Abstract
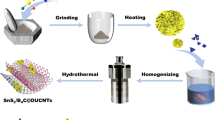
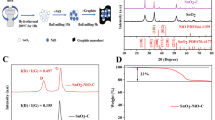
In this report, investigations on pristine vs. hybrid SnO2 modified by four different routes are reported. The pristine SnO2 is a potential anode with high theoretical capacity (1494 mAhg−1), but the volumetric issues prevails the viability of this material in Li-ion battery systems. For enhancing the stability and anode specific properties, SnO2 is incorporated with CNT, ZnO, Li, and amorphous/less crystalline phase of its own. Both physical and electrochemical aspects of the SnO2 experience huge improvement on changing its pristine nature. Structural analysis made by XRD indicates that except SnO2/ZnO matrix, all the samples are phase pure falling into the tetragonal symmetry. SnO2/ZnO sample possesses mixed tetragonal and hexagonal symmetry. The presence of foreign elements greatly impacts the morphological patterns of the sample as ensured by TEM analysis. Do** helps in bandgap engineering of the pristine material by decreasing the energy gap value and thereby improving the redox properties of pristine. Cyclic voltammetry analysis shows betterment in the electrochemical properties of the SnO2 material, whereas galvanostatic charge–discharge analysis shows specific capacitances, 158 mAhg−1, 450 mAhg−1, ~ 225 mAhg−1, 425 mAhg−1, 275 mAhg−1 with respect to SnO2, SnO2/CNT, SnO2/ZnO, SnO2/Li, and composite SnO2 for 1000 cycles with a Coulombic efficiency of ~ 99% except SnO2/Li. Overall, the order of anodic performance of the materials is sorted as, SnO2/CNT, SnO2/Li, SnO2/ZnO, composite SnO2, and pristine SnO2. In summary, the investigations on SnO2 hybrids have shown promising results in improving their properties, especially in capacitance and life time. Inspired by this, future investigations related to SnO2 hybrids will likely revolve around optimizing their anodic properties by improving their long-term stability and compatibility with different battery materials.








Similar content being viewed by others
Data availability
Data can be made available on request.
References
Li L, Zhang D, Deng J, GouFang YJ, Cui H, Zhao Y, Cao M (2021) Carbon-based materials for fast charging lithium-ion batteries. Carbon 183:721–734
Zoller F, Böhm D, BeinFattakhova-Rohlfing TD (2019) Tin oxide based nanomaterials and their application as anodes in lithium-ion batteries and beyond. Chemsuschem 12:4140–4159
Nzereogu PU, Omah AD, Ezema FI, Iwuoha EI, Nwanya AC (2022) Anode materials for lithium-ion batteries: a review. Appl Surf Sci Adv 9:100233
Lakshmi D, Infanta Diana M, Nalini B, Soundarya GG, Priyanka P, Jayapandi S, Christopher Selvin P (2021) Fine-tuning of stannic oxide anodes’ material properties through calcination. J Mater Sci Mater Electron 32:27384–27397
Asenbauer J, Eisenmann T, Kuenzel M, Kazzazi A, Chen Z, Bresser D (2020) The success story of graphite as a lithium-ion anode material—fundamentals, remaining challenges, and recent developments including silicon (oxide) composites. Sustain Energy Fuels 4:5387–5416
Zhang H, Zhang X, ** H, Zong P, Bai Y, Lian K, XuMa HF (2019) A robust hierarchical 3D Si/CNTs composite with void and carbon shell as Li-ion battery anodes. Chem Eng J 360:974–981
Noerochim L, Indra MAT, Purwaningsih H, Subhan A (2018) Porous Fe2O3 microspheres as anode for lithium- ion batteries. IOP Conf Ser Mater Sci Eng 367:012038
Pan Y, Xu M, Yang L, Yu M, Liu H, Zeng F (2020) Porous architectures assembled with ultrathin Cu2O–Mn3O4 hetero-nanosheets vertically anchoring on graphene for high-rate lithium-ion batteries. J Alloys Compd 819:152969
Kebede MA (2020) Tin oxide–based anodes for both lithium-ion and sodium-ion batteries. Curr Opin Electrochem 21:182–187
Ronan O, Roy A, Ryan S, Hughes L, Downing C, Jones L, Nicolosi V (2023) Templated synthesis of Sio2 nanotubes for lithium-ion battery applications: an in situ (scanning) transmission electron microscopy study. ACS Omega 8(1):925–933. https://doi.org/10.1021/acsomega.2c06298
Liang S, Wang X, Qi R, Cheng YJ, **a Y, Buschbaum PM, Hu X (2022) Bronze-phase TiO2 as anode materials in lithium and sodium-ion batteries. Adv Func Mater 32(25):2201675
Cheng YW, Chen CH, Yang SW, Li YC, Peng BL, Chang CC, Wang RC, Liu CP (2018) Freestanding three-dimensional CuO/NiO core-shell nanowire arrays as high-performance lithium-ion battery anode. Sci Rep 8:18034
