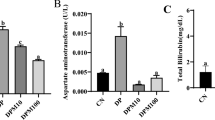
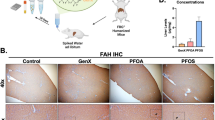
Abstract
In the present study, the toxicity of arsenic trioxide and lead acetate was assessed in adult hepatic stem cells induced in the 2-acetyl-aminofluorene/partial hepatectomy rat model. Isolated oval cells were incubated separately for 6 h with 40 μM each of arsenic trioxide and lead acetate. 3-(4,5-Dimethylthiazole-2-yl)-2,5-diphenyltetrazolium bromide assay denoted significant time-dependent cell death in arsenic and lead treated oval cells. The degree of stress imposed by these metals was evidenced by induction of heat shock protein (HSP) 70 and HSP 90. Arsenic and lead were found to trigger apoptosis as revealed by DNA ladder formation, Western blots of apoptotic factors, and reverse transcriptase polymerase chain reaction analyses of bax and bcl-2. Results clearly indicate that both arsenic and lead induced apoptosis is caspase-mediated and accompanied by extracellular signal-regulated kinase (ERK) dephosphorylation. Full-length BH3-interacting-domain death agonist expression in presence of caspase 3 inhibitor unravels a direct involvement of caspase in As and Pb induced apoptosis. Expression patterns of apoptosis inducing factor, B cell lymphoma-2 (Bcl-2) antagonist of cell death, Bcl-2-associated X protein, and Bcl2 also signify mitochondrial regulation of apoptosis effected by lead and arsenic. It is concluded that stimulation of caspase cascade and simultaneous ERK dephosphorylation are the most significant operative pathways directly associated with apoptotic signals triggered by arsenic and lead in the oval cells.







Similar content being viewed by others
Abbreviations
- AAF:
-
2-acetylaminofluorene
- AIF:
-
Apoptosis-inducing factor
- AP-1:
-
Activator protein-1
- Apaf-1:
-
Apoptotic protease activating factor-1
- Bcl-2:
-
B cell lymphoma-2
- Bax:
-
Bcl-2-associated X protein
- Bad:
-
Bcl-2 antagonist of cell death
- Bid:
-
BH3-interacting-domain death agonist
- CK-19:
-
cytokeratin 19
- DMA:
-
Dimethylarsinic acid
- DMEM:
-
Dulbecco’s minimum essential medium
- ERK:
-
Extracellular signal-regulated kinase
- GAPDH:
-
Glyceraldehyde phosphate dehydrogenase
- HBSS:
-
Hanks balanced salt solution
- HSP:
-
Heat shock protein
- MMA:
-
Monomethylarsonic acid
- MTT:
-
3-(4,5-dimethylthiazole-2-yl)-2,5-diphenyltetrazolium bromide
- NFkB:
-
Nuclear factor kB
- OV1:
-
Oval cell marker 1
- OV6:
-
Oval cell marker 6
- PH:
-
Partial hepatectomy
- RT PCR:
-
Reverse transcriptase polymerase chain reaction
- SCF:
-
Stem cell factor
- ZFL:
-
Zebra fish liver cell line
References
Adams JM. Ways of dying: multiple pathways to apoptosis. Genes Dev. 2003;17:2481–95.
Alison MR, Poulsom R, Forbes SJ. Update on hepatic stem cells. Liver 2001;21:367–73.
Ayala-Fierro F, Barber DS, Rael LT, Carter DE. In vitro tissue specificity for arsine and arsenite toxicity in the rat. Toxicol Sci. 1999;52:122–29.
Best DH, Coleman WB. Treatment with 2-AAF blocks the small hepatocyte-like progenitor cell response in retrorsine-exposed rats. J Hepatol. 2007;46:1055–63.
Bidère N, Lorenzo HK, Carmona S, Laforge M, Harper F, Dumont C, et al. Cathepsin D triggers Bax activation, resulting in selective apoptosis-inducing factor (AIF) relocation in T lymphocytes entering the early commitment phase to apoptosis. J Biol Chem. 2003;278:31401–11.
Bode AM, Dong Z. The paradox of arsenic: molecular mechanisms of cell transformation and chemotherapeutic effects. Crit Rev Oncol Hematol. 2002;42:5–24.
Boudreau RTM, Conrad DM, Hoskin DW. Apoptosis induced by protein phosphatase 2A (PP2A) inhibition in T leukemia cell is negatively regulated by PP2A—associated p38 mitogen-activated protein kinase. Cell Signal. 2007;19:139–51.
Brambilla G, Martelli A. Cytotoxicity, DNA fragmentation and DNA repair synthesis in primary human hepatocytes. In: O’Hare S, Atterwill CK, editors. In vitro toxicity testing protocols. Totowa, New Jersey: Humana; 1995. p. 59–66.
Chen N-Y, Ma W-Y, Yang CS, Dong Z. Inhibition of arsenite-induced apoptosis and AP-1 activity by epigallocatechin-3-gallate and theoflavins. J Environ Pathol Toxicol Oncol. 2000;19:287–95.
