Abstract
Background
Administration of trastuzumab, a fully humanized monoclonal antibody targeted to the human epidermal growth factor receptor 2 (HER2, p185), has improved outcomes for patients with HER2-positive gastric cancer (GC), but some relevant issues remain to be investigated and will emerge with new anti-GC drugs. Gastrin is a major gastrointestinal hormone proven to have an inhibitory effect on GC in vitro and in vivo.
Aim
To explore the sympathetic role of trastuzumab and gastrin on inhibition of GC.
Methods
The HER2-positive and HER2-negative GC cell lines were treated with trastuzumab, gastrin, or their combination in vitro and in xenograft model. The synergistical role of trastuzumab and gastrin and related mechanisms were investigated.
Results
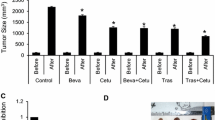
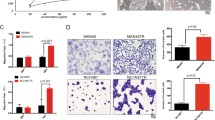
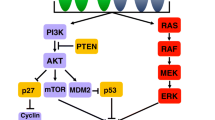
We found the synergistic inhibitory effects of trastuzumab and gastrin on HER2-negative GC cells through the gastrin/cholecystokinin B receptor (CCKBR) pathway. Trastuzumab upregulated CCKBR protein levels but could not initiate its signal transduction, whereas gastrin increased the levels and activation of CCKBR. Molecular experiments indicated that trastuzumab and gastrin co-treatment synergistically enhanced the stability of CCKBR. Moreover, their combined treatment synergistically arrested GC cells at G0/G1 phase, down-regulated levels of GC-related proteins, including anion exchanger 1 (AE1), cyclin D1, β-catenin, and cytoplasmic p16, and promoted nuclear translocation of p16. In addition, combination treatment upregulated AE2 levels, which are reduced in GC tissues. The in vivo synergistic anti-GC effect of combined treatment was confirmed in xenograft experiments.
Conclusions
Trastuzumab plus gastrin inhibit growth of Her2-negative GC by targeting cytoplasmic AE1 and p16.






Similar content being viewed by others
References
Kamangar F, Dores GM, Anderson WF. Patterns of cancer incidence, mortality, and prevalence across five continents: defining priorities to reduce cancer disparities in different geographic regions of the world. J Clin Oncol. 2006;24:2137–2150.
Parkin DM, Pisani P, Ferlay J. Global cancer statistics, 2002. CA Cancer J Clin. 2005;55:74–108.
Nashimoto A, Yabusaki H, Matsuki A, et al. Surgical treatment for curable and incurable recurrent gastric cancer. Gan To Kagaku Ryoho. 2013;40:971–975.
Yang W, Raufi A, Klempner SJ. Targeted therapy for gastric cancer: molecular pathways and ongoing investigations. Biochim Biophys Acta. 2014;1846:232–237.
Jorgensen JT. Targeted HER2 treatment in advanced gastric cancer. Oncology. 2010;78:26–33.
Gomez-Martin C, Lopez-Rios F, Aparicio J, et al. A critical review of HER2-positive gastric cancer evaluation and treatment: from trastuzumab, and beyond. Cancer Lett. 2014;351:30–40.
Kuo HY, Yeh KH. Molecular-targeted therapy for chemotherapy–refractory gastric cancer: a case report and literature review. Anticancer Res. 2014;34:3695–3699.
Bang YJ, Cutsem EV, Feyereislova A, et al. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial. Lancet. 2010;376:687–697.
Sheng WQ, Huang D, Ying JM, et al. HER2 status in gastric cancers: a retrospective analysis from four Chinese representative clinical centers and assessment of its prognostic significance. Ann Oncol. 2013;24:2360–2364.
Gravalos C, Jimeno A. HER2 in gastric cancer: a new prognostic factor and a novel therapeutic target. Ann Oncol. 2008;19:1523–1529.
Sachs G, Prinz C, Loo D, et al. Gastric acid secretion: activation and inhibition. Yale J Biol Med. 1994;67:81–95.
Quattrone A, Barbara D, Wozniak A, et al. Promoting role of cholecystokinin 2 receptor (CCK2R) in gastrointestinal stromal tumour pathogenesis. J Pathol. 2012;228:565–574.
Zhai HH, Meng J, Wang JB, et al. CacyBP/SIP nuclear translocation induced by gastrin promotes gastric cancer cell proliferation. World J Gastroenterol. 2014;20:10062–10070.
Xu W, Chen GS, Shao Y, et al. Gastrin acting on the cholecystokinin2 receptor induces cyclooxygenase-2 expression through JAK2/STAT3/PI3K/Akt pathway in human gastric cancer cells. Cancer Lett. 2013;332:11–18.
Muerkoster S, Isberner A, Arlt A, et al. Gastrin suppresses growth of CCK2 receptor expressing colon cancer cells by inducing apoptosis in vitro and in vivo. Gastroenterology. 2005;129:952–968.
Song LJ, Liu RJ, Zeng Z, et al. Gastrin inhibits a novel, pathological colon cancer signaling pathway involving EGR1, AE2, and P-ERK. J Mol Med (Berl). 2012;90:707–718.
