Abstract
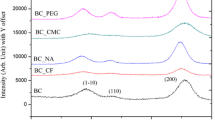
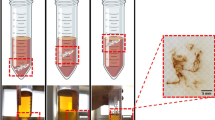
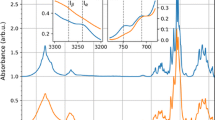
Bacterial cellulose (BC) is a promising biomaterial as well as a model system useful for investigating cellulose biosynthesis. BC produced under static cultivation condition is a hydrous pellicle consisting of an interconnected network of fibrils assembled in numerous dense layers. The mechanisms responsible for this layered BC assembly remain unknown. This study used calcofluor as a fluorescent marker to examine BC layer formation at the air/liquid interface. Layers are found to move downward into the media after formation while new layers continue to form at the air/liquid interface. Calcoflour is also known to reduce the crystallinity of cellulose, changing the mechanical properties of the formed BC microfibrils. Consecutive addition and accumulation of calcofluor in the culture medium is found to disrupt the layered assembly of BC. BC crystalllinity decreased by 22 % in the presence of 12 % calcofluor (v/v) in the medium as compared to BC produced without calcofluor. This result suggests that cellulose crystallinity and the mechanical properties which crystallinity provides to cellulose are major factors influencing the layered BC structure formed during biosynthesis.







Similar content being viewed by others
References
Bielecki S, Krystynowicz A, Turkiewicz M, Kalinowska H (2005) Bacterial cellulose. Biopolym Online. doi:10.1002/3527600035.bpol5003
Borzani W, Souza SJ (1995) Mechanism of the film thickness increasing during the bacterial production of cellulose on non-agitated liquid media. Biotechnol Lett 17:1271–1272. doi:10.1007/BF00128400
Brown MR, Willison JH, Richardson CL (1976) Cellulose biosynthesis in Acetobacter xylinum: visualization of the site of synthesis and direct measurement of the in vivo process. Proc Natl Acad Sci USA 73:4565–4569
Colvin RJ, Witter DE (1983) Congo red and calcofluor white inhibition of Acetobacter xylinum cell growth and of bacterial cellulose microfibril formation: isolation and properties of a transient, extracellular glucan related to cellulose. Protoplasma 116:34–40. doi:10.1007/BF01294228
Fang L, Catchmark JM (2014) Characterization of water-soluble exopolysaccharides from gluconacetobacter xylinus and their impacts on bacterial cellulose crystallization and ribbon assembly. Cellulose 21:3965–3978. doi:10.1007/s10570-014-0443-8
French AD (2014) Idealized powder diffraction patterns for cellulose polymorphs. Cellulose 21:885–896. doi:10.1007/s10570-013-0030-4
French DA, Cintrόn MS (2013) Cellulose polymorphy, crystallite size, and the Segal crystallinity index. Cellulose 20:583–588. doi:10.1007/s10570-012-9833-y
Gu J, Catchmark JM, Kaiser EQ, Archibald DD (2013) Quantification of cellulose nanowhiskers sulfate esterification levels. Carbohydr Polym 92:1809–1816. doi:10.1016/j.carbpol.2012.10.078
Haigler CH, Brown RM, Benziman M (1980) Calcofluor white ST alters the in vivo assembly of cellulose microfibrils. Science 210:903–906. doi:10.1126/science.7434003
Hu Y, Catchmark JM (2010a) Formation and characterization of spherelike bacterial cellulose particles produced by Acetobacter xylinum JCM 9730 strain. Biomacromolecules 11:1727–1734. doi:10.1021/bm100060v
Hu Y, Catchmark JM (2010b) Influence of 1-methylcyclopropene (1-MCP) on the production of bacterial cellulose biosynthesized by Acetobacter xylinum under the agitated culture. Lett Appl Microbiol 51:109–113. doi:10.1111/j.1472-765X.2010.02866.x
Hu Y, Catchmark JM (2011a) Integration of cellulases into bacterial cellulose: toward bioabsorbable cellulose composites. J Biomed Mater Res B 97B:114–123. doi:10.1002/jbm.b.31792
