Abstract
Purpose
The objective of this study is to compare the chromosomal distribution of early miscarriages with or without embryonic poles.
Materials and methods
It was a retrospective study of 223 women who underwent dilation and curettage (D&C) between 1995 and 2013 for early miscarriages. The presence or absence of a fetal pole was evaluated by abdominal or transvaginal ultrasound. Cytogenetic tests of products of conception following culture were determined in both groups.
Results
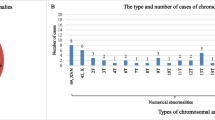
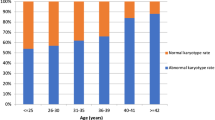
Of the 223 early miscarriages, 143 had embryos and 80 did not. The abnormality rate differed significantly (61.5 % vs. 46.3 %, p < 0.05), with trisomy 18, 21 and 45X found only in miscarriages with embryos. There were no significant differences between groups in rates of triploidy, tetraploidy, mosaicism, structure and double abnormality. The female abortus rate was higher in miscarriages with or without embryonic poles, as well as in groups with normal and abnormal karyotypes.
Conclusions
Chromosome distribution differs in miscarriages with or without embryonic poles. The ultrasound findings might offer different direction to determine the causes of early miscarriages. The higher female abortus rate may be associated with early selection.

Similar content being viewed by others
References
Jurkovic D, Overton C, Bender-Atik R. Diagnosis and management of first trimester miscarriage. BMJ. 2013;346:f3676.
Zhang HK, Luo FW, Geng Q, et al. [Analysis of fetal chromosomal karyotype and etiology in 252 cases of early spontaneous abortion]. Zhonghua yi xue yi chuan xue za zhi = Zhonghua yixue yichuanxue zazhi =. Chin J Med Genet. 2011;28:575–8.
van den Berg, van Maarle, van Wely, et al. Genetics of early miscarriage. Biochim Biophys Acta. 2012;1822(12):1951–9.
Munoz M, Arigita M, Bennasar M, et al. Chromosomal anomaly spectrum in early pregnancy loss in relation to presence or absence of an embryonic pole. Fertil Steril. 2010;94:2564–8.
Lathi RB, Mark SD, Westphal LM, et al. Cytogenetic testing of anembryonic pregnancies compared to embryonic missed abortions. J Assist Reprod Genet. 2007;24:521–4.
Lathi RB, Gustin SL, Keller J, et al. Reliability of 46, XX results on miscarriage specimens: a review of 1,222 first-trimester miscarriage specimens. Fertil Steril. 2014;101:178–82.
Evdokimova VN, Nikitina TV, Lebedev IN, et al. Sex ratio in early embryonal mortality in man. Ontogenez. 2000;31:251–7.
Grech V. Secular trends and latitude gradients in sex ratio at birth in Asia during the past 60 years. Pediatr Int: Off J Jpn Pediatr Soc. 2013;55:219–22.
Lee IW, Lai YC, Kuo PL, et al. Human sex ratio at amniocentesis and at birth in Taiwan. Taiwan J Obstet Gynecol. 2012;51:572–5.
Lathi RB, Milki AA. Tissue sampling technique affects accuracy of karyotype from missed abortions. J Assist Reprod Genet. 2002;19:536–8.
Qin JZ, Pang LH, Li MQ, et al. Risk of chromosomal abnormalities in early spontaneous abortion after assisted reproductive technology: a meta-analysis. PLoS One. 2013;8(10):e75953.
Minelli E, Buchi C, Granata P, et al. Cytogenetic findings in echographically defined blighted ovum abortions. Ann Genet. 1993;36:107–10.
Hsu TY, Liou JD, Copel JA, et al. Prenatal detection of two different monosomic cell lines by chorionic villus sampling. Prenat Diagn. 1996;16:169–72.
Chen C-P. Placental abnormalities and preeclampsia in trisomy 13 pregnancies. Taiwan J Obstet Gynecol. 2009;48(1):3–8.
Philipp T, Kalousek DK. Generalized abnormal embryonic development in missed abortion: embryoscopic and cytogenetic findings. Am J Med Genet. 2002;111:43–7.
Bell KA, Van Deerlin PG, Haddad BR, et al. Cytogenetic diagnosis of “normal 46, XX” karyotypes in spontaneous abortions frequently may be misleading. Fertil Steril. 1999;71:334–41.
Tatyana V, Igor N, Natalia N, et al. A mathematical model for evaluation of maternal cell contamination in cultured cells from spontaneous abortions: significance for cytogenetic analysis of prenatal selection factor. Fertil Steril. 2005;83:964–72.
Avery B, Madison V, Greve T. Sex and development in bovine in-vitro fertilized embryos. Theriogenology. 1991;35:953–63.
Nedambale TL, Dinnyes A, Yang X, et al. Bovine blastocyst development in vitro: timing, sex, and viability following vitrification. Biol Reprod. 2004;71:1671–6.
Hoshi N, Hanatani K, Kishida T, et al. Chromosomal analysis in 894 induced abortuses from women of advanced maternal age in relation to gestational weeks and fetal sex ratio. J Obstet Gynaecol Res. 1997;23:1–7.
Matsuo K, Ushioda N, Udoff LC. Parental aging synergistically decreases offspring sex ratio. J Obstet Gynaecol Res. 2009;35:164–8.
Chan BC, Lao TT. Effect of parity and advanced maternal age on obstetric outcome. Int J Gynaecol Obstet Off Organ Int Fed Gynaecol Obstet. 2008;102:237–41.
Mizuno R. The male/female ratio of fetal deaths and births in Japan. Lancet. 2000;356:738–9.
Morales C, Sanchez A, Bruguera J, et al. Cytogenetic study of spontaneous abortions using semi-direct analysis of chorionic villi samples detects the broadest spectrum of chromosome abnormalities. Am J Med Genet A. 2008;146A:66–70.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Capsule Trisomy 18, 21 and monosomy X were found in miscarriages with embryos. The female abortus rate was higher in miscarriages with or without embryos.
Rights and permissions
About this article
Cite this article
Cheng, HH., Ou, CY., Tsai, CC. et al. Chromosome distribution of early miscarriages with present or absent embryos: female predominance. J Assist Reprod Genet 31, 1059–1064 (2014). https://doi.org/10.1007/s10815-014-0261-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-014-0261-9