Abstract
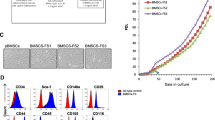
Bone marrow mesenchymal stem cells (bMSCs) are multipotent and preferred for cell therapy. However, the content of bMSCs is very low. To propagate a large number of primary bMSCs rapidly has become a prerequisite for bMSC study and application. Different methods of isolating and culturing bMSC were used and compared among groups: bMSCs of group A are isolated using direct adherence method and cultured by conventional medium changing; of group B are isolated using direct adherence method and cultured by low volume medium changing; of group C are isolated using density gradient centrifugation and cultured by conventional medium changing; of group D are isolated using density gradient centrifugation and cultured by low volume medium changing. The average population doubling time (PDT), average generation time and the cumulative cell doubling level were calculated for every group. bMSCs cultured with complete medium containing 10, 11 and 15 % FBS were allocated into group a, b and c separatedly. Cell numbers were counted everyday under a microscope, the population doubling level curve was plotted and PDT was calculated. The growth curve of bMSC in group a, b and c was made. Both density gradient centrifugation and direct adherence methods obtained relatively pure bMSCs. A larger quantity of primary bMSCs were obtained by direct adherence. bMSC proliferation was faster when cultured via the low volume medium changing method at a serum concentration of 11 % than the other methods. Isolating bMSC by direct adherence and culturing by low volume medium changing at a serum concentration of 11 % is preferential for bMSC propagation.





Similar content being viewed by others
References
Aggarwal S, Pittenger MF (2005) Human mesenchymal stem cells modulate allogeneic immune cell response. Blood 105(4):1815–1822
Bartholomew A, Sturgeon C, Siatska M et al (2002) Mesenchymal stem cues suppress lyruphocyte proliferation in vitro and prolong skin graft Surrlval in vivo. Exp Hematol 30(1):42–48
Bǒcker W, Docheva D, Prall WC et al (2008) IKK-2 is required for TNF-alpha-induced invasion and proliferation of human mesenchymal stem cells. J Mol Med 86(10):1183–1192
Cárcamo-Orive I, Tejados N, Delgado J et al (2008) ERK2 protein regulates the proliferation of human mesenchymal stem cells without affecting their mobilization and differentiation potential. Exp Cell Res 314(8):1777–1788
Chang L, Karin M (2001) Mammalian MAP kinase signalling cascades. Nature 410(6824):37–40
Cheng Z, Zhou D, Song S et al (2004) Oriented induction of umbilical cord blood and bone marrow mesenchymal stem cells into osteoblasts and comparison of the osteogenic activity. Chin J Clin Rehabil 35(8):7973–7975
Conget PA, Minguell JJ (1999) Phenotypical and functional properties of human bone marrow mesenchymal progenitor cells. J Cell Physiol 181(1):67–73
Cowles EA, Brailey LL, Gronowicz GA (2000) Integrin-mediated signaling regulates AP-1 transcription factors and proliferation in osteoblasts. J Biomed Mater Res 52(4):725–737
Dominici M, Le Blanc K, Mueller I et al (2006) Minimal criteria for defining multipotent mesenchymal stromal cells. The internatlonal society for cellular therapy Position statement. Cytotherapy 8:315–317
Erices A, Conget P, Minguell JJ (2000) Mesenchymal progenitor cells in human umbilical cord blood. Br J Haematol 109(1):235–242
Friedenstein AJ, Gorskaja JF, Kulagina NN (1976) Fibroblast precursors in normal and irradiated mouse hematopoietic organs. Exp Hematol 4:267–274