Hu Y, Catchmark JM (2011b) In vitro biodegradability and mechanical properties of bioabsorbable bacterial cellulose incorporating cellulases. Acta Biomater 7:2835–2845. doi:10.1016/j.actbio.2011.03.028
Hu Y, Catchmark JM, Vogler EA (2013) Factors impacting the formation of sphere-like bacterial cellulose particles and their biocompatibility for human osteoblast growth. Biomacromolecules 14:3444–3452. doi:10.1021/bm400744a
Hu Y, Catchmark JM, Zhu Y, Abidi N, Zhou X, Wang J, Liang N (2014) Engineering of porous bacterial cellulose toward human fibroblasts ingrowth for tissue engineering. J Mater Res 29:2682–2693. doi:10.1557/jmr.2014.315
Hu Y, Zhu Y, Zhou X, Ruan C, Pan H, Catchmark JM (2016) Bioabsorbable cellulose composites prepared by an improved mineral-binding process for bone defect repair. J Mater Chem B 4:1235–1246. doi:10.1039/C5TB02091C
Iguchi M, Yamanaka S, Budhiono A (2000) Bacterial cellulose—a masterpiece of nature’s arts. J Mater Sci 35:261–270. doi:10.1023/A:1004775229149
Jonas R, Farah LF (1998) Production and application of microbial cellulose. Polym Degrad Stab 59:101–106. doi:10.1016/S0141-3910(97)00197-3
Klemm D, Schumann D, Udhardt U, Marsch S (2001) Bacterial synthesized cellulose—artificial blood vessels for microsurgery. Prog Polym Sci 26:1561–1603. doi:10.1016/S0079-6700(01)00021-1
Mikkelsen D, Flanagan BM, Dykes GA, Gidley MJ (2009) Influence of different carbon sources on bacterial cellulose production by Gluconacetobacter xylinus strain ATCC 53524. J Appl Microbiol 107:576–583. doi:10.1111/j.1365-2672.2009.04226.x
Park S, Baker JO, Himmel ME, Parilla PA, Johnson DK (2010) Cellulose crystallinity index: measurement techniques and their impact on interpreting cellulase performance. Biotechnol Biofuels 3:1–10. doi:10.1186/1754-6834-3-10
Ruan C, Hu N, Hu Y, Jiang L, Cai Q, Wang H, Pan H, Lu WW, Wang Y (2014a) Piperazine-based polyurethane-ureas with controllable degradation as potential bone scaffolds. Polymer 55:1020–1027. doi:10.1016/j.polymer.2014.01.011
Ruan C, Hu Y, Jiang L, Cai Q, Pan H, Wang H (2014b) Tunable degradation of piperazine-based polyurethane ureas. J Appl Polym Sci 131:40527. doi:10.1002/app.40527
Tomita Y, Kondo T (2009) Influential factors to enhance the moving rate of Acetobacter xylinum due to its nanofiber secretion on oriented templates. Carbohydr Polym 77:754–759. doi:10.1016/j.carbpol.2009.02.022
Valla S, Ertesvåg H, Tonouchi N, Fjærvik E (2009) Bacterial cellulose production: biosynthesis and applications. Microbial production of biopolymers and polymer precursors: applications and perspectives. Caister Academic Press, Norfolk, pp 43–77
Wada M, Okano T, Sugiyama J (2001) Allomorphs of native crystalline cellulose I evaluated by two equatorial d-spacings. J Wood Sci 47:124–128. doi:10.1007/BF00780560
Yamanaka S, Ishihara M, Sugiyama J (2000) Structural modification of bacterial cellulose. Cellulose 7:213–225. doi:10.1023/A:1009208022957
Acknowledgments
This work was supported by the National Natural Science Foundation of China (31570967 and 31370978); the Guangdong Province Science and Technology Planning Project (2015A010105021); the Shenzhen Science and Technology Program (JCYJ20140610152828698 and CXZZ20140417113430716); the Shenzhen Peacock Program (110811003586331).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Changshun Ruan and Yongjun Zhu have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Ruan, C., Zhu, Y., Zhou, X. et al. Effect of cellulose crystallinity on bacterial cellulose assembly. Cellulose 23, 3417–3427 (2016). https://doi.org/10.1007/s10570-016-1065-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-016-1065-0