Abstract
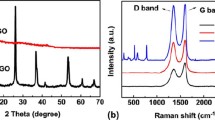
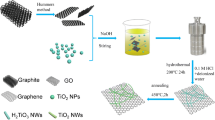
Titanium dioxide/graphene composite is receiving intensive attention because of its potential applications in energy field. Herein, we report the preparation of a three-dimensional TiO2/graphene porous composite prepared by using a sacrificial template strategy, avoiding the usual hydrothermal and freeze-drying processes. Graphene oxide sheets and TiO2 nanoparticles were first dispersed in a block copolymer micelle solution, and then the mixture was exposed to a non-solvent vapor atmosphere to evaporate the solvent. Finally, the resultant intermediate product was calcined in nitrogen to remove polymer template. As a result, the obtained free-standing composite material has a three-dimensional porous microstructure. Scanning electron microscopy and transmission electron microscopy observations indicate that the composite is supported by TiO2-anchored graphene sheets. More interestingly, nano-carbon particles derived from the carbonization of the polymer template are evenly deposited onto both the graphene sheets and the TiO2 nanoparticles. The specific capacitance of the carbon/TiO2/reduced graphene oxide composite has been measured to reach 23.6 mF/cm2. Our results indicate that the enhanced electrochemical properties of the composite are attributed to a synergistic effect of the 3-D porous network and the unique microstructure. The electrochemical stability and the cycle performance of the obtained composite electrode are tested to illustrate its potential applications.








Similar content being viewed by others
References
Huang X, Qi XY, Boey F, Zhang H (2012) Graphene-based composites. Chem Soc Rev 44:666–686
Stankovich S, Dikin DA, Dommett GHB, Kohlhaas KM, Zimney EJ, Stach EA, Piner RD, Nguyen ST, Ruoff RS (2006) Graphene-based composite materials. Nature 442:282–286
Liu GJ, Fan LQ, Yu FD, Wu JH, Liu L, Qiu ZY, Liu Q (2013) Facile one-step hydrothermal synthesis of reduced graphene oxide/Co3O4 composites for supercapacitors. J Mater Sci 48:8463–8470. doi:10.1007/s10853-013-7663-4
de Guzman RC, Yang JH, Ming-Cheng M, Salley SO, Ng KYS (2013) A silicon nanoparticle/reduced grapheme oxide composite anode with excellent nanoparitcle dispersion to improve lithium ion battery performance. J Mater Sci 48:4823–4833. doi:10.1007/s10853-012-7094-7
Qiu YF, Cheng ZY, Guo B, Fan HB, Sun SF, Wu T, ** L, Fan L (2014) Preparation of activated carbon paper through a simple method and application as a supercapacitor. J Mater Sci 50:1586–1593. doi:10.1007/s10853-014-8719-9
Bello A, Makgopa K, Fabiane M, Dodoo-Ahrin D, Ozoemena KI, Manyala N (2013) Chemical adsorption of NiO nanostructures on nickel foam-graphene for supercapacitor applications. J Mater Sci 48:6707–6712. doi:10.1007/s10853-013-7471-x
Han S, Wu DQ, Li S, Zhang F, Feng XL (2014) Porous graphene materials for advanced electrochemical energy storage and conversion devices. Adv Mater 26:849–864
Li W, Wang F, Feng SS, Wang JX, Sun ZK, Li B, Li YH, Yang JP, Elzatahry A, **a YY, Zhao DY (2013) Sol-gel design strategy for ultradispersed TiO2 nanoparticles on graphene for high-performance lithium ion batteries. J Am Chem Soc 135:18300–18303
Zhang ZY, **ao F, Guo YL, Wang S, Liu YQ (2013) One-pot self-assembled three-dimensional TiO2-graphene hydrogel with improved adsorption capacities and photocatalytic and electrochemical activities. ACS Appl Mater Interface 5:2227–2233
