Abstract
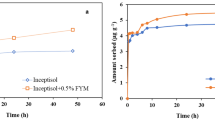
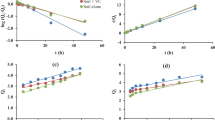
Laboratory study on adsorption–desorption of flubendiamide was conducted in two soil types, varying in their physical and chemical properties, by batch equilibrium method. After 4 h of equilibrium time, adsorption of flubendiamide on soil matrix exhibited moderately low rate of accumulation with 4.52 ± 0.21 % in red soil and low rate with 3.55 ± 0.21 % in black soil. After amending soils with organic manure, adsorption percentage increased to 6.42 ± 0.21 % in red soil and (4.18 ± 0.21 %) in black soil indicating that amendment significantly increased sorption. Variation in sorption affinities of the soils as indicated by distribution coefficient (K d) for sorption was in the range of 2.98–4.32, 4.91–6.64, 1.04–1.45 and 1.92–2.81 ml/g for red soil, organic manure-treated red soil, black soil and organic manure-treated black soil, respectively. Desorption was slightly slower than adsorption indicating a hysteresis effect having hysteresis coefficient ranges between 0.023 and 0.149 in two test soils. The adsorption data for the insecticide fitted well the Freundlich equation. Results revealed that adsorption–desorption was influenced by soil types and showed that the maximum sorption and minimum desorption of the insecticide was observed in soils with higher organic carbon and clay content. It can be inferred that crystal lattice of the clay soil plays a significant role in flubendiamide adsorption and desorption. Adsorption was lower at acidic pH and gradually increased towards alkaline pH. As this insecticide is poorly sorbed in the two Indian soil types, there may be a possibility of their leaching to lower soil profiles.





Similar content being viewed by others
References
Bailey, G. W., & White, J. L. (1964). Review of adsorption and desorption of organic pesticides by soil colloids with implications concerning pesticide bioactivity. Journal of Agriculture & Food Chemistry, 12, 324–332.
Bailey, G. W., White, J. L., & Rothberg, T. (1968). Adsorption of organic herbicides by montmorillonite: role of pH and chemical characters of the adsorbate. Soil Science Society of America, Proceedings, 32, 222–234.
Beyer, L., Ahlsdorf, B., Sorge, C., Schulton, H. R., & Blume, H. P. (1996). Soil organic matter composition and pesticide bonding in sandy soils in relation to groundwater protection in north-west German lower plain. Biology and Fertility of Soils, 23, 266–272.
Black, C. A., Evans, D. D., White, J. L., Ensminger, L. E., & Clark, F. E. (Eds.) (1965). Methods of soil analysis, 2nd edn. In Agronomy monograph 9. Madison: Agronomy Society of America and Soil Science Society of America.
Blakemore, L.C., Searle, P.L., & Daly, B.K. (1987). Methods for chemical analysis of soils. NZ Soil Bureau Scientific Report, series 80. New Zealand: NZ DSIR, 103 p.
Cohen, S.Z., Creeger, S.M., Carsel, R.F., & Enfield, C.G. (1984). Potential pesticide contamination of groundwater from agricultural uses. In R.F. Krueger, & J.N. Seiber (Eds.), Treatment and disposal of pesticide wastes, ACS Symposium Series No. 259 (pp. 297–325). Washington, DC: American Chemical Society.
Cox, L., Koskinen, W. C., & Yen, P. Y. (1997). Sorption–desorption of imidacloprid and its metabolites in soils. Journal of Agricultural and Food Chemistry, 45, 1468–1472.
Das, S. K., & Mukherjee, I. (2011). Effect of light and pH on persistence of flubendiamide. Bulletin of Environmental Contamination and Toxicology, 87, 292–296.
Das, S. K., & Mukherjee, I. (2012). Effect of moisture and organic manure on persistence of flubendiamide in soil. Bulletin of Environmental Contamination and Toxicology, 88, 515–520.
Das, S. K., & Mukherjee, I. (2014). Influence of microbial community on degradation of flubendiamide in two Indian soils. Environmental Monitoring and Assessment, 186, 3213–3219.
Das, S. K., Mukherjee, I., & Das, S. K. (2012). Dissipation of flubendiamide in/on Okra [Abelmoschus esculenta (L.) Moench] Fruits. Bulletin of Environmental Contamination and Toxicology, 88, 381–384.
Das, S. K., Avasthe, R. K., Singh, R., & Babu, S. (2014). Biochar as carbon negative in carbon credit under changing climate. Current Science, 107, 1090–1091.
Deng, J., Jiang, X., Hu, W., & Hu, L. (2010). Quantifying hysteresis of atrazine desorption from a sandy loam soil. Journal of Environmental Sciences, 22, 1923–1929.
Drori, Y., Aizenshtat, Z., & Chefetz, B. (2005). Sorption–desorption behavior of atrazine in soils irrigated with reclaimed wastewater. Soil Science Society of America Journal, 69, 1703–1710.
Ebbinghaus-Kintscher, U., Lummen, P., Raming, K., Masaki, T., & Yasokawa, N. (2007). Flubendiamide, the first insecticide with a novel mode of action on insect ryanodine receptors. Pf lanzenschutz-Nachrichten Bayer, 60, 117–140.