Columbano A, Ledda-Columbano GM, Coni PP, Faa G, Liguori C, Santa Cruz G, et al. Occurrence of cell death (apoptosis) during the involution of liver hyperplasia. Lab Invest. 1985;52:670–5.
Datta S, Saha DR, Ghosh D, Majumdar T, Bhattacharya S, Mazumder S. Sub-lethal concentration of arsenic interferes with the proliferation of hepatocytes and induces in vivo apoptosis in Clarias batrachus L. Comp Biochem Physiol Part C. 2007;145:339–49.
Davila JC, Cezar GG, Thiede M, Strom S, Miki T, Trosko J. Use and application of stem cells in toxicology. Toxicol Sci. 2004;79:214–23.
Dunsford HC, Sell S. Production of monoclonal antibodies to preneoplastic liver cell populations induced by chemical carcinogen in rats and transplantable Morris hepatomas. Cancer Res. 1989;49:4887–93.
Earnshaw WC, Martins LM, Kaufmann SH. Mammalian caspases: structure, activation, substrates, and functions during apoptosis. Annu Rev Biochem. 1999;68:383–424.
Fausto N. Liver regeneration: from laboratory to clinic. Liver Transplant. 2001;276:835–44.
Green DR, Kroemer G. The pathophysiology of mitochondrial cell death. Science 2004;305:626–9.
Gross A, Yin XM, Wang K, Wei MC, Jockel J, Milliman C, et al. Caspase cleaved BID targets mitochondria and is required for cytochrome c release, while BCL-XL prevents this release but not tumor necrosis factor-R1/Fas death. J Biol Chem. 1999;274:1156–63.
He ZP, Tan WQ, Tang YF, Zhang HJ, Feng MF. Activation, isolation, identification and in vitro proliferation of oval cells from adult rat livers. Cell Prolif. 2004;37:177–87.
Ikeyama S, Kusumoto K, Miyake H, Rokutan K, Tashiro S. A non-toxic heat shock protein 70 inducer, geranylgeranylacetone, suppresses apoptosis of cultured rat hepatocytes caused by hydrogen peroxide and ethanol. J Hepatol. 2001;55:53–61.
Inglis JK. Introduction to laboratory animal science and technology. 1st ed. Oxford: Pergamon; 1980. p. 37–126.
Kann S, Estes C, Reichard JF, Huang M, Sartor MA, Schwemberger S, et al. Butylhydroquinone protects cells genetically deficient in glutathione biosynthesis from arsenite-induced apoptosis without significantly changing their prooxidant status. Toxicol Sci. 2005;87:365–84.
Kapur R, Chandra S, Cooper R, McCarthy J, Williams DA. Role of p38 and ERK MAP kinase in proliferation of erythroid progenitors in response to stimulation by soluble and membrane isoforms of stem cell factor. Blood 2002;100:1287–93.
Laemmli UK. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970;227:680–5.
Lanza R. Essentials of stem cell biology. USA: Elsevier; 2006.
Li GC, Werb Z. Correlation between the synthesis of heat shock proteins and the development of thermotolerance in Chinese hamster fibroblasts. Proc Natl Acad Sci USA. 1982;79:3218–22.
Li H, Zhu H, Xu CJ, Yuan J. Cleavage of BID by caspase 8 mediates the mitochondrial damage in the Fas pathway of apoptosis. Cell 1998;94:491–501.
Li L, Luo X, Wang X. Endonuclease G is an apoptotic DNase when released from mitochondria. Nature 2001;412:95–9.
Liedtke C, Trautwein C. The role of JNK2 in toxic liver injury. J Hepatol. 2006;45:762–4.
Lin YW, Chuang SM, Yang JL. Persistent activation of ERK1/2 by lead acetate increases nucleotide excision repair synthesis and confers anti-cytotoxicity and anti-mutagenicity. Carcinogenesis 2003;24:53–61.
Lindquist S, Craig EA. The heat shock proteins. Annu Rev Genet. 1998;22:631–37.
Liu ZM, Huang HS. As2O3-induced c-Src/EGFR/ERK signaling is via Sp1 binding sites to stimulate p21WAF1/CIP1 expression in human epidermoid carcinoma A431 cells. Cell Signal. 2005;18:244–55.
Look AT. Arsenic and apoptosis in the treatment of acute promyelocytic leukemia. J Natl Cancer Inst. 1998;90:86–8.
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. Measurement of protein with the Folin phenol reagent. J Biol Chem. 1951;193:265–75.
Ludwig S, Hoffmeyer A, Goebeler M, Kilian K, Hafner H, Neufeld B, et al. The stress inducer arsenite activates mitogen-activated protein kinases extracellular signal-regulated kinases 1 and 2 via a MAPK kinase 6/p38-dependent pathway. J Biol Chem. 1998;273:1917–22.
Luo X, Budiharjo H, Zou H, Slauhter C, Wang X. Bid, a Bcl2 interacting protein, mediates cytochrome c release from mitochondria in response to activation of cell surface death receptors. Cell 1998;94:481–90.
Marceau N. Biology of disease. Cell lineage and differentiation programs in epidermal; urothelial and hepatic tissues and their neoplasms. Lab Invest. 1990;63:4–20.