Gang EJ, Bosnakovski D, Figueiredo CA et al (2007) SSEA-4 identifies mesenchymal stem cells from bone marrow. Blood 109(4):1743–1751
Giancotti FG, Ruoslahti E (1999) Integrin signaling. Science 285(5430):1028–1032
Illario M, Cavallo AL, Monaco S et al (2005) Fibronectin-induced proliferation in thyroid cells is mediated by αvβ3 integrin through Ras/Raf-1/MEK/ERK and calcium/CaMKII signals. Clin Endocrinol Metab 90(5):2865–2873
In’t Anker PS, Scherjon SA, Kleijburg-van der Keur C et al (2003) Amniotic fluid as a novel source of mesenchymal stem cells for therapeutic transplantation. Blood 102(4):1548–1549
In’t Anker PS, Scherjon SA, Kleijburg-Van Der Keur C et al (2004) Isolation of mesenchymal stem cells of fetal or maternal origin from human placenta. Stem Cells 22(7):1338–1345
Jäger M, Feser T, Denck H et al (2005) Proliferation and osteogenic differentiation of mesenchymal stem cells cultured onto three different polymers in vitro. Ann Biomed Eng 33(10):1319–1332
Kim SM, Jung JU, Ryu JS et al (2008) Effects of gangliosides on the differentiation of human mesenchymal stem cells into osteoblasts by modulating epidermal growth factor receptors. Biochem Biophys Res Commun 371(4):866–871
Ko KS, McCulloch CA (2001) Intercellular mechanotransduction: cellular circuits that coordinate tissue responses to mechanical loading. Biochem Biophys Res Commun 285(5):1077–1083
Kobayashi-Sakamoto M, Isogai E, Hirose K et al (2008) Role of alphav integrin in osteoprotegerin-induced endothelial cell migration and proliferation. Microvasc Res 76(3):139–144
Larsen M, Artym VV, Green JA et al (2006) The matrix reorganized: extracellular matrix remodeling and integrin signaling. Curr Opin Cell Biol 18(5):463–471
Le Blanc K, Tammik L, Sundberg B, Haynesworth SE, Ringdén O (2003) Mesenchymal stem cells inhabit and stimulate mixed lymphocyte cultures and mitogenic responses independently of the major histocompatibility complex. Scand J Immumol 57:11–20
Levy O, Dvir T, Tsur-Gang O et al (2008) Signal transducer and activator of transcription 3-A key molecular switch for human mesenchymal stem cell proliferation. Int J Biochem Cell Biol 40(11):2606–2618
Li YJ, Batra NN, You L et al (2004) Oscillatory fluid flow affects human marrow stromal cell proliferation and differentiation. J Orthop Res 22(6):1283–1289
Liedert A, Kaspar D, Blakytny R et al (2006) Signal transduction pathways involved in mechanotransduction in bone cells. Biochem Biophys Res Commun 349(1):1–5
Mclntosh KR, Bartholomew A (2000) Stromal cell modulation of the immune system: a potential role for mesenchymal stem cell. Graft 3:324–328
Nicola DM, Carlo-Stella C, Magni M et al (2002) Human bone marrow stromal cells suppress T-lymphocyte proliferation induced by cellular or nonspecific mitogenic stimuli. Blood 99:3838–3843
Orlic D, Kajstura J, Chimenti S, Jakoniuk I, Anderson SM, Li B, Pickel J, McKay R, Nadal-Ginard B, Bodine DM, Leri A, Anversa P (2001) Bone marrow cells regenerate infarcted myocardium. Nature 410:701–705
Pelttari K, Steck E, Richter W (2008) The use of mesenchymal stem cells for chondrogenesis. Injury 39(suppl 1):S58–S65
Pittenger MF, Martin BJ (2004) Mesenchymal stem cells and their potential as cardiac therapeutics. Circ Res 95(1):9–20
Pittenger MF, Mackay AM, Beck SC et al (1999) Multilineage potential of adult human mesenchymal stem cells. Science 284(5411):143–147
Reger RL, Tucker AH, Wolfe MR (2008) Differentiation and characterization of human MSCs. Methods Mol Biol 449:93–107