Etacheri V, Yourey JE, Bartlett BM (2014) Chemically bonded TiO2-Bronze nanosheet/reduced graphene oxide hybrid for high-power lithium ion batteries. ACS Nano 8:1491–1499
Ding B, Xu GY, Shen LF, Nie P, Hu PF, Dou H, Zhang XG (2013) Sulfur embedded in metal organic framework-derived hierarchically porous carbon nanoplates for high performance lithium-sulfur battery. J Mater Chem A 1:14280–14285
Liu WW, Yan XB, Xue QJ (2013) Multilayer hybrid films consisting of alternating graphene and titanium dioxide for high-performance supercapacitors. J Mater Chem C 1:1413–1422
Simon P, Gogotsi Y (2008) On the molecular origin of supercapacitance in nanoporous carbon electrodes. Nat Mater 7:45–854
Perez CR, Yeon SH, Segalini J, Presser V, Taberna PL, Simon P, Gogotsi Y (2013) Structure and electrochemical performance of carbide-derived carbon nanopowders. Adv Funct Mater 23:1081–1089
**ong X, Eckelt J, Wolf BA, Zhang Z, Zhang L (2006) Continuous spin fractionation and characterization by size-exclusion chromatography for styrene-butadiene block copolymers. J Chromatogr A 1110:53–60
Marcano DC, Kosynkin DV, Berlin JM (2010) Improved synthesis of graphene oxide. ACS Nano 4:4806–4814
Nian YR, Teng HS (2002) Nitric acid modification of activated carbon electrodes for improvement of electrochemical capacitance. J Electrochem Soc 149:1008–1014
Sliwak A, Grzyb B, Cwikla J, Gryglewicz G (2013) Influence of wet oxidation of herringbone carbon nanofibers on the pseudocapacitance effect. Carbon 64:324–333
Ke QR, Liao YY, Lin MF, Lin SS, Du H, Yao S, **ong XP A superhydrophobic film with high water vapor transmission prepared from block copolymer micelle solution via VIPS method. J Polym Res. doi:10.1007/s10965-015-0850-z
Lee HJ, Jung B, Kang YS, Lee H (2004) Phase separation of polymer casting solution by nonsolvent vapor. J Membr Sci 245:103–112
Khare VP, Greenberg AR, Krantz WB (2005) Vapor-induced phase separation—effect of the humid air exposure step on membrane morphology Part I. Insights from mathematical modeling. J Membr Sci 258:140–156
Yip Y, McHugh AJ (2006) Modeling and simulation of nonsolvent vapor-induced phase separation. J Membr Sci 271:163–176
Peinemann KV, Abetz V, Simon PFW (2007) Asymmetric superstructure formed in a block copolymer via phase separation. Nat Mater 6:992–996
Bouyer D, Werapun W, Pochat-Bohatier C, Deratani A (2010) Morphological properties of membranes fabricated by VIPS process using PEI/NMP/water system: SEM analysis and mass transfer modelling. J Membr Sci 349:97–112
Tsai JT, Su YS, Wang DM, Kuo JL, Lai JY, Deratani A (2010) A retainment of pore connectivity in membranes prepared with vapor-induced phase separation. J Membr Sci 362:360–373
Rangou S, Buhr K, Filiz V, Clodt JI, Lademann B, Hahn J, Jung A, Abetz V (2014) Self-organized isoporous membranes with tailored pore sizes. J Membr Sci 451:266–275
Wu S, Bubeck C (2013) Macro- and microphase separation in block copolymer supramolecular assemblies induced by solvent annealing. Macromolecules 46:3512–3518
She MS, Lo TY, Ho RM (2014) Controlled ordering of block copolymer gyroid thin films by solvent annealing. Macromolecules 47:175–182
Huang WH, Chen PY, Tung SH (2012) Effects of annealing solvents on the morphology of block copolymer-based supramolecular thin films. Macromolecules 45:1562–1569
Wu HB, Chen JS, Lou XW, Hng HH (2011) Asymmetric anatase TiO2 nanocrystals with exposed high-index facets and their excellent lithium storage properties. Nanoscale 3:4082–4084