Cobb S, Wood T, Tessarollo L, et al. Deletion of functional gastrin gene markedly increases colon carcinogenesis in response to azoxymethane in mice. Gastroenterology. 2002;123:516–530.
Kanno N, Glaser S, Chowdhury U, et al. Gastrin inhibits cholangiocarcinoma growth through increased apoptosis by activation of Ca2+-dependent protein kinase C-alpha. J Hepatol. 2001;34:284–291.
Detjen K, Fenrich MC, Logsdon CD. Transfected cholecystokinin receptors mediate growth inhibitory effects on human pancreatic cancer cell lines. Gastroenterology. 1997;112:952–959.
Sebens Muerkoster S, Rausch AV, Isberner A, et al. The apoptosis-inducing effect of gastrin on colorectal cancer cells relates to an increased IEX-1 expression mediating NF-kappa B inhibition. Oncogene. 2008;27:1122–1134.
Tian H, Zhang N, Suo WH, et al. Gastrin suppresses the interdependent expression of p16 and anion exchanger 1 favoring growth inhibition of gastric cancer cells. Int J Cancer. 2010;127:1462–1474.
Kang JM, Kim N, Yoo JY, et al. The role of serum pepsinogen and gastrin test for the detection of gastric cancer in Korea. Helicobacter. 2008;13:146–156.
Ito M, Tanaka S, Maeda M, et al. Role of the gastrin–gastrin receptor system in the expansive growth of human gastric neoplasms. Digestion. 2008;78:163–170.
Konturek SJ, Konturek PC, Bielanski W, et al. Serum progastrin and its products, gastric acid secretion and serum pepsinogen I in gastric cancer. Digestion. 2003;68:169–177.
Yonemura Y, Ninomiya I, Yamaguchi A, et al. Evaluation of immunoreactivity for erbB-2 protein as a marker of poor short term prognosis in gastric cancer. Cancer Res. 1991;51:1034–1038.
Hofmann M, Stoss O, Shi D, et al. Assessment of a HER2 scoring system for gastric cancer: results from a validation study. Histopathology. 2008;52:797–805.
Kimura Y, Oki E, Yoshida A, et al. Significance of accurate human epidermal growth factor receptor-2 (HER2) evaluation as a new biomarker in gastric cancer. Anticancer Res. 2014;34:4207–4212.
Aizawa M, Nagatsuma AK, Kitada K, et al. Evaluation of HER2-based biology in 1,006 cases of gastric cancer in a Japanese population. Gastric Cancer. 2014;17:34–42.
Chao C, Hellmich MR. Gastrin, inflammation, and carcinogenesis. Curr Opin Endocrinol Diabetes Obes. 2010;17:33–39.
Wang T, Zhao L, Yang Y, et al. EGR1 is critical for gastrin-dependent upregulation of anion exchanger 2 in gastric cancer cells. FEBS J. 2013;280:174–183.
Ribeiro MM, Sarmento JA, Simoes MA, et al. Prognostic significance of Lauren and Ming classifications and other pathologic parameters in gastric carcinoma. Cancer. 1981;47:780–784.
Hiromi S, Kimio Y, Norihisa S, et al. Genetic variation in PSCA is associated with susceptibility to diffuse-type gastric cancer. Nat Genet. 2008;40:730–740.
Shan L, Ying J, Lu N. HER2 expression and relevant clinicopathological features in gastric and gastroesophageal junction adenocarcinoma in a Chinese population. Diagn Pathol. 2013;8:76.
Takaoka AS, Kakiuchi H, Itoha F, et al. Infrequent alterations of the p16 (MTS-1) gene in human gastric cancer. Tumour Biol. 1997;18:95–103.
Fu GH, Wang Y, ** YH, et al. Direct interaction and cooperative role of tumor suppressor p16 with band 3 (AE1). FEBS Lett. 2005;579:2105–2110.
Acknowledgments
In vitro drug treatment and cell count experiments were aided by Dr. Ting Wang (Pathology Center, Shanghai Jiao Tong University School of Medicine). Flow cytometry work was assisted by Dong-Jun Qing (Department of Pathophysiology, Shanghai Jiao Tong University School of Medicine). AE2 antibody was kindly provided by Professor Seth L. Alper (Beth Israel Deaconess Medical Center, Harvard Medical School). This work was supported in part by the National Natural Science Foundation of China (Nos. 81171939 and 81372637), The National Basic Research Program (973 Program, No. 2013CB910903), Key Projects in Shanghai Science & Technology Pillar Program for Biomedicine (No. 14431904700), Shanghai Science and Technology Yang Fan Funds (14YF1402300).
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Yan Cui and Shao-Bo Li have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Cui, Y., Li, SB., Peng, XC. et al. Trastuzumab Inhibits Growth of HER2-Negative Gastric Cancer Cells Through Gastrin-Initialized CCKBR Signaling. Dig Dis Sci 60, 3631–3641 (2015). https://doi.org/10.1007/s10620-015-3793-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-015-3793-7