Romanov YA, Darevskaya AN, Merzlikina NV et al (2005) Mesenchymal stem cells from human bone marrow and adipose tissue: isolation, characterization, and differentiation potentialities. Bull Exp Biol Med 140(1):138–143
Rouqer K, Fornasari B, Armenqol V et al (2007) Progenitor cell isolation from muscle derived cells based on adhesion properties. J Histochem Cytochem 55(6):607–618
Runguang L, **gfan S, Mingfa W (2007) Infection of mechanics on mesenchymal stem cells in vitro. J Clin Rehabil Tissue Eng Res 11(3):551–554
Sanchez-Ramos JR (2002) Neural cells derived from adult bone marrow and umbilical cord blood. J Neurosci Res 69(6):880–893
Satija NK, Gurudutta GU, Sharma S et al (2007) Mesenchymal stem cells: molecular targets for tissue engineering. Stem Cells Dev 16(1):7–23
Sekiya I, Larson BL, Vuoristo JT, Cui J-G, Prockop DJ (2004) Adipogenic differentiation of human adult stem cells from bone marrow stroma (MSCs). J Bone Miner Res 19(2):256–264
Sheehan DC, Hrapchak BB (1980) Theory and practice of histotechnology, 2nd edn. Mosby, St. Louis, Mo
Simmons CA, Matlis S, Thornton AJ et al (2003) Cyclic strain enhances matrix mineralization by adult human mesenchymal stem cells via the extracellular signal-regulated kinase (ERK1/2) signaling pathway. J Biomech 36(8):1087–1096
Sotiropoulou PA, Perez SA, Salagianni M et al (2006) Characterization of the optimal culture conditions for clinical scale production of human mesenchymal stem cells. Stem Cells 24(2):462–471
Theise ND, Nimmakayalu M, Gardner R, Illei PB, Morgan G, Eperman L, Henegariu O, Krause DS (2000) Liver from bone marrow in humans. Hepatology 32:11–16
Tse WT, Pendleton JD, Beyer WM et al (2003) Suppression of allogeneic T cell proliferation by human marrow stromal cells: implications in transplantation. Transplantation 75(3):389–397
Vellon L, Menendez JA, Lupu R (2006) A bidirectional “alpha(v)beta(3) integrin-ERK1/ERK2 MAPK” connection regulates the proliferation of breast cancer cells. Mol Carcinog 45(10):795–804
Wilson SH, Ljubimov AV, Morla AO et al (2003) Fibronectin fragments promote human retinal endothelial cell adhesion and proliferation and ERK activation through α5β1 integrin and PI 3-kinase. Invest Ophthalmol Vis Sci 44(4):1704–1715
Woodbury D, Schwarz EJ, Prockop DJ et al (2000) Adult rat and human bone marrow stromal cells differentiate into neurons. J Neurosci Res 61(4):364–370
Woodbury D, Reynolds K, Black IB (2002) Adult bone marrow stromal stem cells express germ Line, ectodermal, endodermal, and mesodermal genes prior to neurogenesis. J Neurosci Res 69(6):908–917
Xu W, Zhang X, Qian H et al (2004) Mesenchymal stem cells from adult human bone marrow differentiate into a cardiomyocyte phenotype in vitro. Exp Biol Med 229(7):623–631
Yang F, Yang N (2008) Comparison of two methods of in vitro isolation of adult bone marrow mesenchymal stem cells. J Clin Rehabil Tissue Eng Res 12(3):473–476
Zannettino AC, Paton S, Kortesidis A et al (2007) Human mulipotential mesenchymal/stromal stem cells are derived from a discrete subpopulation of STRO-1bright/CD34/CD45(-)/glycoPhorin-A-bone marrow cells. Haematoloqica 92(12):1707–1708
Zuk PA, Zhu M, Ashjian P et al (2002) Human adipose tissue is a source of multipotent stem cells. Mol Biol Cell 13(12):4279–4295
Acknowledgments
Authors received fund from Liaoning province high school program (2008s248).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, X., Zhang, Y. & Qi, G. Evaluation of isolation methods and culture conditions for rat bone marrow mesenchymal stem cells. Cytotechnology 65, 323–334 (2013). https://doi.org/10.1007/s10616-012-9497-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10616-012-9497-3