhechi, N.N., Ghorbani, M., Mozammel, M. et al. The optical, photo catalytic behavior and hydrophilic properties of silver and tin co doped TiO2 thin films using sol–gel method. J Mater Sci: Mater Electron 28, 3571–3580 (2017). https://doi.org/10.1007/s10854-016-5958-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-016-5958-3