Fernandes, M. C., Cox, L., Hermosin, M. C., & Cornejo, J. (2006). Organic amendments affecting sorption, leaching and dissipation of fungicides in soils. Pest Management Science, 62, 1207–1215.
Gajbhiye, V. T., & Gupta, S. (2001). Adsorption-desorption behavior of flufenacet in five different soils of India. Pest Management Science, 57, 633–639.
Gannon, T. W., Hixson, A. C., Weber, J. B., Shi, W., Yelverton, F. H., & Rufty, T. W. (2013). Sorption of simazine and S-metolachlor to soils from a chronosequence of turf grass systems. Weed Science, 61, 508–514.
Gustafson, D. I. (2002). Groundwater ubiquity score: a simple method for assessing pesticide leachability. Environmental Toxicology and Chemistry, 8, 339–357.
Hamaker, J. W., & Thompson, J. M. (1992). Adsorption. In C. A. I. Goring & J. W. Hamaker (Eds.), Organic chemical in the soil environment (pp. 49–144). New York: Marcel Dekker Inc.
Helling, C. S., Kearney, P. C., & Alexander, M. (1971). Behavior of pesticides in soils. Advances in Agronomy, 23, 147–240.
Ismail, B. S., & Kalithasan, K. (2004). Adsorption, desorption and mobility of permethrin in Malaysian soils. Journal of Environmental Science and Health. Part. B, 39, 419–429.
Ismail, B. S., & Ooi, K. E. (2012). Sorption, desorption and mobility of metsulfuron-methyl in soils of the oil palm agro ecosystem in Malaysia. Journal of Environmental Biology, 33, 573–577.
Jackson, M. L. (1967). Soil chemical analysis. New Delhi: Prentice Hall.
Jana, T. K., & Das, B. (1997). Sorption of carbaryl (1-Naphthyl N-methyl carbamate) by soil. Bulletin of Environmental Contamination and Toxicology, 59, 65–71.
Khoury, R., Geanchan, A., Corte, C. M., Cooper, J. F., & Bobe, A. (2003). Retention and degradation of metribuzin in sandy loam and clay soils of Lebanon. Weed Research, 43, 252–259.
Kumar, N., & Mukherjee, I. (2013). Effect of soil physicochemical properties on adsorption of tricyclazole. International Journal of Agriculture and Food Science Technology, 4, 391–396.
Li, H., Sheng, G., Teppan, B. J., Johnston, C. T., & Boyd, S. A. (2003). Sorption and desorption of pesticides by clay minerals and humic acid-clay complexes. Soil Science Society America Journal, 67, 122–131.
Masaki, T., Yasokawa, N., Tohnishi, M., Motoba, K., & Hirooka, T. (2006). Flubendiamide: a novel Ca+2 channel modulator, reveals evidence for functional cooperation between Ca+2 pumps and Ca+2 release. Molecular Pharmacology, 69, 1733–1739.
Murphy, E. M., Zachara, J. M., Smith, S. C., & Phillips, J. L. (1992). The sorption of humic acids to mineral surfaces and their role in contaminant binding. Science of Total Environment, 117, 413–423.
Rhodes, R. C., Belasco, I. J., & Pease, H. L. (1970). Determination of mobility and sorption of agrochemicals on soils. Journal of Agricultural and Food Chemistry, 18, 524–527.
Singh, N., Kloeppel, H., & Klein, W. (2001). Sorption behavior of metolachlor, isoproturon and terbuthylazine in soils. Journal of Environmental Science and Health. Part. B, 36, 397–407.
Spark, K. M., & Swift, R. S. (2002). Effect of soil composition and dissolved organic matter on pesticide sorption. Science of Total Environment, 298, 147–161.
USEPA. (2008). Pesticide fact sheet, flubendiamide. Office of prevention, pesticides and toxic substances. http://www.epa.gov/opprd001/factsheet/flubendiamide.pdf. Accessed on 1st July 2010.
Wauchope, R. D., Yeh, S., Linders, J. B., Kloskowski, R., Tanaka, K., Rubin, B., Katayama, A., Kordel, W., Gerstl, Z., Lane, M., & Unsworth, J. B. (2002). Pesticide soil sorption parameters: theory, measurement, uses, limitations and reliability. Pest Management Science, 58(5), 419–445.
Zhang, J., Yang, L., Wei, L., Du, X., Zhou, L., Jiang, L., Ding, Q., & Yang, H. (2012). Environmental impact of two organic amendments on sorption and mobility of propachlor in soils. Journal of Soils Sediments, 12, 1380–1388.
Acknowledgments
The authors thank the head of the Division of Agricultural Chemicals, IARI, New Delhi, India, for providing the facilities to carry out this work (contribution no. 1066).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Das, S.K., Mukherjee, I. & Kumar, A. Effect of soil type and organic manure on adsorption–desorption of flubendiamide. Environ Monit Assess 187, 403 (2015). https://doi.org/10.1007/s10661-015-4623-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-015-4623-2