Milton AG, Zalewski PD, Ratnaike RN. Zinc protects against arsenic-induced apoptosis in a neuronal cell line, measured by DEVD-caspase activity. Biometals 2004;17:707–13.
Mosser DD, Caron AW, Bourget L, Larose CD, Massie B. Role of the human heat shock protein hsp70 in protection against stress-induced apoptosis. Mol Cell Bio. 1997;17:5317–27.
Mosser DD, Caron AW, Bourget L, Meriin AB, Sherman AY, Morimoto RI, et al. The chaperone function of hsp70 is required for protection against stress-induced apoptosis. Mol Cell Biol. 2000;20:7146–59.
Nollen EA, Morimoto RI. Chaperoning signaling pathways: molecular chaperones as stress-sensing ‘heat shock’ proteins. J Cell Sci. 2002;115:2809–16.
Parsell DA, Lindquist S. The function of heat-shock proteins in stress tolerance: degradation and reactivation of damaged proteins. Annu Rev Genet. 1993;27:437–96.
Petit PX, Susin SA, Zamzami N, Mignotte B, Kroemer G. Mitochondria and programmed cell death: back to the future. FEBS Lett. 1996;396:7–13.
Pulido MD, Parrish AR. Metal-induced apoptosis: mechanisms. Mutat Res. 2003;533:227–41.
Qin AL, Zhou XQ, Zhang W, Yu H, **e Q. Characterization and enrichment of hepatic progenitor cells in adult rat liver. World J Gastroenterol. 2004;10:1480–6.
Robinson MJ, Cobb MH. Mitogen-activated protein kinase pathways. Curr Opin Cell Biol. 1997;9:180–86.
Roy S, Bhattacharya S. Arsenic-induced histopathology and synthesis of stress proteins in liver and kidney of Channa punctatus. Ecotox Environ Saf. 2006;65:218–29.
Sambrook J, Fritsch EF, Maniatis T. Molecular cloning—a laboratory manual. 2nd ed. Cold Spring Harbor, New York: Cold Spring Harbor Laboratory; 1989.
Seok SH, Baek MW, Lee HY, Kim DJ, Na YR, Noh YR, et al. Arsenite-induced apoptosis is prevented by antioxidants in zebra fish liver cell line. Toxicol in Vitro. 2007;21:870–7.
Shimano K, Satake M, Okaya A, Kitanaka J, Kitanaka N, Takemura M, et al. Hepatic oval cell have the side population phenotype defined by expression of ATP binding cassette transporter ABCG2/BCRP1. American J Pathol. 2003;163(1):3–9.
Snedecor GW, Cochran WG. Statistical methods. 6th ed. Iowa: The Iowa State University Press; 1967.
Subjeck JR, Sciandra JJ, Johnson RJ. Heat shock proteins and thermotolerance; a comparison of induction kinetics. Br J Radiol. 1982;55:579–84.
Sun X-M, MacFarlane M, Zhuang J, Wolf BB, Green DR, Cohen GM. Distinct caspase cascade are initiated in receptor-mediated and chemical-induced apoptosis. J Biol Chem. 1999;274:5053–60.
Supino R. MTT assays. In: O’Hare S, Atterwill CK, editors. In vitro toxicity testing protocols. Totowa, New Jersey: Humana; 1995. p. 137–49.
Trosko JE. Human stem cells as targets for the aging and diseases of aging processes. Med Hypotheses. 2003;60:439–47.
Tsujimoto Y, Shimizu S. Bcl-2 family: life-or-death switch. FEBS Lett. 2001;466:6–10.
Wang X. The expanding role of mitochondria in apoptosis. Genes Dev. 2002;15:2922–33.
Wang K, Yin XM, Chao DT, Milliman CL, Korsmeyer SJ. BID: a novel BH3 domain-only death agonist. Genes Dev. 1996;10:2859–69.
Waskiewicz AJ, Cooper JA. Mitogen and stress response pathways: MAP kinase cascades and phosphatase regulation in mammals and yeast. Curr Opin Cell Biol. 1995;7:798–805.
Yin XM, Wang K, Gross A, Zhao Y, Zinkel S, Klocke B, et al. Bid-deficient mice are resistant to Fas-induced hepatocellular apoptosis. Nature 1999;400:886–91.
Acknowledgements
SB, SM, and SR gratefully acknowledge the Council of Scientific and Industrial Research for project grant (Project No. 37(1179)/04/EMR-II) and the CAS (UGC) grants to the department which enabled the present study. AR and SA are thankful for the UGC Research Fellowships. Authors are expressing their sincere gratitude to Professor Kaoru Kubokawa, Ocean Research Institute, University of Tokyo, Japan for providing Actinase-E for the present study. Authors also express their sincere gratitude to Mr Hemanta Yadav for his technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Agarwal, S., Roy, S., Ray, A. et al. Arsenic trioxide and lead acetate induce apoptosis in adult rat hepatic stem cells. Cell Biol Toxicol 25, 403–413 (2009). https://doi.org/10.1007/s10565-008-9094-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-008-9094-6