Chen X, Zhou J, Li J, Luo H, Mei L, Wang T, Zhu J, Zhang Y (2019) A germanium and zinc chalcogenide as an anode for a high-capacity and long cycle life lithium battery. RSC Adv 9:35045–35049. https://doi.org/10.1039/C9RA06023E
Guo M, Fu C, Jiang M, Bai Y, Zhang J, Sun B (2020) High performance aluminum foam-graphite dual-ion batteries and failure analysis. J Alloys Compd 838:155640
Kaspar J, Bazarjani MS, Schitco C, Gurlo A, Graczyk-ZajacRiedel MR (2017) Electrochemical study of NiO nanosheets: toward the understanding of capacity fading. J Mater Sci 11:6498–6505
Wang X, Liu Y, Zeng J, Peng C, Wang R (2019) MoO2/C hollow nanospheres synthesized by solvothermal method as anode material for lithium-ion batteries. Ionics 2:437–445
Li H, Wei Y, Zhao Y, Zhang Y, Yin F, Zhang C, Bakenov Z (2016) Simple one-pot synthesis of hexagonal ZnO nanoplates as anode material for lithium-ion batteries. J Nanomater 2016:4675960. https://doi.org/10.1155/2016/4675960
Zhang H, Yang Y, Xu H, Wang L, Lu X, He X (2022) Li4Ti5O12 spinel anode: fundamentals and advances in rechargeable batteries. Infomat 4(4):e12228
Ko M, Oh P, Chae S, Cho W, Cho J (2015) Considering critical factors of Li-rich cathode and Si anode materials for practical Li-ion cell applications. Small 33:4058–4073
Ambalkar AA, Kawade UV, Sethi YA, Kanade SC, Kulkarni MV, Adhyapak PV, Kale BB (2021) A nanostructured SnO2/Ni/CNT composite as an anode for Li-ion batteries. RSC Adv 11:19531–19540
Wang L, Zhang P, Chen X, Fang Y, Wu J, Zhang Q (2022) Synthesis of highly dispersed Zn-doped Sno2 spherical nano materials for anode of lithium-ion batteries. Chem Lett 51(9):957–961
Li Y, Liu R, Wang C, Zhou Y (2021) Uniform nano-SnO2/C composite anodes from coal tar pitch for sodium-ion batteries. Energy Fuels 35(10):9029–9037
Liu S, Tao W, Yu Y, Fakudze S, Wang C, Wang J, Han J, Chen J (2022) Ball milling synthesis of robust sandwich-structured C/Si@SnO2 anode with porous silicon buffer layer for fast charging lithium-ion battery. Colloids Surf A Physicochem Eng 654:130088
Zheng W, Nie S, **n Y, Chen K, Mou H, Qin Y, **ao W (2022) Uniform integration of amorphous SnO2 nanoparticles encapsulated into the carbon matrix as enhanced electrochemical performance anodes for lithium-ion batteries. Ionics 28:4949–4958
Lakshmi D, Nalini B, Sivaraj P, Jayapandi S (2017) Electro analytical studies on indium incorporated SnSb alloy anode for Li-ion batteries. J Electroanal Chem 801:459–465
Jayapandi S, Premkumar S, Lakshmi D, Packiyaraj P, Sivaraj P, Anitha K (2019) Reinforced photocatalytic reduction of SnO2 nanoparticle by La incorporation for efficient photodegradation under visible light irradiation. J Mater Sci 30:8479–8492
Shawuti S, Can MM, Gülgün MA (2020) Complex impedance analyses of Li doped ZnO electrolyte materials. Sci Rep 10:8228
Blessi S, Anand S, Manikandan A, Sonia M, Vinosel VM, Slimani Y, Almessiere MA, Baykal A (2021) Structural, optical, and electrochemical investigations of sb-substituted mesoporous SnO2 nanoparticles. J Mater Sci Mater Electron 4:4132–4145
Lakshmi D, Nalini B (2017) Performance of SnSb: Ce, Co alloy as anode for lithium-ion batteries. J Solid State Electrochem 4:1027–1034
Zhao B, Dhara A, Dendooven J, Detavernier C (2020) Atomic layer deposition of SnO2-based composite anodes for thin-film lithium-ion batteries. Front Energy Res 8:609417. https://doi.org/10.3389/fenrg.2020.609417
Ng B, Peng X, Faegh E, Mustain WE (2020) Using nanoconfinement to inhibit the degradation pathways of conversion-metal oxide anodes for highly stable fast-charging Li-ion batteries. J Mater Chem A 8(5):2712–2727
Author information
Authors and Affiliations
Contributions
DL contributed to conception, writing, and interpretation. MID was involved in experiments and writing. SJ contributed to measurements. PCS was involved in supervision. PB contributed to measurements.
Corresponding authors
Ethics declarations
Conflicts of interest
The authors declare no conflicts of interests.
Additional information
Handling Editor: Jean-Francois Gohy.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lakshmi, D., Diana, M.I., Jayapandi, S. et al. Deciphering the functionalization routes for SnO2 anodes. J Mater Sci 58, 15471–15484 (2023). https://doi.org/10.1007/s10853-023-08986-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-023-08986-3