Abstract
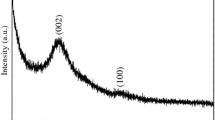
The addition of nano-carbons to composite materials is an area of significant research interest, when their addition results in improved properties. This work reports on the use of detonation nanodiamond (DND) in the preparation of porous carbon monoliths and an investigation of the properties of the final carbon–nanocarbon composite material. Porous carbon–nanodiamond (CND) monoliths, with macro-, meso- and micropores were prepared by carbonisation of a resorcinol-formaldehyde (RF) polymeric rod with an Fe(III) catalyst and spherical silica template. Pore characteristics and BET surface areas were determined from N2 isotherms, with surface areas in the range of 214–461 m2 g−1, depending on DND content. SEM imaging further confirmed the hierarchical pore structure present, where there was a trimodal structure for monoliths containing nanodiamond following pyrolysis up to 900 °C. Thermogravimetric analysis, TEM imaging, energy dispersive X-ray electron spectroscopy and Raman spectroscopy were employed to evaluate the properties of this new composite material. The adsorptions of methylene blue (MB) and neutral red (NR) dyes from water onto the composite monoliths were investigated and compared with activated carbon in order to further evaluate their physical and adsorptive properties. CND materials adsorb these two cationic dyes more effectively than activated carbon, due to a more accessible pore network, and DND content had a direct effect on adsorption capacities for the dyes. The adsorption isotherms coincided with Langmuir and Freundlich adsorption models. Maximum adsorption capacities of 599 and 284 mg g−1 were achieved for NR and MB, respectively, on the CND composites.











Similar content being viewed by others
References
Ruo-wen FU, Zheng-hui LI, Ye-ru L, Eng LI, Eei XU, Ding-cai WU (2011) Hierarchical porous carbons: design: preparation and performance in energy storage. New Carbon Mater 26:171–179
Upare DP, Yoon S, Lee CW (2011) Nano-structured porous carbon materials for catalysis and energy storage. Korean J Chem Eng 28:731–743
Gupta VK, Saleh TA (2013) Sorption of pollutants by porous carbon, carbon nanotubes and fullerene—an overview. Environ Sci Pollut Res 20:2828–2843
Pereira L (2008) Porous graphitic carbon as a stationary phase in hplc: theory and applications. J Liquid Chromatogr Relat Technol 31:1687–1731
Carneiro MC, Puignou L, Galceran MT (2000) Comparison of silica and porous graphitic carbon as solid phase extraction materials for the analysis of cationic herbicides in water by liquid chromatography and capillary electrophoresis. Anal Chim Acta 408:263–269
Hennion MC (2000) Graphitized carbons for solid phase extraction. J Chromatogr A 885:73–95
Eltmimi AH, Barron L, Rafferty A, Hanrahan JP, Fedyanina O, Nesterenko E, Nesterenko PN, Paull B (2010) Preparation, characterisation and modification of carbon-based monolithic rods for chromatographic applications. J Sep Sci 33:1231–1243
Liang C, Dai S, Guichon GA (2003) Graphitized carbon monolithic column. Anal Chem 75:4904–4912
He X, Male KB, Nesterenko PN, Brabazon D, Paull B, Luong JHT (2013) Adsorption and desorption of methylene blue on porous carbon monoliths and nanocrystalline cellulose. ACS Appl Mater Interfaces 5:8796–8804
Kim YS, Guo XF, Kim GJ (2010) Synthesis of carbon monolith with bimodal meso/macroscopic pore structure and its application in asymmetric catalysis. Catal Today 150:91–99
Garcia-Gomez A, Miles P, Centeno TA, Rojo JM (2010) Why carbon monoliths are better supercapacitor electrodes than compacted pellets. Electrochem Solid State Lett 13:112–114
Guichon G (2007) Monolithic columns in high-performance liquid chromatography. J Chromatogr A 1168:101–168
Kilduff JE, Karanfil T, Chin Y-P, Weber WJ (1996) Adsorption of natural organic polyelectrolytes by activated carbon: a size-exclusion chromatography study. Environ Sci Technol 30:1336–1343
Taguchi A, Smått JH, Lindén M (2003) Carbon monoliths possessing a hierarchical fully interconnected porosity. Adv Mater 15:1209–1211
Nesterenko EP, Nesterenko PN, Connolly D, He X, Floris P, Duffy E, Paull B (2013) Nano-particle modified stationary phases for high-performance liquid chromatography. Analyst 138:4229–4254
