Abstract
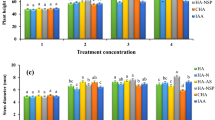
The aim of the present investigation was to study the impact of foliar spraying with Chlorella vulgaris, Arthrospira platensis, and Tetradesmus dimorphus suspensions as biostimulants on growth and yield characteristics of common beans such as Phaseolus vulgaris. Seven treatments were tested during the study: T1 (soil amended with N-urea but no microalgae foliar application), T2 (foliar spraying with C. vulgaris but no N-urea added to the soil), T3 (foliar spraying with T. dimorphus but no N-urea added to the soil), T4 (foliar spraying with A. platensis but no N-urea added to the soil), T5 (foliar spraying with C. vulgaris and soil amended with N-urea), T6 (foliar spraying with T. dimorphus and soil amended with N-urea), T7 (foliar spraying with A. platensis and soil amended with N-urea) and control (untreated). Foliar spraying was applied after 7, 25, and 77 days from sowing using the test microalgae suspensions in concentration of 10 g 100 mL−1. Plant growth and biochemical parameters were measured at the end of both vegetative and fruiting growth stages. Compared with control, the treatments from T1 to T7 showed noticeable increase in all growth parameters and yield attribute. The foliar application with C. vulgaris and chemical fertilizer treated plants (T5) exhibited the maximum increase in total plant height (26.9%), dry weight (37.28%), protein content (48.06 ± 2.403 mg g−1 fresh wt.), and total carbohydrate (394 ± 19.7 mg g−1 dry wt.) during vegetative stage as well as number of pods per plant (5.2 ± 0.26), number of seed/pod (3.5 ± 0.18), pods dry weight (0.95 ± 0.05 g plant−1) during fruiting stage. Thus, it is advisable to use C. vulgaris as a biostimulant for enhancing P. vulgaris growth and crop production.





Similar content being viewed by others
Availability of data
The authors confirm that all the data are available within the article.
References
Anagnostidis K, Komárek J (1988) Modern approach to the classification system of cyanophytes. 3-Oscillatoriales. Algol Stud / Arch Hydrobiol Suppl 50–53:327–472
Arnon DI (1949) Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant Physiol 24:1–15
Barone V, Puglisi I, Fragalà F, Stevanato P, Baglieri A (2019) Effect of living cells of microalgae or their extracts on soil enzyme activities. Arch Agron Soil Sci 65:712–726
Bayona-Morcillo P, Plaza B, Gómez-Serrano C, Rojas E, Jiménez-Becker S (2020) Effect of the foliar application of cyanobacterial hydrolysate (Arthrospira platensis) on the growth of Petunia x hybrida under salinity conditions. J Appl Phycol 32:4003–4011
Battacharyya D, Babgohari MZ, Rathor P, Prithiviraj B (2015) Seaweed extracts as biostimulants in horticulture. Sci Hort 196:39–48
Bischoff H (1963) Some soil algae from enchanted rock and related algal species. Phycological Studies IV. University of Texas Publ No 6318:1–95
Black C (1965) Method of soil analysis part 2. Chem Microbiol Properties 9:1387–1388
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Bumandalai O, Tserennadmid R (2019) Effect of Chlorella vulgaris as a biofertilizer on germination of tomato and cucumber seeds. Int J Aquat Biol 7:95–99
Chiaiese P, Corrado G, Colla G, Kyriacou MC, Rouphael Y (2018) Renewable sources of plant biostimulation: microalgae as a sustainable means to improve crop performance. Front Plant Sci 9:1782
Chookalaii H, Riahi H, Shariatmadari Z, Mazarei Z, SeyedHashtroudi M (2020) Enhancement of total flavonoid and phenolic contents in Plantago major L. with plant growth promoting cyanobacteria. J Agric Sci Technol 22:505–518
Dias GA, Rocha RHC, Araújo JL, Lima JF, Guedes WA (2016) Growth, yield, and postharvest quality in eggplant produced under different foliar fertilizer (Arthrospira platensis) treatments. Semina Ciênc Agrár 37:3893–3902
Díaz-Batalla L, Widholm JM, Fahey GC, Castaño-Tostado E, Paredes-López O (2006) Chemical components with health implications in wild and cultivated Mexican common bean seeds (Phaseolus vulgaris L.). J Agric Food Chem 54:2045–2052
Dineshkumar R, Kumaravel R, Gopalsamy J, Sikder MNA, Sampathkumar P (2018) Microalgae as bio-fertilizers for rice growth and seed yield productivity. Waste Biomass Valor 9:793–800
Dineshkumar R, Subramanian J, Gopalsamy J, Jayasingam P, Arumugam A, Kannadasan S, Sampathkumar P (2019) The impact of using microalgae as biofertilizer in maize (Zea mays L.). Waste Biomass Valor 10:1101–1110
Du Jardin P (2015) Plant biostimulants: definition, concept, main categories and regulation. Sci Hort 196:3–14
Elarroussia H, Elmernissia N, Benhimaa R, El Kadmiria IM, Bendaou N, Smouni A, Wahbya I (2016) Microalgae polysaccharides a promising plant growth biostimulant. J Algal Biomass Utln 7:55–63
Fernández V, Sotiropoulos T, Brown PH (2013) Foliar fertilization: scientific principles and field practices. IFA, Paris pp 12–70
Garcia-Gonzalez J, Sommerfeld M (2016) Biofertilizer and biostimulant properties of the microalga Acutodesmus dimorphus. J Appl Phycol 28:1051–1061
Gorelova O (2006) Communication of cyanobacteria with plant partners during association formation. Microbiology 75:465–469