Liu HP, Ye T, Mao CD (2007) Fluorescent carbon nanoparticles derived from candle soot. Angew Chem Int Ed 46:6473–6475
Ray SC, Saha A, Jana NR, Sarkar R (2009) Fluorescent carbon nanoparticles: synthesis, characterization, and bioimaging application. J Phys Chem C 113:18546–18551
Tian L, Ghosh D, Chen W, Pradhan S, Chang X, Chen S (2009) Nanosized carbon particles from natural gas soot. Chem Mater 21:2803–2809
Sun YQ, Wu Q, Shi GQ (2011) High-performance supercapacitor electrodes based on graphene hydrogels modified with 2-aminoanthraquinone moieties. Phys Chem Chem Phys 13:17249–17254
Kim TY, Lee HW, Stoller M, Dreyer DR, Bielawski CW, Ruoff RS, Suh KS (2011) High-performance supercapacitors based on poly(ionic liquid)-modified graphene electrodes. ACS Nano 5:436–442
Subramanian V, Zhu H, Vajtai R, Ajayan PM, Wei B (2005) Hydrothermal synthesis and pseudocapacitance properties of MnO2 nanostructures. J Phys Chem B 109:20207–20214
Zhang JT, Jiang JW, Zhao XS (2011) Synthesis and capacitive properties of manganese oxide nanosheets dispersed on functionalized graphene sheets. J Phys Chem C 115:6448–6454
Moon GD, Joo JB, Dahl M, Jung H, Yin YD (2014) Nitridation and layered assembly of hollow TiO2 shells for electrochemical energy storage. Adv Funct Mater 24:848–856
Lin Z, Waller G, Liu Y, Liu M, Wong CP (2012) Facile synthesis of nitrogen-doped graphene via pyrolysis of graphene oxide and urea, and its electrocatalytic activity toward the oxygen-reduction reaction. Adv Energy Mater 2:884–888
Yang S, Feng XL, Wang X, Müllen K (2011) Graphene-based carbon nitride nanosheets as efficient metal-free electrocatalysts for oxygen reduction reactions. Angew Chem Int Ed 50:5339–5343
Long D, Li W, Ling L, Miyawaki J, Mochida I, Yoon SH (2010) Preparation of nitrogen-doped graphene sheets by a combined chemical and hydrothermal reduction of graphene oxide. Langmuir 26:16096–16102
Fievet P, Mullet M, Pagetti J (1998) Comparison of two electrokinetic methods-electroosmosis and streaming potential to determine the zeta- potential of plane ceramic membranes. J Membr Sci 149:143–150
Sun F, Liu J, Chen H, Zhang Z, Qiao WM, Long D, Ling L (2013) Nitrogen-rich mesoporous carbons: highly efficient, regenerable metal-free catalysts for low-temperature oxidation of H2S. ACS Catal 3:862–870
Paredes JI, Villar-Rodil S, Martinez-Alonso A, Tascón JMD (2008) Graphene oxide dispersions in organic solvents. Langmuir 24:10560–10564
Gholap S, Badiger M, Gopinath CS (2005) Molecular origins of wettability of hydrophobic poly(vinylidene fluoride) microporous membranes on poly(vinyl alcohol) adsorption: surface and interface analysis by XPS. J Phys Chem B 109:13941–13947
Wang Y, Li Z, Tian YF, Zhao W, Liu XQ, Yang JB (2014) A facile way to fabricate graphene sheets on TiO2 nanotube arrays for dye-sensitized solar cell applications. J Mater Sci 49:7991–7999. doi:10.1007/s10853-014-8506-7
Acknowledgements
The work is financially supported by the Natural Science Foundation of China (51273166), the Natural Science Foundation of the Fujian Province of China (2013J01206), and the Fundamental Research Funds for the Central Universities of China (CXB2014014).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ke, Q., Liao, Y., Yao, S. et al. A three-dimensional TiO2/graphene porous composite with nano-carbon deposition for supercapacitor. J Mater Sci 51, 2008–2016 (2016). https://doi.org/10.1007/s10853-015-9510-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-015-9510-2