Herrera-Herrera AV, Gonzalez-Curbelo MA, Hernández-Borges J, Rodríguez-Delgado MA (2012) Carbon nanotubes applications in separation science: a review. Anal Chim Acta 734:1–30
Scida K, Stege PW, Haby G, Messina GA, Garcia CD (2011) Recent applications of carbon-based nanomaterials in analytical chemistry: critical review. Anal Chim Acta 691:6–17
Valcarel M, Cardenas S, Simonet BM, Moliner-Martinez Y, Lucena R (2008) Carbon nanostructures as sorbent materials in analytical processes. TrAC 27:34–43
Namera A, Nakamoto A, Saito T, Miyazaki S (2011) Monolith as a new sample preparation material: recent devices and applications. J Sep Sci 34:901–924
Moreno-Castilla C, Perez-Cadenas AF (2010) Carbon-based honeycomb monoliths for environmental gas-phase applications. Materials 3:1203–1227
Knox JH, Gilbert MT (1979) US Patent 4263268
Saleh TA, Gupta VK (2014) Processing methods, characteristics and adsorption behaviour of tire derived carbons: a review. Adv Colloid Interface Sci 211:93–101
Yagub MT, Sen TK, Afroze S, Ang HM (2014) Dye and its removal from aqueous solution by adsorption: a review. Adv Colloid Interface Sci 209:172–184
Liu F, Xu Z, Wan H, Wan Y, Zheng S, Zhu D (2011) Enhanced adsorption of humic acids on ordered mesoporous carbon compared with microporous activated carbon. Environ Toxicol Chem 30:793–800
Han S, Sohn K, Hyeon T (2000) Fabrication of new nanoporous carbons through silica templates and their application to the adsorption of bulky dyes. Chem Mater 12:3337–3341
Sevilla M, Fuertes AB (2013) Fabrication of porous carbon monoliths with a graphitic framework. Carbon 56:155–166
Sakintuna B, Yürüm Y (2005) Templated porous carbons: a review article. Ind Eng Chem Res 44:2893–2902
Alvarez S, Fuertes AB (2007) Synthesis of macro/mesoporous silica and carbon monoliths by using a commercial polyurethane foam as sacrificial template. Mater Lett 61:2378–2381
Tao S, Wang Y, Shi D, An Y, Qiu J, Zhao Y, Cao Y, Zhang X (2014) Facile synthesis of highly graphitized porous carbon monoliths with a balance on crystallization and pore-structure. J Mater Chem A 2:12785–12791
He X, Nesterenko EP, Nesterenko PN, Brabazon D, Zhou L, Glennon JD, Luong JHT, Paull B (2013) Fabrication and characterization of nanotemplated carbon monolithic material. ACS Appl Mater Interfaces 5:8572–8580
Duffy E, He X, Nesterenko EP, Dey A, Krishnamurthy S, Brabazon D, Nesterenko PN, Paull B (2015) Thermally controlled growth of carbon onions within porous graphitic carbon-detonation nanodiamond monolithic composites. RSC Adv 5:22906–22915
Nesterenko PN, Haddad PR (2010) Diamond-related materials as potential new media in separation science. Anal Bioanal Chem 396:205–211
Mochalin VM, Shenderova O, Ho D, Gogotsi Y (2012) The properties and applications of nanodiamonds. Nat Nanotechnol 7:11–23
Krueger A (2008) Diamond nanoparticles: jewels for chemistry and physics. Adv Mater 20:2445–2449
Peristyy AA, Fedyanina O, Paull B, Nesterenko PN (2014) Diamond based adsorbents and their application in chromatography. J Chromatogr A 1357:68–86
Wang DH, Tan L-S, Huang H, Dai L, Osawa E (2009) In-situ nanocomposite synthesis: arylcarbonylation and grafting of primary diamond nanoparticles with a poly(ether-ketone) in polyphosphoric acid. Macromolecules 42:114–124
Shenderova O, Jones C, Borjanovic V, Hens S, Cunningham G, Moseenkov S, Kuznetsov V, McGuire G (2008) Detonation nanodiamond and onion-line carbon: applications in composites. Physica Status Solidi (a) 205:2245–2251
Manandhar S, Roder PB, Hanson JL, Lim M, Smith BE, Mann A, Pauzauskie PJ (2014) Rapid sol-gel synthesis of nanodiamond aerogel. J Mater Res 29:2905–2911
Ostrovidova GU, Makeev AV, Biryukov AV, Gordeev SK (2003) Carbon nanocomposite materials as medicinal depot. Mater Sci Eng C 23:377–381
Shenderova OA, Zhirnov VV, Brenner DW (2002) Carbon nanostructures. Crit Rev Solid State Mater Sci 27:227–356
Choma J, Jedynak K, Fahrenholz W, Ludwinowicz J, Jaroniec M (2014) Microporosity development in phenolic resin-based mesoporous carbons for enhancing CO2 adsorption at ambient conditions. Appl Surf Sci 289:592–600
Sajad M, Kazemzad M, Hosseinnia A (2014) Preparation of activated carbon monolith by application of phenolic resins as carbon precursors. Funct Mater Lett 7:1450035
Robertson C, Mokaya R (2013) Microporous activated carbon aerogels via a simple subcritical drying route for CO2 capture and hydrogen storage. Microporous Mesoporous Mater 179:151–156