Hedge J, Hofreiter B, Whistler R (1962) Carbohydrate chemistry. Academic Press, New York
Horvath G, Kissimon J, Faludi-Dániel Á (1972) Effect of light intensity on the formation of carotenoids in normal and mutant maize leaves. Phytochemistry 11:183–187
Kawalekar JS (2013) Role of biofertilizers and biopesticides for sustainable agriculture. J Biol Innov 2:73–78
Khan SA, Hussain MZ, Prasad S, Banerjee U (2009) Prospects of biodiesel production from microalgae in India. Renew Sustain Energy Rev 13:2361–2372
Kim MJ, Shim CK, Kim YK, Ko BG, Park JH, Hwang SG, Kim BH (2018) Effect of biostimulator Chlorella fusca on improving growth and qualities of chinese chives and spinach in organic farm. Plant Pathol J 34:567
Kissimon J (1999) Analysis of the photosynthetic pigment composition. International workshop and training course on microalgal biology and biotechnology. Mosonmagyarouar, Hungary, pp 13–26
Komárek J, Fott B, Huber-Pestalozzi G (1983) Das Phytoplankton des Süßwassers Systematik und Biologie-Teil 7, 1. Schweizerbart Science Publishers, Stuttgart
Lin L-Z, Harnly JM, Pastor-Corrales MS, Luthria DL (2008) The polyphenolic profiles of common bean (Phaseolus vulgaris L.). Food Chem 107:399–410
Mógor ÁF, Ördög V, Lima GPP, Molnár Z, Mógor G (2018) Biostimulant properties of cyanobacterial hydrolysate related to polyamines. J Appl Phycol 30:453–460
Mourice SK, Tryphone GM (2012) Evaluation of common bean (Phaseolus vulgaris L.) genotypes for adaptation to low phosphorus. Int Schl Res Not 2012:309614
Nagy PT, Pintér T (2015) Effects of foliar biofertilizer sprays on nutrient uptake, yield, and quality parameters of Blaufrankish (Vitis vinifera L.) Grapes. Comm Soil Sci Plant Anal 46:219–227
Osman MEH, El-Sheekh MM, El-Naggar AH, Gheda SF (2010) Effect of two species of cyanobacteria as biofertilizers on some metabolic activities, growth, and yield of pea plant. Biol Fertil Soils 46:861–875
Plaza BM, Gómez-Serrano C, Acién-Fernández FG, Jimenez-Becker S (2018) Effect of microalgae hydrolysate foliar application (Arthrospira platensis and Tetradesmus sp.) on Petunia x hybrida growth. J Appl Phycol 30:2359–2365
Porch TG, Beaver JS, Debouck DG, Jackson SA, Kelly JD, Dempewolf H (2013) Use of wild relatives and closely related species to adapt common bean to climate change. Agronomy 3:433–461
Priya TR, Manickavasagan A (2020) Common bean. In: Manickavasagan A, Thirunathan P (eds) Pulses. Springer, Cham, pp 77–97
Puglisi I, La Bella E, Rovetto EI, Lo Piero AR, Baglieri A (2020) Biostimulant effect and biochemical response in lettuce seedlings treated with a Tetradesmus quadricauda extract. Plants 9:123
Raschke RL, Schultz DA (1987) The use of the algal growth potential test for data assessment. J Water Pollut Control Fed 222–227
Ronga D, Biazzi E, Parati K, Carminati D, Carminati E, Tava A (2019) Microalgal biostimulants and biofertilisers in crop productions. Agronomy 9:192
Sadasivam S, Manickam A (1996) Biochemical methods. New Age International (P) Limited, New Delhi 2:124–126
Shaaban MM (2001a) Green microalgae water extract as foliar feeding to wheat plants. Pak J Biol Sci 4:628–632
Shaaban MM (2001b) Nutritional status and growth of maize plants as affected by green microalgae as soil additives. J Biol Sci 1:475–479
Shindy WW, Smith OE (1975) Identification of plant hormones from cotton ovules. Plant Physiol 55:550–554
Siddiq M, Ravi R, Harte J, Dolan K (2010) Physical and functional characteristics of selected dry bean (Phaseolus vulgaris L.) flours. LWT - Food Sci Technol 43:232–237
Starr RC (1978) The culture collection of algae at the University of Texas at Austin. J Phycol 14:47–100
Stein JR, Hellebust JA, Craigie J (1973) Handbook of phycological methods: culture methods and growth measurements. Cambridge University Press, Cambridge
Stoscheck CM (1990) Quantitation of protein. Meth Enzymol 182:50–68
Tarakhovskaya E, Maslov YI, Shishova M (2007) Phytohormones in algae. Russ J Plant Physiol 54:163–170
Van Handel E (1968) Direct micro determination of sucrose. Anal Biochem 22:280–283
Yemm E, Willis A (1954) The estimation of carbohydrates in plant extracts by anthrone. Biochem J 57:508–514
Zarrouk C (1966) Contribution a l'etude d'une Cyanophycee. Influence de Divers Facteurs Physiques et Chimiques sur la croissance et la photosynthese de Spirulina maxima. Thesis. University of Paris, France
Acknowledgements
The authors would like to express their gratitude to the Botany Department at Mansoura University in Egypt for allowing them to complete these experiments and for providing financial support.
Author information
Authors and Affiliations
Contributions
D.A.R. prepared the manuscript. D.A.R., E.M.E., M.I.A, and S.A.H conducted the experiments. S.A.H and D.A.R. designed the study. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Refaay, D.A., El-Marzoki, E.M., Abdel-Hamid, M.I. et al. Effect of foliar application with Chlorella vulgaris, Tetradesmus dimorphus, and Arthrospira platensis as biostimulants for common bean. J Appl Phycol 33, 3807–3815 (2021). https://doi.org/10.1007/s10811-021-02584-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-021-02584-z