Kipling JJ, Wilson RB (1960) Adsorption of methylene blue in the determination of surface areas. J Appl Chem 10:109–113
Los JM, Tompkins CK (1956) Adsorption of methylene blue on a positively charged mercury surface. J Chem Phys 24:630
Taha-Tijerina JJ, Narayanan TN, Tiwary CS, Lozano K, Chipara M, Ajayan PM (2014) Nanodiamond-based thermal fluids. ACS Appl Mater Interfaces 6:4778–4785
Branson BT, Beauchamp PS, Beam JC, Lukehart CM, Davidson JL (2013) Nanodiamond nanofluids for enhanced thermal conductivity. ACS Nano 7:3183–3189
Barnard AS (2008) Self-assembly in nanodiamond agglutinates. J Mater Chem 18:4038–4041
Duffy E, Mitev D, Kazarian AA, Nesterenko PN, Paull B (2014) Separation and characterisation of detonation nanodiamond by capillary zone electrophoresis. Electrophoresis 35:1864–1872
Kuznetsov VL, Chuvilin AL, Butenko YV, Mal’kov IY, Titov VM (1994) Onion-like carbon from ultra-disperse diamond. Chem Phys Lett 222:343–348
Chen J, Deng SZ, Chen J, Yu ZX, Xu NS (1994) Graphitization of nanodiamond powder annealed in argon ambient. Appl Phys Lett 74:3651–3653
Lowell S, Shields JE, Thomas MA, Thommes M (2004) Characterization of porous solids and powders: surface area, pore size and density. Kluwer Academic Publishers, London
Ko T-H, Kuo W-S, Chang Y-H (2001) Microstructural changes of phenolic resin during pyrolysis. J Appl Polym Sci 81:1084–1089
Batsanov SS, Lesnikov EV, Dan’kin DA, Balakhanov DM (2014) Water shells of diamond nanoparticles in colloidal solutions. Appl Phys Lett 104:133105
Fang XW, Mao JD, Levin EM, Schmidt-Rohr K (2009) Nonaromatic core-shell structure of nanodiamond from solid-state NMR spectroscopy. J Am Chem Soc 131:1426–1435
Ko TH, Kuo WS, Lu YR (2000) The influence of post-cure on properties of carbon/phenolic resin cured composites and their final carbon/carbon composites. Polym Compos 21:96–103
Mitev DM, Townsend AT, Paull B, Nesterenko PN (2014) Microwave-assisted purification of detonation nanodiamond. Diam Relat Mater 48:37–46
Xu NS, Chen J, Deng SZ (2002) Effect of heat treatment on the properties of nano-diamond under oxygen and argon ambient. Diam Relat Mater 11:249–256
Tuinstra F, Koenig JL (1970) Raman spectrum of graphite. J Chem Phys 53:1126–1130
Ferrari AC, Robertson J (2000) Interpretation of raman spectra of disordered and amorphous carbon. Phys Rev B 61:14095–14107
Yuan X, Shu-** Z, Wei X, Hong-You C, **ao-Dong D, **n-Mei L, Zi-Feng Y (2007) Aqueous dye adsorption on ordered mesoporous carbons. J Colloid Interface Sci 310:83–89
Wu F-C, Tseng R-L, Juang R-S (2005) Preparation of highly microporous carbons from fir wood by KOH activation for adsorption of dyes and phenols from water. Sep Purif Technol 47:10–19
Dabrowski A (2001) Adsorption—from theory to practice. Adv Colloid Interface Sci 93:135–224
Wang S, Wei J, Lv S, Guo Z, Jiang F (2013) Removal of organic dyes in environmental water onto magnetic-sulfonic graphene nanocomposite. CLEAN 41:751–764
Desta MB (2013) Batch sorption experiments: langmuir and freundlich isotherm studies for the adsorption of textile metal ions onto teff straw (Eragrostis Tef) agricultural waste. J Thermodyn 2013:375830
Poots VJP, McKay G, Healy JJ (1978) Removal of basic dye from effluent using wood as an adsorbent. J Water Pollut Control F 50:926–935
Hall KR, Eagleton LC, Acrivos A, Vermeulen T (1966) Pore- and solid-diffusion kinetics in fixed-bed adsorption under constant-pattern conditions. Ind Eng Chem Fundamen 5:212–223
Acknowledgements
The authors are grateful to the Australian Research Council for the financial support in the form of Australian Research Council Discovery Grants DP110102046 and DP150102608. E.D. would also like to thank Dr. Ekaterina P. Nesterenko and Mrs. Heather Davies for technical assistance. The authors also acknowledge the technical support imaging materials received from Dr. Satheesh Krishnamurthy (TEM), Dr. Karsten Goemann and Dr. Sandrin Feig (SEM).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Duffy, E., He, X., Nesterenko, P.N. et al. Hierarchical porous graphitic carbon monoliths with detonation nanodiamonds: synthesis, characterisation and adsorptive properties. J Mater Sci 50, 6245–6259 (2015). https://doi.org/10.1007/s10853-015-9195-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-015-9195-6